Abstract
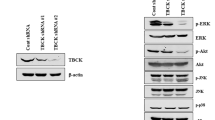
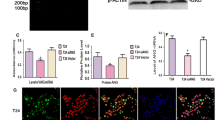
The urokinase-type plasminogen activator receptor (uPAR) serves not only as an anchor for urokinase-type plasminogen activator but also participates in intracellular signal transduction events. In this study, we investigated whether uPAR could modulate TRAIL-induced apoptosis in human colon cancer cells HCT116. Using an antisense strategy, we established a stable HCT116 cell line with down-regulated uPAR. The sensitivity to TRAIL-induced apoptosis was evaluated by FACS analysis. Our results show that the inhibition of uPAR could sensitize HCT116 to TRAIL-induced apoptosis. uPAR inhibition changed the expression of mitochondrial apoptotic pathway proteins, including Bcl-2, Bax, Bid and p53, in a pro-apoptotic manner. We also found that the inhibition of uPAR down-regulated the phosphorylation of FAK, ERK and JNK. The inhibition of p53 by RNA interference rescued cells from enhanced apoptosis, thus indicating that p53 is critical for enhancing TRAIL-induced apoptosis. Furthermore, JNK, but not ERK, inhibition involved in the up-regulation of p53. JNK negatively regulated p53 protein level. Overall, our results show that uPAR inhibition can sensitize colon cancer cells HCT116 to TRAIL-induced apoptosis via active p53 and mitochondrial apoptotic pathways that JNK inhibition is involved.






Similar content being viewed by others
Abbreviations
- uPAR:
-
Urokinase-type plasminogen activator receptor
- uPA:
-
Urokinase-type plasminogen activator
- AS:
-
Antisense
- TRAIL:
-
Tumor necrosis factor (TNF)-related apoptosis-inducing ligand
- DR4:
-
Death receptor 4
- DR5:
-
Death receptor 5
- ECM:
-
Extracellular matrix
- FAK:
-
Focal adhesion kinase
- ERK:
-
Extracellular signal-regulated kinases
- JNK:
-
Jun N-terminal kinase
- Bcl-2:
-
B cell lymphoma 2
References
Almasi CE, Christensen IJ, Høyer-Hansen G, Danø K, Pappot H, Dienemann H, Muley T (2001) Urokinase receptor forms in serum from non-small cell lung cancer patients: Relation to prognosis. Lung Cancer 74(3):510–515
Sorio C, Mafficini A, Furlan F, Barbi S, Bonora A, Brocco G, Blasi F, Talamini G, Bassi C, Scarpa A (2011) Elevated urinary levels of urokinase-type plasminogen activator receptor (uPAR) in pancreatic ductal adenocarcinoma identify a clinically high-risk group. BMC Cancer 11:448
Pedersen H, Brünner N, Francis D, Osterlind K, Rønne E, Hansen HH, Danø K, Grøndahl-Hansen J (1994) Prognostic impact of urokinase, urokinase receptor, and type 1 plasminogen activator inhibitor in squamous and large cell lung cancer tissue. Cancer Res 54(17):4671–4675
de Vries TJ, van Muijen GN, Ruiter DJ (1996) The plasminogen activation system in tumour invasion and metastasis. Pathol Res Pract 192(7):718–733
Smith HW, Marshall CJ (2010) Regulation of cell signalling by uPAR. Nat Rev Mol Cell Biol 11(1):23–36
Resnati M, Pallavicini I, Wang JM, Oppenheim J, Serhan CN, Romano M, Blasi F (2002) The fibrinolytic receptor for urokinase activates the G protein-coupled chemotactic receptor FPRL1LXA4R. Proc Natl Acad Sci USA 99(3):1359–1364
Tang CH, Wei Y (2008) The urokinase receptor and integrins in cancer progression. Cell Mol Life Sci 65(7):1916–1932
Franco P, Vocca I, Carriero MV, Alfano D, Cito L, Longanesi-Cattani I, Grieco P, Ossowski L, Stoppelli MP (2006) Activation of urokinase receptor by a novel interaction between the connecting peptide region of urokinase and alpha v beta 5 integrin. J Cell Sci 119(16):3424–3434
