Abstract
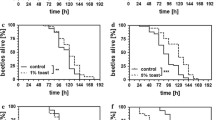
Mitochondria are suggested to play a central role in ageing and evolution of longevity. Gradual decline in mitochondrial function during ageing and concomitant increase in production of reactive oxygen species (ROS) leads to oxidative damage of macromolecules and impairment of ATP synthesis. To assess relationship between ageing and oxidative stress resistance we exposed different longevity lines of the seed beetle (Acanthoscelides obtectus) to four concentrations of tebufenpyrad, mitochondrial complex I inhibitor. Complex I is one of main sites of ROS production during normal respiration and its inhibition elevates oxidative stress. Our results showed that 24 h of exposure to tebufenpyrad decreased survival and post-stress longevity due to increased baseline mortality. Higher resistance was recorded in beetles from lines selected for late reproduction and extended longevity (L) than in early reproducing beetles (E). Also, females were more resistant than males. Since complex I is under dual genetic control, our second aim was to disentangle relative contribution of nuclear and mitochondrial genes to the variation in longevity. We used crossed combinations of distinct mitochondrial and nuclear genotypes (E × L, L × E) and compared them to control hybrids where mitochondrial genome was “transplanted” onto the original background (E × E, L × L). Our study revealed significant effect of nucleus, i.e. higher survival and post-stress longevity in beetles harbouring L nucleus. Mitochondrion effect was significant only within L nuclear background where E mitochondrion gave advantage.




Similar content being viewed by others
References
Andreyev AYu, Kushnareva YuE, Starkov AA (2005) Mitochondrial metabolism of reactive oxygen species. Biochemistry (Moscow) 70:200–214
Arking R, Buck S, Novoseltev VN, Hwangbo D-S, Lane M (2002) Genomic plasticity, energy allocations, and the extended longevity phenotypes of Drosophila. Ageing Res Rev 1:209–228
Arnqvist G, Dowling DK, Eady P, Gay L, Tregenza T, Tuda M, Hosken DJ (2010) Genetic architecture of metabolic rate: environment specific epistasis between mitochondrial and nuclear genes in an insect. Evolution 64:3354–3363
Ballard JWO, Whitlock MC (2004) The incomplete natural history of mitochondria. Mol Ecol 13:729–744
Blier PU, Dufresne F, Burton RS (2001) Natural selection and the evolution of mtDNA-encoded peptides: evidence for intergenomic co-adaptation. Trends Genet 17:400–406
Bokov A, Chaudhuri A, Richardson A (2004) The role of oxidative damage and stress in aging. Mech Ageing Dev 125:811–826
Brand MD, Affourtit C, Esteves TC, Green K, Lambert AJ, Miwa S, Pakay JL, Parker N (2004) Mitochondrial superoxide: production, biological effects, and activation of uncoupling proteins. Free Radical Bio Med 37:755–767
Bratić I, Trifunović A (2010) Mitochondrial energy metabolism and ageing. Biochim Biophys Acta 1797:961–967
Budovsky A, Abramovich A, Cohen R, Chalifa-Caspi V, Fraifel V (2007) Longevity network: construction and implications. Mech Ageing Dev 128:117–124
Camus MF, Clancy DJ, Dowling DK (2012) Mitochondria, maternal inheritance and male aging. Curr Biol 22:1717–1721
Copeland JM, Cho J, Lo T Jr, Hur JH, Bahadorani S, Arabyan T, Rabie J, Soh J, David W, Walker DW (2009) Extension of Drosophila life span by RNAi of the mitochondrial respiratory chain. Curr Biol 19:1591–1598
