Abstract
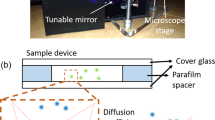
This paper reports the in situ detection of C-reactive protein (CRP) and antibody-coated nanobead interaction by measuring the variation of the Brownian diffusion of the nanobeads. The measurements are carried out by using total internal reflective fluorescence-enhanced (TIRF-enhanced) micro-particle-tracking velocimetry (micro-PTV), which is highly sensitive for analyzing nanobead movements in the near-wall region. The characteristics of evanescent wave penetration of various liquid samples, which have different refractive indices, are investigated for nanobead illumination. The Brownian velocities of the nanobeads in various concentrations of CRPs within glycerol solution and serum are measured in real time. The detectable CRP concentration range of 1–6 mg/ml in human serum is found, and the detecting process can be finished in 10 min. From the results, the dissociation constant of CRP and anti-CRP in various viscosity solutions can be determined through the developed kinetic analysis.







Similar content being viewed by others
References
Brenner H (1961) The slow motion of a sphere through a viscous fluid towards a plane surface. Chem Eng Sci 16:242–251
Burmeister JS, Olivier LA, Reichert WM, Truskey GA (1998) Application of total internal reflection fluorescence microscopy to study cell adhesion to biomaterials. Biomaterials 19:307–325
Cao Y et al (2006) Preparation of silica encapsulated quantum dot encoded beads for multiplex assay and its properties. Anal Chem 351:193–200
Choi CK, Margraves CH, Kihm KD et al (2007) Examination of near-wall hindered Brownian diffusion of nanoparticles: experimental comparison to theories by Brenner (1961) and Goldman et al. (1967). Phys Fluids 19:103305
Devadhasan JP, Marimuthu M, Kim S, Kim M-G (2012) A CMOS image sensor to recognize the cardiovascular disease markers troponin I and C-reactive protein. Anal Bioanal Chem 402:813–821
Einstein A (1905) Investigations on the theory of the Brownian movement. Annu Phys 17:549–560
Fan YJ, Sheen HJ, Hsu CJ, Liu CP, Lin S, Wu KC (2009) A quantitative immunosensing technique based on the measurement of nanobeads’ Brownian motion. Biosens Bioelectron 25:688–694
Fan YJ, Sheen HJ, Liu YH, Tsai JF, Wu TH, Wu KC, Lin S (2010) Detection of C-reactive protein in evanescent wave field using microparticle-tracking velocimetry. Langmuir 26:13751–13754
Goldman AJ, Cox RG, Brenner H (1967) Slow viscous motion of a sphere parallel to a plane wall. I. Motion through a quiescent fluid. Chem Eng Sci 22:637–651
Gorti VM, Shang H, Wereley ST, Lee GU (2008) Immunoassays in nanoliter volume reactors using fluorescent particle diffusometry. Langmuir 24:2947–2952
Hu WP, Hsu HY, Chiou A, Tseng K, Lin HY, Chang GL, Chen SJ (2006) Immunodetection of pentamer and modified C-reactive protein using surface plasmon resonance biosensing. Biosens Bioelectron 21:1631–1637
Huang P, Breuer KS (2007) Direct measurement of slip length in electrolyte solutions. Phys Fluids 19:028104
Jin S, Huang P, Park J, Yoo JY, Breuer KS (2004) Near-surface velocimetry using evanescent wave illumination. Exp Fluids 37:825–833
Kihm KD, Banerjee A, Choi CK, Takagi T (2004) Near-wall hindered Brownian diffusion of nanoparticles examined by three-dimensional ratiometric total internal reflection fluorescence microscopy (3-D R-TIRFM). Exp Fluids 37:811–824
Kim JY, Choi K, Moon DI, Ahn JH, Park TJ, Lee SY, Choi YK (2013) Surface engineering for enhancement of sensitivity in an underlap-FET biosensor by control of wettability. Biosens Bioelectron 41:867–870
Kulin S, Kishore R, Hubbard JB, Helmerson K (2002) Real-time measurement of spontaneous antigen–antibody dissociation. Biophys J 83:1965–1973
Kurosawa S, Nakamura M, Park J, Aizawa H, Yamada K, Hirata M (2004) Evaluation of a high-affinity QCM immunosensor using antibody fragmentation and 2-methacryloyloxyethyl phosphorylcholine (MPC) polymer. Biosens Bioelectron 20:1134–1139
Langevin P (1908) Theory of Brownian movement. CR Acad Sci 146:530
Lin B, Yu J, Rice SA (2000) Direct measurements of constrained Brownian motion of an isolated sphere between two walls. Phys Rev E 62:3909–3919
Lin S et al (2006a) Measurement of dimensions of pentagonal doughnut-shaped C-reactive protein using an atomic force microscope and a dual polarisation interferometric biosensor. Biosens Bioelectron 22:323–327
Lin S, Lee CK, Lin YH, Lee SY, Sheu BC, Tsai JC, Hsu SM (2006b) Homopolyvalent antibody–antigen interaction kinetic studies with use of a dual-polarization interferometric biosensor. Biosens Bioelectron 22:715–721
Margraves CH, Choi CK, Kihm KD (2006) Measurements of the minimum elevation of nano-particles by 3D nanoscale tracking using ratiometric evanescent wave imaging. Exp Fluids 41:173–183
Meyer M, Hartmann M, Keusgen M (2006) SPR-based immunosensor for the CRP detection–a new method to detect a well known protein. Biosens Bioelectron 21:1987–1990
Pultar J, Sauer U, Domnanich P, Preininger C (2009) Aptamer–antibody on-chip sandwich immunoassay for detection of CRP in spiked serum. Biosens Bioelectron 24:1456–1461
Rocha SM, Suzuki LA, Silva ADT, Arruda GC, Rossi CL (2002) A rapid latex agglutination test for the detection of anti-cysticercus antibodies in cerebrospinal fluid (CSF). Rev Inst Med Trop Sao Paulo 44:57–58
Thompson PA, Troian SM (1997) A general boundary condition for liquid flow at solid surfaces. Nature 389:360–362
Toomre D, Manstein DJ (2001) Lighting up the cell surface with evanescent wave microscopy. Trends Cell Biol 11:298–303
Tracy RP (2003) Emerging relationships of inflammation, cardiovascular disease and chronic diseases of aging. Int J Obes 27:S29–S34
Tsai H, Hsu C, Chiu I, Fuh C (2007) Detection of C-reactive protein based on immunoassay using antibody-conjugated magnetic nanoparticles. Anal Chem 79:8416–8419
Wee K, Kang G, Park J, Kang J, Yoon D, Park J, Kim T (2005) Novel electrical detection of label-free disease marker proteins using piezoresistive self-sensing micro-cantilevers. Biosens Bioelectron 20:1932–1938
Wu TL, Tsao KC, Chang CPY, Li CN, Sun CF, Wu JT (2002) Development of ELISA on microplate for serum C-reactive protein and establishment of age-dependent normal reference range. Clin Chim Acta 322:163–168
Zettner C, Yoda M (2003) Particle velocity field measurements in a near-wall flow using evanescent wave illumination. Exp Fluids 34:115–121
Acknowledgments
This work was supported by National Science Council of Taiwan under grant numbers NSC 102-2627-E-002-002 and NSC 100-2221-E-002-107-MY3.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Fan, YJ., Sheen, HJ., Chen, ZY. et al. TIRF-enhanced nanobeads’ Brownian diffusion measurements for detecting CRP in human serum. Microfluid Nanofluid 19, 85–94 (2015). https://doi.org/10.1007/s10404-015-1551-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10404-015-1551-y