Abstract
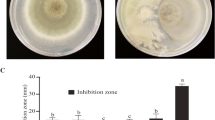
Bacterial endophytes offer control against many diseases of crop plants as potential biocontrol agents. Antagonistic bacterial endophytes acting against Phytophthora meadii have been screened from leaf, petiole and root tissues of Hevea brasiliensis. Six bacterial endophytes could exhibit more than 50 % inhibition of P. meadii, among which EIL-2, from disease-free zones showed a maximum of 62.5 % inhibition. The isolate EIL-2 was characterized as Alcaligenes sp. and the other isolates were identified as Pseudomonas aeruginosa. 16S rDNA sequence analysis showed that there existed genetic variation among the five isolates of P. aeruginosa from different tissues of the plant indicating the tissue type adaptation of the isolates. Dual culture technique with endophyte EIL-2 completely arrested the growth of P. meadii when inoculated prior to pathogen. The bioassay with EIL-2 in H. brasiliensis clones, RRII 105 showed 43 % reduction of lesion size on infected leaves whereas in RRIM 600 it was only 30 %.


Similar content being viewed by others
References
Alexander RM, Richard AS (2009) Biological control of Radopholus similis in banana by combined application of the mutualistic endophyte Fusarium oxysporum strain 162, the egg pathogen Paecilomyces lilacinus strain 251 and the antagonistic bacteria Bacillus firmus. BioControl 54:263–272
Aravind R, Kumar A, Eapen SJ, Ramana KV (2009) Endophytic bacterial flora in root and stem tissues of black pepper (Piper nigrum L.) genotype: isolation, identification and evaluation against Phytophthora capsici. Lett Appl Microbiol 48:64–67
Arnold AE, Majia LC, Kyllo D, Rojas EI, Maynard Z, Robbins N, Herre A (2003) Fungal endophytes limit pathogen damage in a tropical tree. Proc Natl Acad Sci USA 100:15649–15654
Ashplant HT (1928) Bordeaux and burgundy spraying mixtures. Scientific Department Bulletin, United Planter’s Association of South India, Coonoor, TamilNadu, India
Benhamou N, Kolepper JW, Quadt- Hallman A, Tuzun S (1996) Induction of defence related ultrastuctural modifications in pea root tissues inoculated with endophytic bacteria. Plant Physiol 112:919–929
Brooks DS, Gonzalez CF, Appel DN, Filer TH (1994) Evaluation of endophytic bacteria as potential biological control agents for oak wilt. Biol Control 4:373–381
Brown AE, Soepena H (1994) Pathogenicity of Colletotrichum acutatum and C. gloeosporioides on Hevea spp. Mycol Res 98:264–266
Cactano-Anolles G, Faueluken G, Beber WD (1993) Optimizations of surface sterilization for legume seed. Crop Sci 87:561–568
Cho KM, Hong SY, Lee SM, Kim YH, Kahng GG, Lim YP, Kim H, Yun HD (2007) Endophytic bacterial communities in ginseng and their antifungal activity against pathogens. Microb Ecol 54:341–351
Conn VM, Franco CMM (2004) Analysis of the endophytic actinobacterial population in the roots of wheat (Triticum aestivum L.) by terminal restriction fragment length polymorphism and sequencing of 16S rRNA clones. Appl Environ Microbiol 70:1787–1794
Coombs JT, Michelsen PP, Franco CMM (2004) Evaluation of endophytic actinobacteria as antagonists of Gaeumannomyces graminis var. tritici in wheat. Biol Control 29:359–366
Delaney SM, Mavrodi DV, Bonsall RF, Thomashow LS (2001) phzO, a gene for biosynthesis of 2-hydroxylated phenazine compounds in Pseudomonas aureofaciens 30–84. J Bacteriol 183:318–327
Felsenstein J (1985) Confidence limits on phylogenies: an approach using the bootstrap. Evolution 39:783–791
Fravel DR (1988) Role of antibiosis in the biocontrol of plant diseases. Annu Rev Phytopathol 26:75–91
Galindo JJ (1992) Prospects for biological control of cacao. In: Keane PJ, Putter CA (eds) Cocoa pest and disease management in Southeast Asia and Australasia. FAO Plant Production and Protection Paper, Rome, Italy pp 31–36
He RL, Wang GP, Liu XH, Zhang CL, Lin FC (2009) Antagonistic bioactivity of an endophytic bacterium isolated from Epimedium brevicornu Maxim. Afr J Biotechnol 8:191–195
Jacob CK, Edathil TT, Idicula SP, Jayarathnam K (1989) Effect of abnormal leaf fall disease caused by Phytophthora sp. on the yield of rubber tree. Indian J Nat Rubber Res 2:77–80
Jacobs MJ, Bugbee WM, Gabrielson DA (1985) Enumeration, location, and characterization of endophytic bacteria within sugar beet roots. Can J Microbiol 63:1262–1265
Jayaraman J, Parthasarathi T, Radhakrishnan NV (2007) Characterization of a Pseudomonas fluorescens strain from tomato rhizosphere and its use for integrated management of tomato damping-off. BioControl 52:683–702
Kim BS, Moon SS, Hwang BK (1999) Isolation, identification and antifungal activity of a macrolide antibiotic, oligomycin A, produced by Streptomyces libani. Can J Bot 77:850–858