Curtis C, Landis GM, Folk D, Wehr NB, Hoe N, Waskar M, Abdueva D, Skvortsov D, Ford D, Luu A, Badrinath A, Levine RL, Bradley TJ, Tavaré S, Tower J (2007) Transcriptional profiling of MnSOD-mediated lifespan extension in Drosophila reveals a species-general network of aging and metabolic genes. Genome Biol 8:R262
Das J (2006) The role of mitochondrial respiration in physiological and evolutionary adaptation. BioEssays 28:890–901
Degli Esposti M (1998) Inhibitors of NADH-ubiquinone reductase: an overview. Biochim Biophys Acta 1364:222–235
Djawdan M, Sugiyama TT, Schlaeger LK, Bradley TJ, Rose MR (1996) Metabolic aspects of the trade-off between fecundity and longevity in Drosophila melanogaster. Physiol Zool 69:1176–1195
Dowling DK, Simmons LW (2009) Reactive oxygen species as universal constraints in life-history evolution. Proc R Soc 276B:1737–1745
Dowling DK, Abiega KC, Arnqvist G (2007) Temperature-specific outcomes of cytoplasmic-nuclear interactions on egg-to-adult development time in seed beetles. Evolution 61:194–201
Dowling DK, Friberg U, Lindell J (2008) Evolutionary implications of non-neutral mitochondrial genetic variation. Trends Ecol Evol 23:546–554
Ellison CK, Burton RS (2006) Disruption of mitochondrial function in interpopulation hybrids of Tigriopus californicus. Evolution 60:1382–1391
Finkel T, Holbrook NJ (2000) Oxidants, oxidative stress and the biology of ageing. Nature 408:239–247
Gemmell NJ, Metcalf VJ, Allendorf FW (2004) Mother’s curse: the effect of mtDNA on individual fitness and population viability. Trends Ecol Evol 19:238–244
Harman D (1956) Aging: a theory based on free radical radiation chemistry. J Gerontol 11:298–300
Harman D (1972) The biologic clock: the mitochondria? J Am Geriatrics Soc 20:145–147
Hirst J, Carroll J, Fearnley IM, Shannon RJ, Walker JE (2003) The nuclear encoded subunits of complex I from bovine heart mitochondria. Biochim Biophys Acta 1604:135–150
Innocenti P, Morrow EH, Dowling DK (2011) Experimental evidence supports a sex-specific selective sieve in mitochondrial genome evolution. Science 332:845–848
Jacobs HT (2003) The mitochondrial theory of aging: dead or alive? Aging Cell 2:11–17
James AC, Ballard JWO (2003) Mitochondrial genotype affects fitness in Drosophila simulans. Genetics 164:187–194
Katewa SD, Ballard WO (2007) Sympatric Drosophila simulans flies with distinct mtDNA show age related differences in mitochondrial metabolism. Insect Biochem Mol Biol 37:923–932
Kim H-J, Morrow G, Westwood JT, Michaud S, Tanguay RM (2010) Gene expression profiling implicates OXPHOS complexes in lifespan extension of flies over-expressing a small mitochondrial chaperone, Hsp22. Exp Gerontol 45:611–620
Kirkwood TBL, Kowald A (2012) The free-radical theory of ageing—older, wiser and still alive. BioEssays 34:692–700
Krementsova AV, Roshina NV, Tsybul’ko EA, Rybina OY, Symonenko AV, Pasyukova EG (2012) Reproducible effects of the mitochondria-targeted plastoquinone derivative SkQ1 on Drosophila melanogaster lifespan under different experimental scenarios. Biogerontology 13:595–607
Kuang J, Ebert PR (2012) The failure to extend lifespan via disruption of complex II is linked to preservation of dynamic control of energy metabolism. Mitochondrion 12:280–287
Kuether K, Arking R (1999) Drosophila selected for extended longevity are more sensitive to heat shock. Age 22:175–180
Lambert AJ, Buckingham JA, Boysen HM, Brand MD (2010) Low complex I content explains the low hydrogen peroxide production rate of heart mitochondria from the long-lived pigeon, Columba livia. Aging Cell 9:78–91
Lane N (2011) Mitonuclear match: optimizing fitness and fertility over generations drives ageing within generations. BioEssays 33:860–869
