Abstract
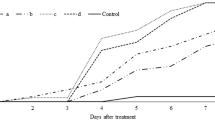
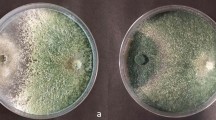
The mechanism of biocontrol of brown rot in stone fruit by Penicillium frequentans Westling (Pf909) was investigated using in vitro and in vivo growth assays and a benomyl-resistant strain of Monilinia fructicola (G Winter) Honey (Mf3C). For the in vitro assays, Pf909 and Mf3C conidia were suspended in Czapek-Dox broth, which was amended or not amended with a skin extract of mature peaches. The growth and germination of Pf909 and Mf3C conidia were determined by counting the number of colony-forming units on potato dextrose agar plates, which were amended or not amended with 0.5 g ml−1 benomyl. In some of the assays, germinated Pf909 conidia were used before their exposure to Mf3C conidia. For the in vivo assays, healthy cherries were inoculated with Mf3C conidia before and after applying Pf909 conidia on the cherry surface and the incidence of brown rot was recorded for seven days. Since we found that Pf909 conidia compete with Mf3C conidia for space and nutrients in the different assays, we concluded that competition is the probable primary mechanism of biocontrol of Pf909.




Similar content being viewed by others
References
Abdee SR, Pfender WF, Hartnett DC (1990) Competition between Pyrenophora tritici-repentis and Septoria nodorum in the wheat leaf as measured with de Wit replacement series. Phytopathology 80:1177–1182
Blakeman JP, Fokkema NJ (1982) Potential for biological control of plant diseases on the phylloplane. Ann Rev Phytopathology 20:167–190
Brodie IDS, Blakeman JP (1976) Competition for exogenous substrates in vitro by leaf surface micro-organisms and germination of conidia of Botrytis cinerea. Physiol Plant Pathol 9:227–239
Campbell CL, Madden LV (1990) Introduction to plant disease epidemiology. Wiley-Interscience, New York
De Cal A, M-Sagasta E, Melgarejo P (1988) Antifungal substances produced by Penicillium frequentans and their relationship to the biocontrol of Monilinia laxa. Phytopathology 78:888–893
De Cal A, Mateo-Sagasta E, Melgarejo P (1990) Biological control of peach twig blight (Monilinia laxa) with Penicillium frequentans. Plant Pathol 39:612–618
De Cal A, Pascual S, Melgarejo P (1994) In vitro studies on the effects of fungicides on beneficial fungi of peach twig mycoflora. Mycopathologia 126:15–20
De Cal A, Larena I, Guijarro B, Melgarejo P (2002) Solid state fermentation to produce conidia of Penicillium frequentans, a biocontrol agent against brown rot on stone fruits. Biocontrol Sci Technol 12:715–725
De Cal A, Larena I, Guijarro B, Melgarejo P (2012) Use of biofungicides for controlling plant diseases to improve food availability. Agriculture 2:109–124
Doan HK, Leveau JHJ (2015) Artificial surfaces in phyllosphere microbiology. Phytopathology 105:1036–1042
Guijarro B, Larena I, Melgarejo P, De Cal A (2006) Effect of drying on conidial viability of Penicilllium frequentans, a biological control agent against peach brown rot disease caused by Monilinia spp. Biocontrol Sci Tecnol 16:257–269
Guijarro B, Melgarejo P, Torres R, Lamarca N, Usall J, De Cal A (2007) Effect of different biological formulations of Penicillium frequentans on brown rot of peach. Biol Control 42:86–96
Guijarro B, Melgarejo P, Torres R, Lamarca N, Usall J, De Cal A (2008) Penicillium frequentans population dynamics on peach surfaces after its application against brown rot in orchards. J Appl Microbiol 104:659–671
Hjeljord LG, Tronsmo A (2003) Effect of germination initiation on competitive capacity of Trichoderma atroviride P1 conidia. Phytopathology 93:1593–1598