Kugler MC, Wei Y, Chapman HA (2003) Urokinase receptor and integrin interactions. Curr Pharm Des 9(19):1565–1574
Cavallo-Medved D, Dosescu J, Linebaugh BE, Sameni M, Rudy D, Sloane BF (2003) Mutant K-ras regulates cathepsin B localization on the surface of human colorectal carcinoma cells. Neoplasia 5(6):507–519
Kotipatruni RR, Nalla AK, Asuthkar S, Gondi CS, Dinh DH, Rao JS (2012) Apoptosis induced by knockdown of uPAR and MMP-9 is mediated by inactivation of EGFR/STAT3 signaling in medulloblastoma. PLoS ONE 7(9):e44798
Gondi CS, Kandhukuri N, Dinh DH, Gujrati M, Rao JS (2007) Down-regulation of uPAR and uPA activates caspase-mediated apoptosis and inhibits the PI3K/AKT pathway. Int J Oncol 31(1):19–27
Gopinath S, Malla RR, Gondi CS, Alapati K, Fassett D, Klopfenstein JD, Dinh DH, Gujrati M, Rao JS (2010) Co-depletion of cathepsin B and uPAR induces G0/G1 arrest in glioma via FOXO3a mediated p27Kip1 upregulation. PLoS ONE 5(7):e11668
Smith CA, Ferrah T, Goodwin RG (1995) The TNF receptor superfamily of cellular and viral proteins: activation costimulation and death. Cell 76:959–962
Srivastava RK (2001) TRAIL/Apo-2L: mechanisms and clinical applications in cancer. Neoplasia 3(6):535–546
Suliman A, Lam A, Datta R, Srivastava RK (2001) Intracellular mechanisms of TRAIL: apoptosis through mitochondrial-dependent and independent pathways. Oncogene 20(17):2122–2133
Gross A, McDonnell JM, Korsmeyer SJ (1999) BCL-2 family members and the mitochondria in apoptosis. Genes Dev 13(15):1899–1911
Schug ZT, Gonzalvez F, Houtkooper RH, Vaz FM, Gottlieb E (2011) BID is cleaved by caspase-8 within a native complex on the mitochondrial membrane. Cell Death Differ 18(3):538–548
Kroemer G, Galluzzi L, Brenner C (2007) Mitochondrial membrane permeabilization in cell death. Physiol Rev 87(1):99–163
Park EJ, Choi KS, Yoo YH, Kwon TK (2013) Nutlin-3, a small-molecule MDM2 inhibitor, sensitizes caki cells to TRAIL-induced apoptosis through p53-mediated PUMA upregulation and ROS-mediated DR5 upregulation. Anticancer Drugs 24(3):260–269
Kim JY, Kim EH, Kim SU, Kwon TK, Choi KS (2010) Capsaicin sensitizes malignant glioma cells to TRAIL-mediated apoptosis via DR5 upregulation and survivin downregulation. Carcinogenesis 31(3):367–375
Moon DO, Asami Y, Long H, Jang JH, Bae EY, Kim BY, Choi YH, Kang CH, Ahn JS, Kim GY (2013) Verrucarin A sensitizes TRAIL-induced apoptosis via the upregulation of DR5 in an eIF2α/CHOP-dependent manner. Toxicol In Vitro 27(1):257–263
Kim HB, Kim MJ, Lee SH, Lee JW, Bae JH, Kim DW, Dao TT, Oh WK, Kang CD, Kim SH (2012) Amurensin G, a novel SIRT1 inhibitor, sensitizes TRAIL-resistant human leukemic K562 cells to TRAIL-induced apoptosis. Biochem Pharmacol 84(3):402–410
Shin SW, Park JW (2013) Ursolic acid sensitizes prostate cancer cells to TRAIL-mediated apoptosis. Biochim Biophys Acta 1833(3):723–730
Kim K, Nakagawa H, Fei P, Rustgi AK, El-Deiry WS (2004) Targeting Bcl-xL in esophageal squamous cancer to sensitize to chemotherapy plus TRAIL-induced apoptosis while normal epithelial cells are protected by blockade of caspase 9. Cell Death Differ 11(5):583–587
Ray S, Bucur O, Almasan A (2005) Sensitization of prostate carcinoma cells to Apo2L/TRAIL by a Bcl-2 family protein inhibitor. Apoptosis 10(6):1411–1418