Kwon SW, Go SJ, Kang HW, Ryu JC, Jo JK (1997) Phylogenetic analysis of Erwinia species based on 16S rRNA gene sequences. Int J Syst Bacteriol 47:1061–1067
Lamb TG, Tonkyn DW, Kluepfel DA (1996) Movement of Pseudomonas aureofaciens from the rhizosphere to aerial plant tissue. Can J Microbiol 42:1112–1120
Lingfei X, Yanmin D (2012) Effects of yeast antagonist in combination with UV-C treatment on postharvest diseases of pear fruit. BioControl 57:451–461
Lodewyckx C, Vangronsveld J, Porteous F, Moore ERB, Taghavi S, Mezgeay M, der Lelie D (2002) Endophytic bacteria and their potential applications. Crit Rev Plant Sci 21:583–606
Mclnroy JA, Kloepper JW (1995) Population dynamics of endophytic bacteria in field-grown sweet corn and cotton. Can J Microbiol 41:895–901
M’piga P, Bélanger RR, Paulitz TC, Benhamou N (1997) Increased resistance to Fusarium oxysporum f. sp. radicis-lycopersici in tomato plants treated with the endophytic bacterium Pseudomonas fluorescens strain 63–28. Physiol Mol Plant Path 50:301–320
Nielsen MN, Serensen J, Fels J, Pedersen HC (1998) Secondary metabolite- and endochitinase-dependent antagonism toward plant-pathogenic microfungi of Pseudomonas fluorescens isolates from sugar beet rhizosphere. Appl Environ Microbiol 64:3563–3569
Pirttilä AM, Pospiech H, lakkanen H, Myllyalä R, Pospiech H, Hohtola A (2005) Seasonal variations in location and population structure of endophytes in buds of Scots pine. Tree Physiol 25:289–297
Pleban S, Cherian L, Chet I (1997) Chitinolytic activity of an endophytic strain of Bacillus cereus. Lett Appl Microbiol 25:284–288
Quadt-Hallman A, Benhamou N, Kloepper JW (1997) Bacterial endophytes in cotton: mechanisms of entering the plant. Can J Microbiol 43:557–582
Rademaker JLW, Louws FJ, de Bruijn FJ (1998) Characterization of the diversity of ecologically important microbes by rep-PCR genomic fingerprinting. In: Akkermans ADL, van Elsas JD, de Bruijn FJ (eds) Molecular microbial ecology manual. Kluwer Academic, Dordrecht, The Netherland pp 1–27
Sandhiya GS, Sugitha TCK, Balachandar D, Kumar K (2005) Encophytic colonization and in planta nitrogen and nitrogen fixation by a diazatrophic Serratia sp. in rice. Indian J Exp Biol 43:802–807
Sneath PHA, Sokal RR (1973) Numerical taxonomy. Freeman, San Francisco, USA
Sturz AV, Christie BR, Matheson BG (1998) Association of bacterial endophyte populations from red clover and potato crops with potential for beneficial allelopathy. Can J Microbiol 44:162–167
Sturz AV, Chiristie BR, Matheson BG, Arsenault WJ, Buchanan NA (1999) Endophytic bacterial communities in the periderm of potato tubers and their potential to improve resistance to soil born plant pathogens. Plant Pathol 48:360–369
Sturz AV, Christie BR, Novak J (2000) Bacterial endophytes: potential role in developing sustainable systems of crop production. Crit Rev Plant Sci 19:1–30
Sun L, Qiu F, Zhang X, Dai X, Dong X, Song W (2008) Endophytic bacterial diversity in rice (Oryza sativa L.) roots estimated by 16S rDNA sequence analysis. Microb Ecol 55:415–424
Suseela BR, Kumar A (2008) Effect of rhizobacteria on Phytophthora meadii, Fusariumoxysporum f.sp. vanillae and Colletotrichum vanillaeinfecting vanilla. J Biol Control 22:33–41
Tamura K, Dudley J, Nei M, Kumar S (2007) MEGA4: molecular evolutionary genetics analysis (MEGA) software version 4.0. Mol Biol Evol 24:1596–1599
Valerie G, Carole M, Hani A, Russell JT (2005) Antagonist microorganisms with the ability to control Pythium damping-off of tomato seeds in Rockwool. BioControl 50:771–786
Whipps JM (1997) Developments in the biological control of soil-borne plant pathogens. Adv Bot Res 26:1–134
Whitesides SK, Spottas RA (1991) Frequency, distribution and characteristics of endophytic Pseudomonas syringae in pear trees. Phytopathology 81:453–457
Yin XT, Xu LN, Xu L, Fan SS, Liu ZU, Zhang XY (2011) Evaluation of the efficacy of endophytic Bacillus amyloliquefaciens against Botryosphaeria dothidea and other phytopathogenic microorganisms. Afr J Microbiol Res 5:340–345
You CB, Zhou FY (1989) Non-nodular endorhizospheric nitrogen fixation in wetland rice. Can J Microbiol 35:403–408
Acknowledgments
The authors are highly thankful for the facilities provided at Rubber Research Institute of India and Mahatma Gandhi University, Kottayam, India.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Monica Hofte
Rights and permissions
About this article
Cite this article
Abraham, A., Philip, S., Kuruvilla Jacob, C. et al. Novel bacterial endophytes from Hevea brasiliensis as biocontrol agent against Phytophthora leaf fall disease. BioControl 58, 675–684 (2013). https://doi.org/10.1007/s10526-013-9516-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10526-013-9516-0