Lazarević J, Tucić N, Šešlija Jovanović D, Večera J, Kodrik D (2012) The effects of selection for early and late reproduction on metabolite pools in Acanthoscelides obtectus Say. Insect Sci 19:303–314
Lazarević J, Đorđević M, Stojković B, Tucić N (2013a) Resistance to prooxidant agent paraquat in the short- and long-lived lines of the seed beetle (Acanthoscelides obtectus). Biogerontology 14:141–152
Lazarević J, Stojković B, Tucić N (2013b) Sexual dimorphism in insect longevity: insights from experimental evolution. In: Geldani RM, Davin MA (eds) Sexual selection: evolutionary perspectives, mating strategies and long-term effects on genetic variation. Nova Science Publisher, New York, pp 1–44
Le Bourg É (2009) Hormesis, aging and longevity. Biochim Biophys Acta 1790:1030–1039
Min KJ, Tatar M (2006) Restriction of amino acids extends lifespan in Drosophila melanogaster. Mech Ageing Dev 127:643–646
Miquel J, Economos AC, Fleming J, Johnson JE (1980) Mitochondrial role in cell aging. Exp Gerontol 15:575–591
Mohammadi-Bardbori A, Ghazi-Khansari M (2008) Alternative electron acceptors: proposed mechanism of paraquat mitochondrial toxicity. Environ Toxicol Phar 26:1–5
Morrow G, Tanguay RM (2008) Mitochondria and ageing in Drosophila. Biotechnol J 3:728–739
O’Neill SL, Giordano R, Colbert AM, Karr TL, Robertson HM (1992) 16S rRNA phylogenetic analysis of the bacterial endosymbionts associated with cytoplasmic incompatibility in insects. Proc Natl Acad Sci 89:2699–2702
Pérez VI, Bokov A, Van Remmen H, Mele J, Ran Q, Ikeno Y, Richardson A (2009) Is the oxidative stress theory of aging dead? Biochim Biophys Acta 179:1005–1014
Pletcher SD (1999) Model fitting and hypothesis testing for age-specific mortality data. J Evol Biol 12:430–439
Prasad NG, Joshi A (2003) What have two decades of laboratory life-history evolution studies on Drosophila melanogaster taught us? J Genet 82:45–46
Pujol C, Bratić-Hench I, Šumakovic M, Hench J, Mourier A, Baumann L, Pavlenko V, Trifunović A (2013) Succinate dehydrogenase upregulation destabilize complex I and limits the lifespan of gas-1 mutant. PLoS One 8:e59493
Rand DM, Haney RA, Fry AJ (2004) Cytonuclear coevolution: the genomics of cooperation. Trends Ecol Evol 19:645–653
Rand DM, Fry A, Sheldahl L (2006) Nuclear-mitochondrial epistasis and Drosophila aging: introgression of Drosophila simulans mtDNA modifies longevity in D. melanogaster nuclear backgrounds. Genetics 172:329–341
Rattan SIS (2012) Biogerontology: from here to where? The Lord Cohen Medal Lecture—2011. Biogerontology 13:83–91
Ristow M, Zarse K (2010) How increased oxidative stress promotes longevity and metabolic health: the concept of mitochondrial hormesis (mitohormesis). Exp Gerontol 45:410–418
Sanz A, Fernández-Ayala DJM, Stefanatos RKA, Jacobs HT (2010) Mitochondrial ROS production correlates with, but does not directly regulate lifespan in Drosophila. Aging 2:200–223
Sarup P, Loeschcke V (2011) Life extension and the position of the hormetic zone depends on sex and genetic background in Drosophila melanogaster. Biogerontology 12:109–117
SAS Institute, Inc. (2010) The SAS system for windows, release 9.3. SAS Institute, Cary
Schuler F, Casida JE (2001) The insecticide target in the PSST subunit of complex I. Pest Manag Sci 57:932–940
Šešlija D (2005) Analysis of the selection effects on fitness components in the bean weevil (Acanthoscelides obtectus). Doctoral dissertation, University of Belgrade, Belgrade
Šešlija D, Blagojević D, Spasić M, Tucić N (1999) Activity of superoxide dismutase and catalase in the bean weevil (Acanthoscelides obtectus) selected for postponed senescence. Exp Gerontol 34:185–195
