Abstract
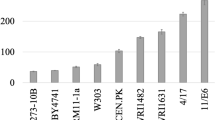
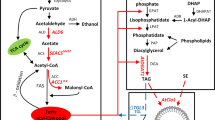
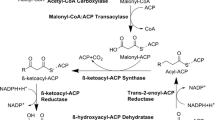
Fatty acid ethyl esters are fatty acid derived molecules similar to first generation biodiesel (fatty acid methyl esters; FAMEs) which can be produced in a microbial cell factory. Saccharomyces cerevisiae is a suitable candidate for microbial large scale and long term cultivations, which is the typical industrial production setting for biofuels. It is crucial to conserve the metabolic design of the cell factory during industrial cultivation conditions that require extensive propagation. Genetic modifications therefore have to be introduced in a stable manner. Here, several metabolic engineering strategies for improved production of fatty acid ethyl esters in S. cerevisiae were combined and the genes were stably expressed from the organisms’ chromosomes. A wax ester synthase (ws2) was expressed in different yeast strains with an engineered acetyl-CoA and fatty acid metabolism. Thus, we compared expression of ws2 with and without overexpression of alcohol dehydrogenase (ADH2), acetaldehyde dehydrogenase (ALD6) and acetyl-CoA synthetase (acs L641P SE ) and further evaluated additional overexpression of a mutant version of acetyl-CoA decarboxylase (ACC1 S1157A,S659A ) and the acyl-CoA binding protein (ACB1). The combined engineering efforts of the implementation of ws2, ADH2, ALD6 and acs L641P SE , ACC1 S1157A,S659A and ACB1 in a S. cerevisiae strain lacking storage lipid formation (are1Δ, are2Δ, dga1Δ and lro1Δ) and β-oxidation (pox1Δ) resulted in a 4.1-fold improvement compared with sole expression of ws2 in S. cerevisiae.



Similar content being viewed by others
References
Chen Y, Daviet L, Schalk M, Siewers V, Nielsen J (2013) Establishing a platform cell factory through engineering of yeast acetyl-CoA metabolism. Metab Eng 15:48–54
Chen Y, Siewers V, Nielsen J (2012) Profiling of cytosolic and peroxisomal acetyl-CoA metabolism in Saccharomyces cerevisiae. PLoS One 7:e42475
Choi JW, Da Silva NA (2014) Improving polyketide and fatty acid synthesis by engineering of the yeast acetyl-CoA carboxylase. J Biotechnol 187:56–59
de Jong B, Siewers V, Nielsen J (2012) Systems biology of yeast: enabling technology for development of cell factories for production of advanced biofuels. Curr Opin Biotechnol 23:624–630
de Jong BW, Shi S, Siewers V, Nielsen J (2014) Improved production of fatty acid ethyl esters in Saccharomyces cerevisiae through up-regulation of the ethanol degradation pathway and expression of the heterologous phosphoketolase pathway. Microb Cell Fact 13(1):39
de Kroon AIPM, Rijken PJ, De Smet CH (2013) Checks and balances in membrane phospholipid class and acyl chain homeostasis, the yeast perspective. Prog Lipid Res 52(4):374–394
Garbarino J, Padamsee M, Wilcox L, Oelkers PM, D’Ambrosio D, Ruggles KV, Ramsey N, Jabado O, Turkish A, Sturley SL (2009) Sterol and diacylglycerol acyltransferase deficiency triggers fatty acid-mediated cell death. J Biol Chem 284(45):30994–31005
Hauf J, Zimmermann FK, Muller S (2000) Simultaneous genomic overexpression of seven glycolytic enzymes in the yeast Saccharomyces cerevisiae. Enzym Microb Technol 26(9–10):688–698
Hofbauer HF, Schopf FH, Schleifer H, Knittelfelder OL, Pieber B, Rechberger GN, Wolinski H, Gaspar ML, Kappe CO, Stadlmann J, Mechtler K, Zenz A, Lohner K, Tehlivets O, Henry SA, Kohlwein SD (2014) Regulation of gene expression through a transcriptional repressor that senses acyl-chain length in membrane phospholipids. Dev Cell 29(6):729–739
Hong S-P, Xue Z (2008) Manipulation of acyl-CoA binding protein expression for altered lipid production in microbial hosts. Patent: US20090291479 A1
Jensen NB, Strucko T, Kildegaard KR, David F, Maury J, Mortensen UH, Forster J, Nielsen J, Borodina I (2013) EasyClone: method for iterative chromosomal integration of multiple genes in Saccharomyces cerevisiae. FEMS Yeast Res 14(2):238–248
Kalscheuer R, Luftmann H, Steinbuchel A (2004) Synthesis of novel lipids in Saccharomyces cerevisiae by heterologous expression of an unspecific bacterial acyltransferase. Appl Environ Microbiol 70:7119–7125
Kalscheuer R, Stolting T, Steinbuchel A (2006) Microdiesel: escherichia coli engineered for fuel production. Microbiology 152:2529–2536
Keasling JD (2012) Synthetic biology and the development of tools for metabolic engineering. Metab Eng 14(3):189–195
Knudsen J, Faergeman NJ, Skott H, Hummel R, Borsting C, Rose TM, Andersen JS, Hojrup P, Roepstorff P, Kristiansen K (1994) Yeast acyl-CoA-binding protein: acyl-CoA-binding affinity and effect on intracellular acyl-CoA pool size. Biochem J 302:479–485
Knudsen J, Jensen MV, Hansen JK, Faergeman NJ, Neergaard TBF, Gaigg B (1999) Role of acylCoA binding protein in acylCoA transport, metabolism and cell signaling. Mol Cell Biochem 192(1–2):95–103