Johnson KB, Dileone JA (1999) Effect of antibiosis on antagonist dose-plant disease response relationships for the biological control of crown gall of tomato and cherry. Phytopathology 89:974–980
Köhl J, Lombaers-van der Plas CH, Molhoek WML, Kessel GJT, Goossen-van der Geijn HM (1999) Competitive ability of the antagonist Ulocladium atrum at temperatures favourable for Botrytis spp. development. BioControl 44:329–346
Larkin RP, Fravel DR (1999) Mechanisms of action and dose response relationships governing biological control of Fusarium wilt of tomato by nonpathogenic Fusarium spp. Phytopathol 89:1152–1161
Martinuz A, Schouten A, Sikora RA (2012) Systemically induced resistance and microbial competitive exclusion: implications on biological control. Phytopathology 102:260–266
Melgarejo P, M-Sagasta E (1984) Fungal antagonism in relation to peaches. In: Woodbine M (ed) Antimicrobials and agriculture. Butterworths, London, pp 127–136
Melgarejo P, Carrillo R, Sagasta EM (1985) Mycoflora of peach twigs and flowers and its possible significance in biological control of Monilinia laxa. Trans Br Mycol Soc 85:313–317
Melgarejo P, Carrillo R, Sagasta EM (1986) Potential for biological control of Monilinia laxa in peach twigs. Crop Prot 5:422–426
Montesinos E, Bonaterra A (1996) Dose-response models in biological control of plant pathogen: an empirical verification. Phytopathology 86:464–472
Newton MR, Kinkel LL, Leonard KJ (1998) Determinants of density- and frequency-dependent fitness in competing plant pathogens ecology and population biology. Phytopathology 88:45–51
Pal KK, Gardener BM (2006) Biological control of plant pathogens. Plant Health Instr. doi:10.1094/PHI-A-2006-1117-02
Sauer DB, Burroughs R (1986) Disinfection of seed surfaces with sodium hypochlorite. Phytopathology 76:745–749
Shams-Ghahfarokhi M, Kalantari S, Razzaghi-Abyaneh M (2013) Terrestrial bacteria from agricultural soils: versatile weapons against aflatoxigenic fungi. In: Razzaghi-Abyaneh M (ed) Aflatoxins – recent advances and future prospects. InTech, Rijeka, pp 23–40
Sikora RA, Schäfer K, Dababat AA (2007) Modes of action associated with microbially induced in planta suppression of plant-parasitic nematodes. Australas Plant Pathol 36(2):124–134
Smilanick JL, Denis-Arrue R, Bosch JR, Gonzalez AR, Henson D, Janisiewicz WJ (1993) Control of postharvest brown rot of nectarines and peaches by Pseudomonas species. Crop Prot 12:513–520
Snedecor GW, Cochran WG (1980) Statistical methods, 7th edn. Iowa State University Press, Ames
Spadaro D, Gullino ML (2004) State of the art and future prospects of the biological control of postharvest fruit diseases. Int J Food Microbiol 91:185–194
Villarino M, Melgarejo P, De Cal A (2016) Growth and aggressiveness factors affecting Monilinia spp. survival peaches. Int J Food Microbiol 227:6–12
Xu XM, Jeger MJ (2013) Combined use of two biocontrol agents with different biocontrol mechanisms most likely results in less than expected efficacy in controlling foliar pathogens under fluctuating conditions: a modeling study. Phytopathology 103:108–116
Acknowledgements
This work was carried out with financial support from BIOCOMES project (EU FP7 612713). We wish to thank to Y. Herranz and R. Castillo for technical support. The authors would also like to acknowledge Dr. Arieh Bomzon, Consul Write (http://www.consulwrite.com).
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Monica Hofte.
Rights and permissions
About this article
Cite this article
Guijarro, B., Hernandez-Escribano, L., Larena, I. et al. Competition is the mechanism of biocontrol of brown rot in stone fruit by Penicillium frequentans . BioControl 62, 557–566 (2017). https://doi.org/10.1007/s10526-017-9808-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10526-017-9808-x