Abstract
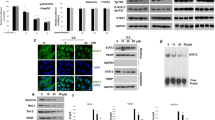
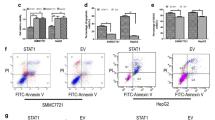
Growing evidence has demonstrated that signal transducer and activator of transcription 3 (STAT3), which is constitutively activated in patients with hepatocellular carcinoma (HCC), plays an important role in HCC development, progression, and prognosis. Interleukin-6 (IL-6) is a multifunctional inflammatory cytokine, which may induce STAT3 activation in a variety of human cancers. In this study, we demonstrated that blocking STAT3 phosphorylation with STAT3 small molecule inhibitors SD-1029 and Stattic caused apoptosis in HCC cells. Then we characterized the inhibitory effects of a novel small molecule XZH-5 on STAT3 phosphorylation in HCC cell lines. XZH-5 reduced constitutive STAT3 phosphorylation at Tyr705 and the expression of STAT3 downstream genes. The inhibition of STAT3 in HCC cells resulted in the induction of apoptosis and reduction of colony forming ability. In addition, XZH-5 also inhibited IL-6-induced STAT3 phosphorylation, nuclear translocation and STAT3 DNA binding activity. In contrast, it had no effect on IFN-γ-induced STAT1 phosphorylation, indicating the more selective effects on STAT3. These results suggested that XZH-5 may serve as a lead compound for further development of STAT3 specific small molecule inhibitors for HCC therapy.








Similar content being viewed by others
References
Greenlee RT, Hill-Harmon MB, Murray T, Thun M (2001) Cancer statistics. CA Cancer J Clin 51:15–36
Berasain C, Castillo J, Perugorria MJ, Latasa MU, Prieto J, Avila MA (2009) Inflammation and liver cancer: new molecular links. Ann N Y Acad Sci 1155:206–221
Sherman M (2005) Hepatocellular carcinoma: epidemiology, risk factors, and screening. Semin Liver Dis 25:143–154
Wilson JF (2005) Liver cancer on the rise. Ann Intern Med 142:1029–1032
Bosch FX, Ribes J, Cleries R, Diaz M (2005) Epidemiology of hepatocellular carcinoma. Clin Liver Dis 9:191–211
Bromberg JF, Wrzeszczynska MH, Devgan G et al (1999) Stat3 as an oncogene. Cell 98:295–303
Buettner R, Mora LB, Jove R (2002) Activated STAT signaling in human tumors provides novel molecular targets for therapeutic intervention. Clin Cancer Res 8:945–954
Yu H, Kortylewski M, Pardoll D (2007) Crosstalk between cancer and immune cells: role of STAT3 in the tumour microenvironment. Nat Rev Immunol 7:41–51
Yu H, Pardoll D, Jove R (2009) STATs in cancer inflammation and immunity: a leading role for STAT3. Nat Rev Cancer 9:798–809
Niu G, Wright KL, Huang M et al (2002) Constitutive Stat3 activity up-regulates VEGF expression and tumor angiogenesis. Oncogene 21:2000–2008
Real PJ, Sierra A, De Juan A, Segovia JC, Lopez-Vega JM, Fernandez-Luna JL (2002) Resistance to chemotherapy via Stat3-dependent overexpression of Bcl-2 in metastatic breast cancer cells. Oncogene 21:7611–7618
Wei LH, Kuo ML, Chen CA et al (2003) Interleukin-6 promotes cervical tumor growth by VEGF-dependent angiogenesis via a STAT3 pathway. Oncogene 22:1517–1527
Wang T, Niu G, Kortylewski M et al (2004) Regulation of the innate and adaptive immune responses by Stat-3 signaling in tumor cells. Nat Med 10:48–54
Turkson J, Zhang S, Mora LB, Burns A, Sebti S, Jove R (2005) A novel platinum compound inhibits constitutive Stat3 signaling and induces cell cycle arrest and apoptosis of malignant cells. J Biol Chem 280:32979–32988