Kocharin K, Chen Y, Siewers V, Nielsen J (2012) Engineering of acetyl-CoA metabolism for the improved production of polyhydroxybutyrate in Saccharomyces cerevisiae. AMB Express 2:52
Lee FWF, DaSilva NA (1997) Improved efficiency and stability of multiple cloned gene insertions at the delta sequences of Saccharomyces cerevisiae. Appl Microbiol Biotechnol 48(3):339–345
Mikkelsen MD, Buron LD, Salomonsen B, Olsen CE, Hansen BG, Mortensen UH, Halkier BA (2012) Microbial production of indolylglucosinolate through engineering of a multi-gene pathway in a versatile yeast expression platform. Metab Eng 14(2):104–111
Okuyama H, Saito M, Joshi VC, Gunsberg S, Wakil SJ (1979) Regulation by temperature of the chain-lenght of fatty-acids in yeast. J Biol Chem 254(24):2281–2284
Petschnigg J, Wolinski H, Kolb D, Zellnig G, Kurat CF, Natter K, Kohlwein SD (2009) Good fat, essential cellular requirements for triacylglycerol synthesis to maintain membrane homeostasis in yeast. J Biol Chem 284(45):30981–30993
Rasmussen JT, Rosendal J, Knudsen J (1993) Interaction of acyl-CoA-binding protein (Acbp) on processes for which acyl-CoA is a substrate, product or inhibitor. Biochem J 292:907–913
Runguphan W, Keasling JD (2014) Metabolic engineering of Saccharomyces cerevisiae for production of fatty acid-derived biofuels and chemicals. Metab Eng 21:103–113
Sambrook J, Russell DW (2001) Molecular cloning: a laboratory manual. In: Argentine J (ed), 3rd ed. Cold Spring Harbor Laboratory Press, New York, ISBN 0-87969-87579-87965
Shi S, Chen Y, Siewers V, Nielsen J (2014) Improving production of malonyl coenzyme A-derived metabolites by abolishing Snf1-dependent regulation of Acc1. MBio 5(3):e01130–e01140
Shi S, Valle-Rodriguez JO, Khoomrung S, Siewers V, Nielsen J (2012) Functional expression and characterization of five wax ester synthases in Saccharomyces cerevisiae and their utility for biodiesel production. Biotechnol Biofuels 5:7
Shi S, Valle-Rodriguez JO, Siewers V, Nielsen J (2014) Engineering of chromosomal wax ester synthase integrated Saccharomyces cerevisiae mutants for improved biosynthesis of fatty acid ethyl esters. Biotechnol Bioeng 111(9):1740–1747
Shiba Y, Paradise EM, Kirby J, Ro D-K, Keasling JD (2007) Engineering of the pyruvate dehydrogenase bypass in Saccharomyces cerevisiae for high-level production of isoprenoids. Metab Eng 9(2):160–168
Steen EJ, Kang YS, Bokinsky G, Hu ZH, Schirmer A, McClure A, del Cardayre SB, Keasling JD (2010) Microbial production of fatty-acid-derived fuels and chemicals from plant biomass. Nature 463(7280):559–562
Stoveken T, Kalscheuer R, Malkus U, Reichelt R, Steinbuchel A (2005) The wax ester synthase/acyl coenzyme A : diacylglycerol acyltransferase from Acinetobacter sp strain ADP1: characterization of a novel type of acyltransferase. J Bacteriol 187(4):1369–1376
Thompson RA, Trinh CT (2014) Enhancing fatty acid ethyl ester production in Saccharomyces cerevisiae through metabolic engineering and medium optimization. Biotechnol Bioeng 111(11):2200–2208
Tyo KEJ, Ajikumar PK, Stephanopoulos G (2009) Stabilized gene duplication enables long-term selection-free heterologous pathway expression. Nature Biotechnol 27(8):760–765
Valle-Rodriguez JO, Shi S, Siewers V, Nielsen J (2014) Metabolic engineering of Saccharomyces cerevisiae for production of fatty acid ethyl esters, an advanced biofuel, by eliminating non-essential fatty acid utilization pathways. Appl Energy 115:226–232
Wang XH, Wang ZJ, DaSilva NA (1996) G418 selection and stability of cloned genes integrated at chromosomal delta sequences of Saccharomyces cerevisiae. Biotechnol Bioeng 49(1):45–51
Woods A, Munday MR, Scott J, Yang XL, Carlson M, Carling D (1994) Yeast snf1 is functionally related to mammalian AMP-activated protein-kinase and regulates acetyl-CoA carboxylase in-vivo. J Biol Chem 269(30):19509–19515
Zhou YJJ, Gao W, Rong QX, Jin GJ, Chu HY, Liu WJ, Yang W, Zhu ZW, Li GH, Zhu GF, Huang LQ, Zhao ZBK (2012) Modular pathway engineering of diterpenoid synthases and the mevalonic acid pathway for miltiradiene production. J Am Chem Soc 134(6):3234–3241
Acknowledgments
This research has been funded by the Knut and Alice Wallenberg Foundation, Vetenskapsrådet, FORMAS, and Ångpanneföreningens Forskningsstiftelse.
Author information
Authors and Affiliations
Corresponding author
Additional information
Special Issue: Metabolic Engineering.
Rights and permissions
About this article
Cite this article
de Jong, B.W., Shi, S., Valle-Rodríguez, J.O. et al. Metabolic pathway engineering for fatty acid ethyl ester production in Saccharomyces cerevisiae using stable chromosomal integration. J Ind Microbiol Biotechnol 42, 477–486 (2015). https://doi.org/10.1007/s10295-014-1540-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-014-1540-2