Zhao J, Lu Y, Shen HM (2012) Targeting p53 as a therapeutic strategy in sensitizing TRAIL-induced apoptosis in cancer cells. Cancer Lett 314(1):8–23
Rieger J, Naumann U, Glaser T, Ashkenazi A, Weller M (1998) APO2 ligand: a novel lethal weapon against malignant glioma. FEBS Lett 427(1):124–128
Ashkenazi A, Dixit VM (1998) Death receptors: signaling and modulation. Science (Washington, DC) 281(5381):1305–1308
Chipuk JE, Kuwana T, Bouchier-Hayes L, Droin NM, Newmeyer DD, Schuler M, Green DR (2004) Direct activation of Bax by p53 mediates mitochondrial membrane permeabilization and apoptosis. Science 303(5660):1010–1014
Cheng J, Cui R, Chen CH, Du J (2007) Oxidized low-density lipoprotein stimulates p53-dependent activation of proapoptotic Bax leading to apoptosis of differentiated endothelial progenitor cells. Endocrinology 148(5):2085–2094
Yamaguchi H, Chen J, Bhalla K, Wang HG (2004) Regulation of Bax activation and apoptotic response to microtubule-damaging agents by p53 transcription dependent and independent pathways. J Biol Chem 279(38):39431–39437
Liu X, Yue P, Khuri FR, Sun SY (2004) p53 Upregulates death receptor 4 expression through an intronic p53 binding site. Cancer Res 64(20):5078–5083
Zhuang H, Jiang W, Zhang X, Qiu F, Gan Z, Cheng W, Zhang J, Guan S, Tang B, Huang Q, Wu X, Huang X, Jiang W, Hu Q, Lu M, Hua ZC (2013) Suppression of HSP70 expression sensitizes NSCLC cell lines to TRAIL-induced apoptosis by upregulating DR4 and DR5 and downregulating c-FLIP-L expressions. J Mol Med 91(2):219–235
Srsen V, Gnadt N, Dammermann A, Merdes A (2006) Inhibition of centrosome protein assembly leads to p53-dependent exit from the cell cycle. J Cell Biol 174(5):625–630
Tsujimoto Y (2002) Bcl-2 family of proteins: life-or-death switch in mitochondria. Biosci Rep 22(1):47–58
D’Alessio S, Gerasi L, Blasi F (2008) uPAR-deficient mouse keratinocytes fail to produce EGFR-dependent laminin-5, affecting migration in vivo and in vitro. J Cell Sci 121(Pt 23):3922–3932
Margheri F, D’Alessio S, Serratí S, Pucci M, Annunziato F, Cosmi L, Liotta F, Angeli R, Angelucci A, Gravina GL, Rucci N, Bologna M, Teti A, Monia B, Fibbi G, Del Rosso M (2005) Effects of blocking urokinase receptor signaling by antisense oligonucleotides in a mouse model of experimental prostate cancer bone metastase. Gene Ther 12(8):702–714
Lin A, Dibling B (2002) The true face of JNK activation in apoptosis. Aging Cell 1(2):112–116
Nalla AK, Asuthkar S, Bhoopathi P, Gujrati M, Dinh DH, Rao JS (2010) Suppression of uPAR retards radiation-induced invasion and migration mediated by integrin β1/FAK signaling in medulloblastoma. PLoS ONE 5(9):e13006
Kin Y, Chintala SK, Go Y, Sawaya R, Mohanam S, Kyritsis AP, Rao JS (2000) A novel role for the urokinase-type plasminogen activator receptor in apoptosis of malignant gliomas. Int J Oncol 17(1):61–65
Hildenbrand R, Gandhari M, Stroebel P, Marx A, Allgayer H, Arens N (2008) The urokinase system role of cell proliferation and apoptosis. Histol Histopathol 23(2):227–236
Besch R, Berking C, Kammerbauer C, Degitz K (2007) Inhibition of urokinase-type plasminogen activator receptor induces apoptosis in melanoma cells by activation of p53. Cell Death Differ 14(4):818–829
Prager GW, Breuss JM, Steurer S, Olcaydu D, Mihaly J, Brunner PM, Stockinger H, Binder BR (2004) Vascular endothelial growth factor receptor-2-induced initial endothelial cell. Migration depends on the presence of the urokinase receptor. Circ Res 94(12):1562–1570
