Abstract
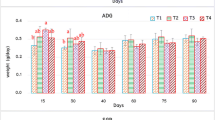
Six types of Chinese tea were incorporated into diets at four different levels (0, 1, 2 and 4 %) in order to determine their effects on the growth performance, fatty acid composition of muscle lipids and resistance to Aeromonas hydrophila infection of Ictalurus punctatus (channel catfish) fingerlings. Each diet was fed to catfish in triplicate tanks for 8 weeks. Results show that a 1 % Dragon Well tea (DWT) and Maofeng tea were significantly better than other diets (including the control) in improving weight gain (WG) and feed conversion rate (FCR) of fish; while the 4 % DWT increased FCR (P < 0.01) but decreased WG (P < 0.05). In addition, 4 % DWT resulted in a significant decrease in muscle lipid content. Pu-erh tea in 2 and 4 % dietary addition decreased saturated fatty acid and increased monounsaturated fatty acid content in muscle. The hepatosomatic index decreased significantly in fish fed the 4 % DWT diet. The mortality (%) of catfish challenged with A. hydrophila decreased with increasing addition of tea to the diet, and the mortality in DWT and Huangya tea (HYT) was lower than control groups (P < 0.05). In conclusion, diets supplemented with a low dose (1 %) of Chinese tea increased growth performance and enhanced the fish fillet quality. Addition of DWT or HYT tea at 4 % in channel catfish diets can improve resistance against A. hydrophila, but growth performance is reduced.

Similar content being viewed by others
Abbreviations
- GT:
-
Green tea
- FT:
-
Fermented tea
- SFA:
-
Saturated fatty acid
- MUFA:
-
Monounsaturated fatty acid
- PUFA:
-
Polyunsaturated fatty acid
- DWT:
-
Dragon Well tea
- PFT:
-
Pan-fired tea
- HYT:
-
HuangYa tea
- MFT:
-
MaoFeng tea
- PRT:
-
Pu-erh tea
- KMT:
-
Keemun tea
- FCR:
-
Feed conversion ratio
- PVC:
-
Polyvinyl chloride
- LDPE:
-
Low-density polyethylene
- WG:
-
Weight gain
- HSI:
-
Hepatosomatic index
- VSI:
-
Viscerosomatic index
- IFR:
-
Intraperitoneal fat ratio
References
Abdel-Tawwab M, Ahmad MH, Seden MEA, Sakr SFM (2010) Use of green tea, Camellia sinensis L., in practical diet for growth and protection of Nile Tilapia, Oreochromis niloticus (L.), against Aeromonas hydrophila infection. J World Aquac Soc 41:203–213
Balentine DA, Wiseman SA, Bouwens LC (1997) The chemistry of tea flavonoids. Crit Rev Food Sci 37:693–704
Bandyopadhyay D, Chatterjee TK, Dasgupta A, Lourduraja J, Dastidar SG (2005) In vitro and in vivo antimicrobial action of tea: the commonest beverage of Asia. Biol Pharm Bull 28(11):2125–2127
Bandyopadhyay P, Ghosh AK, Ghosh C (2012) Recent developments on polyphenol-protein interactions: effects on tea and coffee taste, antioxidant properties and the digestive system. Food Funct 3:592–605
Chacko SM, Thambi PT, Kuttan R, Nishigaki I (2010) Beneficial effects of green tea: a literature review. Chin Med 5:1–9
Cho SH, Kim CI (2009) Effect of dietary inclusion of various sources of additives on growth and body composition of juvenile olive flounder Paralichthys olivaceus. Aquac Res 40:625–629
Cho SH, Lee SM, Park BH, Ji SC, Lee J, Bae J, Oh SY (2007) Effect of dietary inclusion of various sources of green tea on growth, body composition and blood chemistry of the juvenile olive flounder, Paralichthys olivaceus. Fish Physiol Biochem 33:49–57
Dal Bosco A, Castellini C, Bianchi L, Mugnai C (2004) Effect of dietary alpha-linolenic acid and vitamin E on the fatty acid composition, storage stability and sensory traits of rabbit meat. Meat Sci 66:407–413
Dembele S, Wang DF, Yu LN, Sun JP, Dong SY (2010) Effects of added crude green tea polyphenol on the lipid oxidation of common carp (Cyprinus Carpio L.) and catfish (Clarias gariepinus Burchell) during refrigerated storage. J Muscle Foods 21:738–756
Dong GF, Yang YO, Song XM, Yu L, Zhao TT, Huang GL, Zhang JL (2013) Comparative effects of dietary supplementation with maggot meal and soybean meal in gibel carp (Carassius auratus gibelio) and darkbarbel catfish (Pelteobagrus vachelli): growth performance and antioxidant responses. Aquac Nutr 19:543–554
