Abstract
Purpose
To evaluate the changes of orbital tissue volumes and proptosis after methylprednisolone pulse therapy in thyroid-associated ophthalmopathy (TAO).
Methods
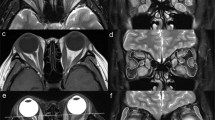
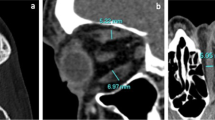
The cross-sectional areas of orbital tissues and proptosis were measured with magnetic resonance imaging in 40 orbits of 20 patients with TAO before and after methylprednisolone pulse therapy. The volumes of the whole orbit, orbital fatty tissue, and extraocular muscles were calculated. The volumes and proptosis were compared before and after treatment using a paired t test.
Results
Before treatment, the mean volumes were 33.0 ± 4.8 cm3 in the whole orbit, 19.9 ± 4.1 cm3 in the orbital fatty tissue, and 4.6 ± 1.2 cm3 in the total extraocular muscles. After treatment, the mean volumes were 32.5 ± 4.4 cm3 in the whole orbit, 19.9 ± 3.7 cm3 in the orbital fatty tissue, and 4.0 ± 1.0 cm3 in the total extraocular muscles. The mean volumes of the whole orbit (P = 0.17) and orbital fatty tissue (P = 0.82) were not significantly decreased after treatment, while the mean volume of total extraocular muscles was significantly decreased (P < 0.001). The mean proptosis value was 18.9 ± 2.8 mm before treatment and 18.6 ± 3.4 mm after treatment. The mean proptosis value was not significantly decreased after treatment (P = 0.30).
Conclusions
The volume of orbital fatty tissue seemed to be unchanged after methylprednisolone pulse therapy while that of total extraocular muscles was decreased. The proptosis value seemed to be unchanged after treatment.








Similar content being viewed by others
References
Bahn RS. Thyrotropin receptor expression in orbital adipose/connective tissues from patients with thyroid-associated ophthalmopathy. Thyroid. 2002;12:193–5.
Bahn RS. Graves’ ophthalmopathy. N Engl J Med. 2010;362:726–38.
Nishida Y, Tian S, Isberg B, Tallstedt L, Lennerstrand G. MRI measurements of orbital tissues in dysthyroid ophthalmopathy. Graefes Arch Clin Exp Ophthalmol. 2001;239:824–31.
Burch HB, Wartofsky L. Graves’ ophthalmopathy: current concepts regarding pathogenesis and management. Endocr Rev. 1993;14:747–93.
Hatton MP, Rubin PA. The pathophysiology of thyroid’associated ophthalmopathy. Ophthalmol Clin North Am. 2002;15:113–9.
Lehmann GM, Feldon SE, Smith TJ, Phipps RP. Immune mechanisms in thyroid eye disease. Thyroid. 2008;18:959–65.
Wiersinga WM, Regensburg NI, Mourits MP. Differential involvement of orbital fat and extraocular muscles in Graves’ ophthalmopathy. Eur Thyroid J. 2013;2:14–21.
Nishida Y, Tian S, Isberg B, Hayashi O, Tallstedt L, Lennerstrand G. Significance of orbital fatty tissue for exophthalmos in thyroid-associated ophthalmopathy. Graefes Arch Clin Exp Ophthalmol. 2002;240:515–20.
Bartalena L, Baldeschi L, Dickinson A, Eckstein A, Kendall-Taylor P, Marcocci C, et al. Consensus statement of the European group on Graves’ orbitopathy (EUGOGO) on management of GO. Eur J Endocrinol. 2008;158:273–85.
Bartalena L, Marcocci C, Bogazzi F, Bruno-Bossio G, Pinchera A. Glucocorticoid therapy of Graves’ ophthalmopathy. Exp Clin Endocrinol. 1991;97:320–7.
Bartalena L, Pinchera A, Marcocci C. Management of Graves’ ophthalmopathy: reality and perspectives. Endocr Rev. 2000;21:168–99.
Kauppinen-Mäkelin R, Karma A, Leinonen E, Löyttyniemi E, Salonen O, Sane T, et al. High dose intravenous methylprednisolone pulse therapy versus oral prednisone for thyroid-associated ophthalmopathy. Acta Ophthalmol Scand. 2002;80:316–21.
Ohtsuka K, Sato A, Kawaguchi S, Hashimoto M, Suzuki Y. Effect of high-dose intravenous steroid pulse therapy followed by 3-month oral steroid therapy for Graves’ ophthalmopathy. Jpn J Ophthalmol. 2002;46:563–7.
Van Geest RJ, Sasim IV, Koppeschaar HP, Kalmann R, Stravers SN, Bijlsma WR, et al. Methylprednisolone pulse therapy for patients with moderately severe Graves’ orbitopathy: a prospective, randomized, placebo-controlled study. Eur J Endocrinol. 2008;158:229–37.
Higashiyama T, Nishida Y, Morino K, Ugi S, Nishio Y, Maegawa H, et al. Use of MRI signal intensity of extraocular muscles to evaluate methylprednisolone pulse therapy in thyroid-associated ophthalmopathy. Jpn J Ophthalmol. 2015;59:124–30.
Schneider CA, Rasband WS, Eliceiri KW. NIH image to imageJ: 25 years of image analysis. Nat Methods. 2012;9:671–5.
Peyster RG, Ginsberg F, Silber JH, Adler LP. Exophthalmos caused by excessive fat: CT volumetric analysis and differential diagnosis. AJR Am J Roentgenol. 1986;146:459–64.
Tachibana S, Murakami T, Noguchi H, Noguchi Y, Nakashima A, Ohyabu Y, et al. Orbital magnetic resonance imaging combined with clinical activity score can improve the sensitivity of detection of disease activity and prediction of response to immunosuppressive therapy for Graves’ ophthalmopathy. Endocr J. 2010;57:853–61.
Hiromatsu Y, Eguchi H, Tani J, Kasaoka M, Teshima Y. Graves’ ophthalmopathy: epidemiology and natural history. Intern Med. 2014;53:353–60.
Acknowledgments
Supported in part by a grant from the Ministry of Education, Culture, Sports, Science and Technology of Japan (#21592255) and a grant from the Ministry of Health, Labour and Welfare.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
T. Higashiyama, Lecture fees (Senju Pharmaceutical); Y. Nishida, Lecture fees (Senju Pharmaceutical); M. Ohji, Lecture fees (Senju Pharmaceutical, Carl Zeiss Japan, Otsuka Pharmaceutical, MSD, Kowa Pharmaceutical, Alcon Japan, Novartis Pharmaceutical, Senju Pharmaceutical, Bayer, Shionogi & Co.), Consultant fees (Alcon Japan, Novartis Pharmaceutical, Santen Pharmaceutical, Bayer, Shionogi & CO., Pfizer Japan, Sanwa Kagaku Institute).
About this article
Cite this article
Higashiyama, T., Nishida, Y. & Ohji, M. Changes of orbital tissue volumes and proptosis in patients with thyroid extraocular muscle swelling after methylprednisolone pulse therapy. Jpn J Ophthalmol 59, 430–435 (2015). https://doi.org/10.1007/s10384-015-0410-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10384-015-0410-4