Cortese K, Sahores M, Madsen CD, Tacchetti C, Blasi F (2008) Clathrin and LRP-1-independent constitutive endocytosis and recycling of uPAR. PLoS ONE 3(11):e3730
Xiaowen H, Yi S (2012) Triptolide sensitizes TRAIL-induced apoptosis in prostate cancer cells via p53-mediated DR5 up-regulation. Mol Biol Rep 39(9):8763–8770
Lan YH, Chiang JH, Huang WW, Lu CC, Chung JG, Wu TS, Jhan JH, Lin KL, Pai SJ, Chiu YJ, Tsuzuki M, Yang JS (2012) Activations of both extrinsic and intrinsic pathways in HCT 116 human colorectal cancer cells contribute to apoptosis through p53-mediated ATM/Fas signaling by emilia sonchifolia extract, a folklore medicinal plant. Evid Based Complement Altern Med 2012:178
Takimoto R, El-Deiry WS (2000) Wild-type p53 transactivates the KILLER/DR5 gene through an intronic sequence-specific DNA-binding site. Oncogene 19(14):1735–1743
Guan B, Yue P, Clayman GL, Sun SY (2001) Evidence that the death receptor DR4 is a DNA damage-inducible, p53-regulated gene. J Cell Physiol 188(1):98–105
Shetty S, Velusamy T, Idell S, Shetty P, Mazar AP, Bhandary YP, Shetty RS (2007) Regulation of urokinase receptor expression by p53: novel role in stabilization of uPAR mRNA. Mol Cell Biol 27(16):5607–5618
Pulukuri SM, Gondi CS, Lakka SS, Jutla A, Estes N, Gujrati M, Rao JS (2005) RNA interference-directed knockdown of urokinase plasminogen activator receptor inhibits MMPs expression via dephosphorylation of Erk1/2 and Stat3 in AM. J Biol Chem 280(43):36529–36540
Malla RR, Gopinath S, Alapati K, Gorantla B, Gondi CS, Rao JS (2012) uPAR and cathepsin B inhibition enhanced radiation-induced apoptosis in glioma initiating cells. Neuro Oncol 14(6):745–760
Das M, Jiang F, Sluss HK, Zhang C, Shokat KM, Flavell RA, Davis RJ (2007) Suppression of p53-dependent senescence by the JNK signal transduction pathway. Proc Natl Acad Sci USA 104(40):15759–15764
Fuchs SY, Adler V, Buschmann T, Yin Z, Wu X, Jones SN, Ronai Z (1998) JNK targets p53 ubiquitination and degradation in nonstressed cells. Genes Dev 12(17):2658–2663
Rahman M, Zhang Z, Mody AA, Su DM, Das HK (2012) Intraperitoneal injection of JNK-specific inhibitor SP600125 inhibits the expression of presenilin-1 and Notch signaling in mouse brain without induction of apoptosis. Brain Res 1448:117–128
Saha MN, Jiang H, Yang Y, Zhu X, Wang X, Schimmer AD, Qiu L, Chang H (2012) Targeting p53 via JNK pathway: a novel role of RITA for apoptotic signaling in multiple myeloma. PLoS ONE 7(1):e30215
Acknowledgments
The authors are grateful to grants from the Ministry of Science and Technology (2012CB967004, 2014CB744501, 2012AA020304 and 2012ZX09401012), the Chinese National Natural Sciences Foundation (81121062, 31200572, 31070706 and 31071196), the Jiangsu Provincial Nature Science Foundation (BE2013630 and BZ2012050), the Bureau of Science and Technology of Changzhou, Jiangsu, China (CZ20130011, CE20135013, CZ20120004, CM20122003 and WF201207).
Conflict of interest
The authors declare that there are no conflicts of interest.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Liu, X., Qiu, F., Liu, Z. et al. Urokinase-type plasminogen activator receptor regulates apoptotic sensitivity of colon cancer HCT116 cell line to TRAIL via JNK-p53 pathway. Apoptosis 19, 1532–1544 (2014). https://doi.org/10.1007/s10495-014-1025-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-014-1025-9