Abstract
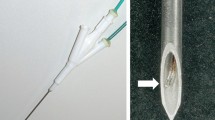
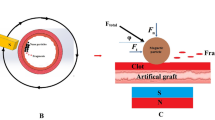
The purpose of the work is to investigate whether the electromagnetic properties of multi-walled carbon nanotubes (MWCNT) in the presence of radiofrequency (RF) energy is (1) safe, and (2) improves the precision of the therapeutic efficiency of the RF-ablation (RFA) procedure. An in vitro phantom was created for evaluating temperature near RF treated nanotubes. For the in vivo study, three baboons and six pigs were submitted for RFA procedure in superior/inferior kidney poles embolized with a non-adherent, lipophilic embolic agent (marsembol) with or without MWCNT. Tissue damage in the surrounding kill zone was assayed through caspase-3 activation. The in vitro results showed marked heat increase only in the region of the nanotubes. In vivo, necrosis/ischemic damage resulted from RFA therapy alone, RFA plus marsembol only. In marsembol + MWCNT condition, dramatic disruption of cell membranes and sub-cellular organelles was found whereas the nuclear membranes and basal cell membranes remained largely intact. The marsembol vaporized under RFA and tissue fluid filled the space. This caused the MWCNT to cluster within the new aqueous environment. RFA plus marsembol + MWCNT created a well-defined demarcation between healthy and apoptotic cells as evidenced by a marked reduction of caspase-3 expression. By contrast, there was a much less defined ablation zone in the absence of MWCNT. In conclusion, the combination of RFA plus marsembol + MWCNT embolization delineated the kill zone in vitro and in vivo. We demonstrate that MWCNTs remain in the ablation region thus minimizing their migration to the systemic circulation.







Similar content being viewed by others
References
Ahmed, M., C. L. Brace, F. T. Lee, and S. N. Goldberg. Principles of and advances in percutaneous ablation. Radiology 258:351–369, 2011.
Ahmed, M., Z. J. Liu, K. S. Afzal, D. Weeks, S. M. Lobo, J. B. Kruskal, R. E. Lenkinski, and S. N. Goldberg. Radiofrequency ablation: effect of surrounding tissue composition on coagulation necrosis in a canine tumor model. Radiology 230:761–767, 2004.
Ahmed, M., Z. J. Liu, A. N. Lukyanov, S. Signoretti, C. Horkan, W. L. Monsky, V. P. Torchilin, and S. N. Goldberg. Combination radiofrequency ablation with intratumoral liposomal doxorubicin: effect on drug accumulation and coagulation in multiple tissues and tumor types in animals. Radiology 235:469–477, 2005.
Altunrende, F., R. Autorino, S. Hillyer, B. Yang, H. Laydner, M. A. White, R. Khanna, W. Isac, G. Spana, R. J. Stein, G. P. Haber, C. M. O’Malley, E. M. Remer, and J. H. Kaouk. Image guided percutaneous probe ablation for renal tumors in 65 solitary kidneys: functional and oncological outcomes. J. Urol. 186:35–41, 2011.
Burke, A., X. F. Ding, R. Singh, R. A. Kraft, N. Levi-Polyachenko, M. N. Rylander, C. Szot, C. Buchanan, J. Whitney, J. Fisher, H. C. Hatcher, R. D’Agostino, N. D. Kock, P. M. Ajayan, D. L. Carroll, S. Akman, F. M. Torti, and S. V. Torti. Long-term survival following a single treatment of kidney tumors with multiwalled carbon nanotubes and near-infrared radiation. Proc. Natl. Acad. Sci. U.S.A. 106:12897–12902, 2009.
Cheng, X., J. Zhong, J. Meng, M. Yang, F. Jia, Z. Xu, H. Kong, and H. Xu. Characterization of multiwalled carbon nanotubes dispersing in water and association with biological effects. J. Nanomater. 2011:1, 2011.
Cooperberg, M. R., C. J. Kane, K. Mallin, and P. R. Carroll. National trends in treatment of stage I renal cell carcinoma. J. Urol. 181:319, 2009.