Erba D, Riso P, Bordoni A, Foti P, Biagim PL, Testolin G (2005) Effectiveness of moderate green tea consumption on antioxidative status and plasma lipid profile in humans. J Nutr Biochem 16(3):144–149
FAO Fisheries & Aquaculture (2011) World aquaculture 2010. Food and Agriculture Organization of the United Nations, Rome
FAO Fisheries and Aquaculture (2012) The state of world fisheries and aquaculture: 2012. Food and Agriculture Organization of the United Nations, Rome
Feng WY (2006) Metabolism of green tea catechins: an overview. Curr Drug Metab 7:755–809
Folch J, Lees M, Sloane Stanley GH (1957) A simple method for the isolation and purification of total lipides from animal tissues. J Biol Chem 226:497–509
Frejnagel S, Wroblewska M (2010) Comparative effect of green tea, chokeberry and honeysuckle polyphenols on nutrients and mineral absorption and digestibility in rats. Ann Nutr Metab 56:163–169
Gao J, Koshio S, Ishikawa M, Yokoyama S, Mamauag REP, Han Y (2012) Effects of dietary oxidized fish oil with vitamin E supplementation on growth performance and reduction of lipid peroxidation in tissues and blood of red sea bream Pagrus major. Aquaculture 356:73–79
Harikrishnan R, Balasundaram C, Heo M-S (2011) Impact of plant products on innate and adaptive immune system of cultured finfish and shellfish. Aquaculture 317:1–15
Haslam E (2003) Thoughts on thearubigins. Phytochemistry 64:61–73
He Q, Lv Y, Yao K (2007) Effects of tea polyphenols on the activities of alpha-amylase, pepsin, trypsin and lipase. Food Chem 101:1178–1182
Higdon JV, Frei B (2003) Tea catechins and polyphenols. Health effects, metabolism, and antioxidant functions. Crit Rev Food Sci 43:89–143
Hrelia S, Bordoni A, Angeloni C, Leoncini E, Toschi TG, Lercker G, Biagi PL (2002) Green tea extracts can counteract the modification of fatty acid composition induced by doxorubicin in cultured cardiomyocytes. Prostaglandins Leukot Essent Fat Acids 66:519–524
Huang J, Zhang Y, Zhou Y, Zhang Z, Xie Z, Zhang J, Wan X (2013) Green tea polyphenols alleviate obesity in broiler chickens through the regulation of lipid-metabolism-related genes and transcription factor expression. J Agric Food Chem 61(36):8565–8572
Hwang JH, Lee SW, Rha SJ, Yoon HS, Park ES, Han KH, Kim SJ (2013) Dietary green tea extract improves growth performance, body composition, and stress recovery in the juvenile black rockfish, Sebastes schlegeli. Aquac Int 21:525–538
Ji SC, Jeong GS, Im GS, Lee SW, Yoo JH, Takii K (2007) Dietary medicinal herbs improve growth performance, fatty acid utilization, and stress recovery of Japanese flounder. Fish Sci 73:70–76
Kamboh AA, Zhu WY (2013) Effect of increasing levels of bioflavonoids in broiler feed on plasma anti-oxidative potential, lipid metabolites, and fatty acid composition of meat. Poult Sci 92:454–461
Khan N, Mukhtar H (2007) Tea polyphenols for health promotion. Life Sci 81:519–533
Klaus S, Pultz S, Thone-Reineke C, Wolfram S (2005) Epigallocatechin gallate attenuates diet-induced obesity in mice by decreasing energy absorption and increasing fat oxidation. Int J Obes 29:615–623
Li SY, Tse IMY, Li ETS (2012a) Maternal green tea extract supplementation to rats fed a high-fat diet ameliorates insulin resistance in adult male offspring. J Nutr Biochem 23:1655–1660
Li MH, Oberle DF, Lucas PM (2012b) Effects of dietary fiber concentrations supplied by corn bran on feed intake, growth, and feed efficiency of channel catfish. N Am J Aquac 74:148–153
Maqsood S, Benjakul S, Shahidi F (2013) Emerging role of phenolic compounds as natural food additives in fish and fish products. Crit Rev Food Sci Nutr 53:162–179
McKay DL, Blumberg JB (2002) The role of tea in human health: an update. J Am Coll Nutr 21:1–13
Mendilaharsu M, De Stefani E, Deneo-Pellegrini H, Carzoglio JC, Ronco A (1998) Consumption of tea and coffee and the risk of lung cancer in cigarette-smoking men: a case–control study in Uruguay. Lung Cancer 19(2):101–117
Morrison WR, Smith LM (1964) Preparation of fatty acid methyl esters and dimethylacetals from lipids with boron fluoride–methanol. J Lipid Res 5:600–608