Nam S, Buettner R, Turkson J et al (2005) Indirubin derivatives inhibit Stat3 signaling and induce apoptosis in human cancer cells. Proc Natl Acad Sci USA 102:5998–6003
Rahaman SO, Harbor PC, Chernova O, Barnett GH, Vogelbaum MA, Haque SJ (2002) Inhibition of constitutively active Stat3 suppresses proliferation and induces apoptosis in glioblastoma multiforme cells. Oncogene 21:8404–8413
Li X, Wang H, Lu X, Di B (2010) STAT3 blockade with shRNA enhances radiosensitivity in Hep-2 human laryngeal squamous carcinoma cells. Oncol Rep 23:345–353
Liu Y, Li PK, Li C, Lin J (2010) Inhibition of STAT3 signaling blocks the anti-apoptotic activity of IL-6 in human liver cancer cells. J Biol Chem 285:27429–27439
Lin L, Amin R, Gallicano GI et al (2009) The STAT3 inhibitor NSC 74859 is effective in hepatocellular cancers with disrupted TGF-beta signaling. Oncogene 28:961–972
Zhang B, Zhong DW, Wang QW, et al. (2010) Study on correlation of JAK/STAT signal pathway with progression and prognosis in hepatocellular carcinoma. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi 26:368–370, 373
Hosui A, Kimura A, Yamaji D, Zhu BM, Na R, Hennighausen L (2009) Loss of STAT5 causes liver fibrosis and cancer development through increased TGF-{beta} and STAT3 activation. J Exp Med 206:819–831
Saxena NK, Sharma D, Ding X et al (2007) Concomitant activation of the JAK/STAT, PI3K/AKT, and ERK signaling is involved in leptin-mediated promotion of invasion and migration of hepatocellular carcinoma cells. Cancer Res 67:2497–2507
Li WC, Ye SL, Sun RX et al (2006) Inhibition of growth and metastasis of human hepatocellular carcinoma by antisense oligonucleotide targeting signal transducer and activator of transcription 3. Clin Cancer Res 12:7140–7148
Li J, Piao YF, Jiang Z, Chen L, Sun HB (2009) Silencing of signal transducer and activator of transcription 3 expression by RNA interference suppresses growth of human hepatocellular carcinoma in tumor-bearing nude mice. World J Gastroenterol 15:2602–2608
Choudhari SR, Khan MA, Harris G et al (2007) Deactivation of Akt and STAT3 signaling promotes apoptosis, inhibits proliferation, and enhances the sensitivity of hepatocellular carcinoma cells to an anticancer agent, Atiprimod. Mol Cancer Ther 6:112–121
Lin L, Deangelis S, Foust E et al (2010) A novel small molecule inhibits STAT3 phosphorylation and DNA binding activity and exhibits potent growth suppressive activity in human cancer cells. Mol Cancer 9:217
Bollrath J, Phesse TJ, von Burstin VA et al (2009) gp130-mediated STAT3 activation in enterocytes regulates cell survival and cell-cycle progression during colitis-associated tumorigenesis. Cancer Cell 15:91–102
Berishaj M, Gao SP, Ahmed S et al (2007) STAT3 is tyrosine-phosphorylated through the interleukin-6/glycoprotein 130/Janus kinase pathway in breast cancer. Breast Cancer Res 9:R32
Soresi M, Giannitrapani L, D’Antona F et al (2006) Interleukin-6 and its soluble receptor in patients with liver cirrhosis and hepatocellular carcinoma. World J Gastroenterol 12:2563–2568
Park EJ, Lee JH, Yu GY et al (2010) Dietary and genetic obesity promote liver inflammation and tumorigenesis by enhancing IL-6 and TNF expression. Cell 140:197–208
Duan Z, Bradner JE, Greenberg E et al (2006) SD-1029 inhibits signal transducer and activator of transcription 3 nuclear translocation. Clin Cancer Res 12:6844–6852
McMurray JS (2006) A new small molecule STAT3 inhibitor. Chem Biol 13:1123–1124
Schust J, Sperl B, Hollis A, Mayer TU, Berg T (2006) Stattic: a small molecule inhibitor of STAT3 activation and dimerization. Chem Biol 13:1235–1242