Cornelis, F., X. Buy, M. Andre, R. Oyen, J. Bouffard-Vercelli, A. Blandino, J. Auriol, J. M. Correas, A. Pluvinage, S. Freeman, S. B. Solomon, and N. Grenier. De novo renal tumors arising in kidney transplants: midterm outcome after percutaneous thermal ablation. Radiology 260:900–907, 2011.
Ding, X. F., R. Singh, A. Burke, H. Hatcher, J. Olson, R. A. Kraft, M. Schmid, D. Carroll, J. D. Bourland, S. Akman, F. M. Torti, and S. V. Torti. Development of iron-containing multiwalled carbon nanotubes for MR-guided laser-induced thermotherapy. Nanomedicine 6:1341–1352, 2011.
Fraczek-Szczypta, A., E. Menaszek, and S. Blazewicz. Some observations on carbon nanotubes susceptibility to cell phagocytosis. J. Nanomater. 2011:473516, 2011.
Gasser, M., B. Rothen-Rutishauser, H. F. Krug, P. Gehr, M. Nelle, B. Yan, and P. Wick. The adsorption of biomolecules to multi-walled carbon nanotubes is influenced by both pulmonary surfactant lipids and surface chemistry. J. Nanobiotechnol. 8:31, 2010.
Gebauer, B., M. Werk, E. Lopez-Haenninen, R. Felix, and P. Althaus. Radiofrequency ablation in combination with embolization in metachronous recurrent renal cancer in solitary kidney after contralateral tumor nephrectomy. Cardiovasc. Intervent. Radiol. 30:644–649, 2007.
Gill, I. S., T. H. S. Hsu, R. L. Fox, A. Matamoros, C. D. Miller, R. F. LeVeen, M. T. Grune, G. T. Sung, and M. E. Fidler. Laparoscopic and percutaneous radiofrequency ablation of the kidney: acute and chronic porcine study. Urology 56:197–200, 2000.
Kang, D. C., D. A. Palmer, M. Zarei, P. Shah, C. Folsom, R. J. Beyth, T. L. Stoffs, M. M. Neuberger, and P. Dahm. A systematic review of the quality of evidence of ablative therapy for small renal masses. J. Urol. 187:44–47, 2012.
Krug, H. F., and P. Wick. Nanotoxicology: an interdisciplinary challenge. Angew. Chem. Int. Ed. Engl. 50:1260–1278, 2011.
Lavoie, P., P. Metellus, L. Velly, V. Vidal, P. H. Rolland, M. Mekaouche, G. Dubreuil, and O. Levrier. Functional cerebral venous outflow in swine and baboon: feasibility of an intracranial venous hypertension model. J. Invest. Surg. 21:323–329, 2008.
Lee, S., and J. W. Peng. Effect of plasma treatment on electrical conductivity and Raman spectra of carbon nanotubes. J. Phys. Chem. Solids 72:1101–1103, 2011.
Levrier, O., C. Mekkaoui, P. H. Rolland, K. Murphy, P. Cabrol, G. Moulin, J. M. Bartoli, and C. Raybaud. Efficacy and low vascular toxicity of embolization with radical versus anionic polymerization of n-butyl-2-cyanoacrylate (NBCA)—an experimental study in the swine. J. Neuroradiol. 30:95–102, 2003.
Lewandowski, R. J., J. F. Geschwind, E. Liapi, and R. Salem. Transcatheter intraarterial therapies: rationale and overview. Radiology 259:641–657, 2011.
Madani, S. Y., A. Mandel, and A. M. Seifalian. A concise review of carbon nanotube’s toxicology. Nano Rev. 4:1–14, 2013.
Margulis, V., E. D. Matsumoto, G. Lindberg, L. Tunc, G. Taylor, A. I. Sagalowsky, and J. A. Cadeddu. Acute histologic effects on temperature-based radiofrequency ablation on renal tumor pathologic interpretation. Urology 64:660–663, 2004.