Sherer TB, Richardson JR, Testa CM, Seo BB, Panov AV, Yagi T, Matsuno-Yagi A, Miller GW, Greenamyre JT (2007) Mechanism of toxicity of pesticides acting at complex I: relevance to environmental etiologies of Parkinson’s disease. J Neurochem 100:1469–1479
Sohal RS (2002) Oxidative stress hypothesis of aging. Free Radic Biol Med 33:573–574
Sohal RS, Orr WC (2012) The redox stress hypothesis of aging. Free Radic Biol Med 52:539–555
Sőti C, Csermely P (2007) Aging cellular networks: chaperones as major participants. Exp Gerontol 42:113–119
Sriram A (2012) Effect of the reduction of respiratory complex I levels on Drosophila melanogaster lifespan. MS Thesis, Tampere University of Technology, Tampere
Stojković B, Savković U (2011) Gender differences in longevity in early and late reproduced lines of the seed beetle. Arch Biol Sci (Belgrade) 63:129–136
St-Pierre J, Buckingham AJ, Roebuck SJ, Brand MD (2002) Topology of superoxide production from different sites in the mitochondrial electron transport chain. J Biol Chem 277:44784–44790
Tower J (2009) Hsps and aging. Trends Endocrin Met 20:216–222
Tucić N, Gliksman I, Šešlija D, Milanović D, Mikuljanac S, Stojković O (1996) Laboratory evolution of longevity in the Bean weevil (Acanthoscelides obtectus). J Evol Biol 9:485–503
Tucić N, Šešlija D, Stanković V (2004) The short-term and long-term effects of parental age in the bean weevil (Acanthoscelides obtectus). Evol Ecol 18:187–201
Van Pottelberge S, Van Leeuwen T, Nauen R, Tirry L (2009) Resistance mechanisms to mitochondrial electron transport inhibitors in a field-collected strain of Tetranychus urticae Koch (Acari: Tetranychidae). Bull Entomol Res 99:23–31
Vermeulen CJ, Loeschcke V (2007) Longevity and the stress response in Drosophila. Exp Gerontol 42:153–159
Vettraino J, Buck S, Arking R (2001) Direct selection for paraquat resistance in Drosophila results in a different extended longevity phenotype. J Gerontol 56A:B415–B425
Willlett CS, Burton RS (2003) Environmental influences on epistatic interactions: viabilities of cytochrome c genotypes in interpopulation crosses. Evolution 57:2286–2292
Yashin AI, Cypser JW, Johnson TE, Michalski AI, Boyko SI, Vasili N, Novoseltsev VN (2002) Heat shock changes the heterogeneity distribution in populations of Caenorhabditis elegans: does it tell us anything about the biological mechanism of stress response? J Gerontol Biol Sci 57A:B83–B92
Zera AJ, Harshman LG (2010) Laboratory selection studies of life-history physiology in insects. In: Garland T Jr, Rose MR (eds) Experimental evolution: concepts, methods, and applications of selection experiments. University of California Press, Berkeley, pp 217–262
Zid BM, Rogers A, Katewa SD, Vargas MA, Kolipinski M, Lu TA, Benzer S, Pankaj Kapahi P (2009) 4E-BP extends lifespan upon dietary restriction by enhancing mitochondrial activity in Drosophila. Cell 139:149–160
Acknowledgements
We thank the Editor and an anonymous referee for constructive comments and helpful suggestions. This work was supported by Ministry of Education, Science and Technological Development, Grant No. 173007.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jovanović, D.Š., Đorđević, M., Savković, U. et al. The effect of mitochondrial complex I inhibitor on longevity of short-lived and long-lived seed beetles and its mitonuclear hybrids. Biogerontology 15, 487–501 (2014). https://doi.org/10.1007/s10522-014-9520-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10522-014-9520-5