Mukhtar H, Ahmad N (2000) Tea polyphenols: prevention of cancer and optimizing health. Am J Clin Nutr 71(6):1698s–1702s. http://ajcn.nutrition.org/content/71/6/1698s.full
Murase T, Nagasawa A, Suzuki J, Hase T, Tokimitsu I (2002) Beneficial effects of tea catechins on diet-induced obesity: stimulation of lipid catabolism in the liver. Int J Obes 26:1459–1464
Murase T, Haramizu S, Shimotoyodome A, Nagasawa A, Tokimitsu I (2005) Green tea extract improves endurance capacity and increases muscle lipid oxidation in mice. Am J Physiol Regul Integr Comp Physiol 288:R708–R715
Murase T, Haramizu S, Shimotoyodome A, Tokimitsu I, Hase T (2006) Green tea extract improves running endurance in mice by stimulating lipid utilization during exercise. Am J Physiol Regul Integr Comp Physiol 290:R1550–R1556
Naldi L, Gallus S, Tavani A, Imberti GL, La Vecchia C (2004) Risk of melanoma and vitamin A, coffee and alcohol: a case–control study from Italy. Eur J Cancer Prev 13(6):503–508
Nance CL, Shearer WT (2003) Is green tea good for HIV-1 infection. J Allergy Clin Immunol 112(5):851–853
Ota N, Soga S, Shimotoyodome A, Haramizu S, Inaba M, Murase T, Tokimitsu I (2005) Effects of combination of regular exercise and tea catechins intake on energy expenditure in humans. J Health Sci 51:233–236
Sheikhzadeh N, Nofouzi K, Delazar A, Oushani AK (2011) Immunomodulatory effects of decaffeinated green tea (Camellia sinensis) on the immune system of rainbow trout (Oncorhynchus mykiss). Fish Shellfish Immunol 31:1268–1269
Shimotoyodome A, Haramizu S, Inaba M, Murase T, Tokimitsu I (2005) Exercise and green tea extract stimulate fat oxidation and prevent obesity in mice. Med Sci Sports Exerc 37:1884–1892
Shishikura Y, Khokhar S, Murray BS (2006) Effects of tea polyphenols on emulsification of olive oil in a small intestine model system. J Agric Food Chem 54:1906–1913
Sueoka N, Suganuma M, Sueoka E, Okabe S, Matsuyama S, Imai K, Nakachi K, Fujiki H (2001) A new function of green tea: prevention of lifestyle-related diseases. Ann NY Acad Sci 928:274–280
Tan CY, Zhong RZ, Tan ZL, Han XF, Tang SX, Xiao WJ, Sun ZH, Wang M (2011) Dietary inclusion of tea catechins changes fatty acid composition of muscle in goats. Lipids 46:239–247
Thawonsuwan J, Kiron V, Satoh S, Panigrahi A, Verlhac V (2010) Epigallocatechin-3-gallate (EGCG) affects the antioxidant and immune defense of the rainbow trout, Oncorhynchus mykiss. Fish Physiol Biochem 36:687–697
Trattner S, Pickova J, Park KH, Rinchard J, Dabrowski K (2007) Effects of alpha-lipoic and ascorbic acid on the muscle and brain fatty acids and antioxidant profile of the South American pacu Piaractus mesopotamicus. Aquaculture 273:158–164
Unno T, Osada C, Motoo Y, Suzuki Y, Kobayashi M, Nozawa A (2009) Dietary tea catechins increase fecal energy in rats. J Nutr Sci Vitaminol 55:447–451
Wang F, Ma XZ, Wang W, Liu JY (2012) Comparison of proximate composition, amino acid and fatty acid profiles in wild, pond- and cage-cultured long snout catfish (Leiocassis longirostris). Int J Food Sci Technol 47:1772–1776
Weisburger JH, Chung FL (2002) Mechanisms of chronic disease causation by nutritional factors and tobacco products and their prevention by tea polyphenols. Food Chem Toxicol 40:1145–1154
Zakes Z, Kowalska A, Demska-Zakes K, Jeney G, Jeney Z (2008) Effect of two medicinal herbs (Astragalus radix and Lonicera japonica) on the growth performance and body composition of juvenile pikeperch Sander lucioperca (L.). Aquac Res 39:1149–1160
Zhang L, Zhang ZZ, Zhou YB, Ling TJ, Wan XC (2013) Chinese dark teas: postfermentation, chemistry and biological activities. Food Res Int 53:600–607
Acknowledgments
This research was supported by the International Science and Technology Cooperation Programme of the Ministry of Science and Technology (Project Number: 2011DFG33280).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, Ybp., Zhou, Yb., Sang, By. et al. Effect of dietary Chinese tea on growth performance, disease resistance and muscle fatty acid profile of channel catfish (Ictalurus punctatus). Aquacult Int 23, 683–698 (2015). https://doi.org/10.1007/s10499-014-9846-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-014-9846-7