Matlaga, B. R., R. J. Zagoria, R. D. Woodruff, F. M. Torti, and M. C. Hall. Phase II trial of radio frequency ablation of renal cancer: evaluation of the kill zone. J. Urol. 168:2401–2405, 2002.
Matsumoto, E. D., L. Watumull, D. B. Johnson, K. Ogan, G. D. Taylor, S. Josephs, and J. A. Cadeddu. The radiographic evolution of radio frequency ablated renal tumors. J. Urol. 172:45–48, 2004.
Michaels, M. J., H. K. Rhee, A. P. Mourtzinos, I. C. Summerhayes, M. L. Silverman, and J. A. Libertino. Incomplete renal tumor destruction using radio frequency interstitial ablation. J. Urol. 168:2406–2409, 2002.
Murr, L. E., K. M. Garza, K. F. Soto, A. Carrasco, T. G. Powell, D. A. Ramirez, P. A. Guerrero, D. A. Lopez, and J. Venzor. Cytotoxicity assessment of some carbon nanotubes and related carbon nanoparticle aggregates and the implications for anthropogenic carbon nanotube aggregates in the environment. Int. J. Environ. Res. Public Health 2:31–42, 2005.
Osmond-McLeod, M. J., C. A. Poland, F. Murphy, L. Waddington, H. Morris, S. C. Hawkins, S. Clark, R. Aitken, M. J. McCall, and K. Donaldson. Durability and inflammogenic impact of carbon nanotubes compared with asbestos fibres. Part. Fibre Toxicol. 8:15, 2011.
Pacurari, M., V. Castranova, and V. Vallyathan. Single- and multi-wall carbon nanotubes versus asbestos: are the carbon nanotubes a new health risk to humans? J. Toxicol. Environ. Health A 73:378–395, 2010.
Pacurari, M., Y. Qian, D. Porter, M. Wolfarth, Y. Wan, D. Luo, M. Ding, V. Castranova, and N. Guo. Multi-walled carbon nanotube-induced gene expression in the mouse lung: association with lung pathology. Toxicol. Appl. Pharmacol. 255:18–31, 2011.
Polascik, T. J. Ablation of renal tumors in a rabbit model with interstitial saline-augmented radiofrequency energy—reply by the authors. Urology 54:382–383, 1999.
Ravichandran, P., S. Baluchamy, B. Sadanandan, R. Gopikrishnan, S. Biradar, V. Ramesh, J. C. Hall, and G. T. Ramesh. Multiwalled carbon nanotubes activate NF-kappa B and AP-1 signaling pathways to induce apoptosis in rat lung epithelial cells. Apoptosis 15:1507–1516, 2010.
Rendon, R. A., M. R. Gertner, M. D. Sherar, M. R. Asch, J. R. Kachura, J. Sweet, and M. A. S. Jewett. Development of a radiofrequency based thermal therapy technique in an in vivo porcine model for the treatment of small renal masses. J. Urol. 166:292–298, 2001.
Rendon, R. A., J. R. Kachura, J. M. Sweet, M. R. Gertner, M. D. Sherar, M. Robinette, J. Tsihlias, J. Trachtenberg, H. Sampson, and M. A. S. Jewett. The uncertainty of radio frequency treatment of renal cell carcinoma: findings at immediate and delayed nephrectomy. J. Urol. 167:1587–1592, 2002.
Rodemann, H. P., A. Binder, A. Burger, N. Guven, H. Loffler, and M. Bamberg. The underlying cellular mechanism of fibrosis. Kidney Int. Suppl. 54:S32–S36, 1996.
Rolland, P. H., A. B. Charifi, C. Verrier, H. Bodard, A. Friggi, P. Piquet, G. Moulin, and J. M. Bartoli. Hemodynamics and wall mechanics after stent placement in swine iliac arteries: comparative results from six stent designs. Radiology 213:229–246, 1999.