Puthier D, Bataille R, Amiot M (1999) IL-6 up-regulates mcl-1 in human myeloma cells through JAK/STAT rather than ras/MAP kinase pathway. Eur J Immunol 29:3945–3950
Fukada T, Hibi M, Yamanaka Y et al (1996) Two signals are necessary for cell proliferation induced by a cytokine receptor gp130: involvement of STAT3 in anti-apoptosis. Immunity 5:449–460
Dauer DJ, Ferraro B, Song L et al (2005) STAT3 regulates genes common to both wound healing and cancer. Oncogene 24:3397–3408
Garcia R, Bowman TL, Niu G et al (2001) Constitutive activation of STAT3 by the Src and JAK tyrosine kinases participates in growth regulation of human breast carcinoma cells. Oncogene 20:2499–2513
Burke WM, Jin X, Lin HJ et al (2001) Inhibition of constitutively active STAT3 suppresses growth of human ovarian and breast cancer cells. Oncogene 20:7925–7934
Chen CL, Hsieh FC, Lieblein JC et al (2007) STAT3 activation in human endometrial and cervical cancers. Br J Cancer 96:591–599
Yeh HH, Lai WW, Chen HH, Liu HS, Su WC (2006) Autocrine IL-6-induced STAT3 activation contributes to the pathogenesis of lung adenocarcinoma and malignant pleural effusion. Oncogene 25:4300–4309
Shin DS, Kim HN, Shin KD et al (2009) Cryptotanshinone inhibits constitutive signal transducer and activator of transcription 3 function through blocking the dimerization in DU145 prostate cancer cells. Cancer Res 69:193–202
Frank DA (2007) STAT3 as a central mediator of neoplastic cellular transformation. Cancer Lett 251:199–210
Aggarwal BB, Kunnumakkara AB, Harikumar KB et al (2009) Signal transducer and activator of transcription-3, inflammation, and cancer: how intimate is the relationship? Ann N Y Acad Sci 1171:59–76
Masuda M, Suzui M, Yasumatu R et al (2002) Constitutive activation of signal transducers and activators of transcription 3 correlates with cyclin D1 overexpression and may provide a novel prognostic marker in head and neck squamous cell carcinoma. Cancer Res 62:3351–3355
Kiuchi N, Nakajima K, Ichiba M et al (1999) STAT3 is required for the gp130-mediated full activation of the c-myc gene. J Exp Med 189:63–73
Shirogane T, Fukada T, Muller JM, Shima DT, Hibi M, Hirano T (1999) Synergistic roles for Pim-1 and c-Myc in STAT3-mediated cell cycle progression and antiapoptosis. Immunity 11:709–719
Catlett-Falcone R, Landowski TH, Oshiro MM et al (1999) Constitutive activation of STAT3 signaling confers resistance to apoptosis in human U266 myeloma cells. Immunity 10:105–115
Mahboubi K, Li F, Plescia J et al (2001) Interleukin-11 up-regulates survivin expression in endothelial cells through a signal transducer and activator of transcription-3 pathway. Lab Invest 81:327–334
Grivennikov S, Karin E, Terzic J et al (2009) IL-6 and STAT3 are required for survival of intestinal epithelial cells and development of colitis-associated cancer. Cancer Cell 15:103–113
Acknowledgment
This study was partly supported by a grant from NIHR21 to Jiayuh Lin.
Author information
Authors and Affiliations
Corresponding author
Additional information
Yan Liu, Aiguo Liu contributed equally.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Liu, Y., Liu, A., Xu, Z. et al. XZH-5 inhibits STAT3 phosphorylation and causes apoptosis in human hepatocellular carcinoma cells. Apoptosis 16, 502–510 (2011). https://doi.org/10.1007/s10495-011-0578-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-011-0578-0