Rolland, P. H., V. Vidal, C. Mekkaoui, M. F. Bertrand, O. Levrier, and J. M. Bartoli. Embolization-driven occlusion of the abdominal aortic aneurysmal sac as the basis of prevention of endoleaks in a new swine model. Eur. J. Vasc. Endovasc. Surg. 31:28–35, 2006.
Savolainen, K., L. Pylkkanen, H. Norppa, G. Falck, H. Lindberg, T. Tuomi, M. Vippola, H. Alenius, K. Hameri, J. Koivisto, D. Brouwer, D. Mark, D. Bard, M. Berges, E. Jankowska, M. Posniak, P. Farmer, R. Singh, F. Krombach, P. Bihari, G. Kasper, and M. Seipenbusch. Nanotechnologies, engineered nanomaterials and occupational health and safety—a review. Saf. Sci. 48:957–963, 2010.
Shuba, M. V., G. Y. Slepyan, S. A. Maksimenko, and G. W. Hanson. Radiofrequency field absorption by carbon nanotubes embedded in a conductive host. J. Appl. Phys. 108:114302, 2010.
Sohaebuddin, S. K., P. T. Thevenot, D. Baker, J. W. Eaton, and L. Tang. Nanomaterial cytotoxicity is composition, size, and cell type dependent. Part. Fibre Toxicol. 7:22, 2010.
Sommer, C., N. Kortes, S. Zelzer, F. Arnegger, U. Stampfl, N. Bellemann, T. Gehrig, F. Nickel, H. Kenngott, C. Mogler, T. Longerich, H. Meinzer, G. Richter, H. Kauczor, and B. Radeleff. Renal artery embolization combined with radiofrequency ablation in a porcine kidney model: effect of small and narrowly calibrated microparticles as embolization material on coagulation diameter, volume, and shape. Cardiovasc. Intervent. Radiol. 34:156–165, 2011.
Terrones, M., P. M. Ajayan, F. Banhart, X. Blase, D. L. Carroll, J. C. Charlier, R. Czerw, B. Foley, N. Grobert, R. Kamalakaran, P. Kohler-Redlich, M. Ruhle, T. Seeger, and H. Terrones. N-doping and coalescence of carbon nanotubes: synthesis and electronic properties. Appl. Phys. A 74:355–361, 2002.
Torti, S. V., F. Byrne, O. Whelan, N. Levi, B. Ucer, M. Schmid, F. M. Torti, S. Akman, J. Liu, P. M. Ajayan, O. Nalamasu, and D. L. Carroll. Thermal ablation therapeutics based on CN(x) multi-walled nanotubes. Int. J. Nanomed. 2:707–714, 2007.
Vidal, V., P. H. Rolland, L. Daniel, G. Moulin, J. M. Bartoli, and O. Levrier. Effectiveness of endovascular embolization with a collagen-based embolic agent (marsembol) in an animal model. J. Vasc. Interv. Radiol. 21:1419–1423, 2010.
Wang, L., V. Castranova, A. Mishra, B. Chen, R. R. Mercer, D. Schwegler-Berry, and Y. Rojanasakul. Dispersion of single-walled carbon nanotubes by a natural lung surfactant for pulmonary in vitro and in vivo toxicity studies. Part. Fibre Toxicol. 7:31, 2010.
Wirnitzer, U., B. Herbold, M. Voetz, and J. Ragot. Studies on the in vitro genotoxicity of baytubes (R), agglomerates of engineered multi-walled carbon-nanotubes (MWCNT). Toxicol. Lett. 186:160–165, 2009.
Yang, C., V. Kaushal, S. V. Shah, and G. P. Kaushal. Autophagy is associated with apoptosis in cisplatin injury to renal tubular epithelial cells. Am. J. Physiol. Renal Physiol. 294:F777–F787, 2008.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Agata A. Exner oversaw the review of this article.
Rights and permissions
About this article
Cite this article
Rolland, P.H., Berry, J.L., Louis, G. et al. A Nanoengineered Embolic Agent for Precise Radiofrequency Ablation. Ann Biomed Eng 42, 940–949 (2014). https://doi.org/10.1007/s10439-014-0977-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-014-0977-9