Abstract
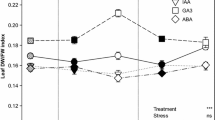
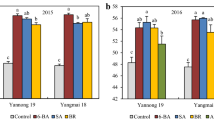
The effect of the auxin physiological analogues analogues 1-[2-chloroethoxycarbonylmethyl]-4-naphthalenesulfonic acid calcium salt (TA-12) and 1-[2-dimethylaminoethoxicarbonylmethyl]naphthalene chlormethylate (TA-14) TA-14 on different winter rapeseed cultivars were studied with regard to their autumnal growth, cold hardening, accumulation of the stress-protective metabolites proline and saccharide in plant organs: apical bud and root collum, winter survival and productivity formation. The test cultivars were the very early ‘Libea’ medium-resistant to wintering, the medium-early ‘Sunday’ resistant to wintering, the medium–early ‘Valesca’ less than medium resistant to wintering, and the early ‘Hornet’ (hybrid) tolerant to stress growth conditions. During the period of cold hardening in natural field conditions, the test compounds TA-12 (2 mM) and TA-14 (4 mM), applied to different winter rapeseed cultivars at the 4th–5th leaf stage, stimulate accumulation proline and saccharides (sucrose and glucose) in the root collum and apical bud tissues, influence plants acclimation to cold, overwintering and productivity formation. Compounds TA-12 and especially TA-14 produced a stable effect on seed and crude fat yield in cvs. ‘Hornet’, ‘Sunday’ and ‘Libea’. The genotypic peculiarities of a cultivar and the meteorological conditions of the plant vegetation period were the factors that mostly determined fatty acid content in seed oil.





Similar content being viewed by others
Abbreviations
- TA-12:
-
1-[2-Chloroethoxycarbonyl-methyl]-4-naphthalenesulfonic acid calcium salt
- TA-14:
-
1-[2-Dimethylaminoethoxicarbonylmethyl]naphthalene chlormethylate
- DM:
-
Dry mass
- DW:
-
Dry weight
References
Bates LS, Waldren RP, Teare ID (1973) Rapid determination of free proline content for water-stress studies. Plant Soil 39:205–207
Bravo LA, Zúñiga GE, Alberdi M, Corcuera LJ (1998) The role of ABA in freezing tolerance and cold acclimation in barley. Physiol Plant 103:17–23
Burbulis N, Kuprienė R, Blinstrubienė A (2008) Investigation of cold resistance of winter rapeseed in vitro. Sodininkystė ir daržininkystė 27:223–232
Butkutė B (2004) Factors influencing accuracy of NIR calibrations for the prediction of quality of Lithuania grown rapeseed. In: Davies AMC, Garrido-Varo A (eds) Near infrared spectroscopy. Proceedings of the 11th international conference on near infrared spectroscopy. NIR publications, Chichester, UK, pp 405–410
Christopherson SW, Glass RL (1969) Preparation of milk fat methylesters by alcoholysis in an essentially non alcoholic solution. J Diary Sci 52:1289–1290
Crowe JH, Carpenter FF, Crowe LM, Anchordoguy TJ (1990) Are freezing and dehydration similar stress vectors? A comparison of modes of interaction of stabilizing solutes with biomolecules. Cryobiol 27:219–231
Cuevas JC, López-Cobollo R, Alcázar R, Zarza X, Koncz C, Altabella T, Salinas J, Tiburcio AF, Ferrando A (2008) Putrescine is involved in Arabidopsis freezing tolerance and cold acclimation by regulating abscisic acid levels in response to low temperature. Plant Physiol 148:1094–1105
Cuevas JC, López-Cobollo R, Alcázar R, Zarza X, Koncz C, Altabella T, Salinas J, Tiburcio AF, Ferrando A (2009) Putrescine as a signal to modulate the indispensable ABA increase under cold stress. Plant Signal Behav 4:219–220 (PMCID: PMC2652534)
Dionne J, Castonguary Y, Nadeau P, Desjardins Y (2001) Amino acids and protein changes during cold acclimation of green-type annual bluegrass (Poa annua L.) ecotypes. Crop Sci 41:1862–1870
Folch J, Less M, Sloanc-Stanly GH (1957) A simple method for isolation and purification of total lipids from animal tissues. J Biol Chem 226:497–509
Gavelienė V, Novickienė L, Miliuvienė L (2007) Improving of oilseed rape lateral root formation by physiological analogues of auxin. Acta Physiol Plant 29:291–295
Gilmour SJ, Hajela RK, Thomashow MF (1988) Cold acclimation in Arabidopsis thaliana. Plant Physiol 87:745–750
Gothandam KM, Nalini E, Karthikeyan S, Shin JS (2010) OsPRP3, a flower specific proline-rich protein of rice, determines extracellular matrix structure of floral organs and its overexpression confers cold-tolerance. Plant Mol Biol 72:125–135
Hunter JE (1990) n-3 fatty acids from vegetable oils. Am J Clin Nutr 51:809–814
Klotke J, Kopta J, Gatzke N, Heyer AG (2004) Impact of soluble sugar concentrations on the acquisition of freezing tolerance in accessions of Arabidopsis thaliana with contrasting cold adaptation—evidence for a role of raffinose in cold acclimation. Plant Cell Environ 27:1395–1404
Lecomte C, Girand A, Aubert V (2003) Testing a predicting model for frost resistance of winter wheat under natural conditions. Agronomie 23:51–66. doi:10.1051/agro:2002068
Mahajan S, Tuteja N (2005) Cold, salinity and drought stresses: an overview. Arch Biochem Biophys 444:139–158
McKown R, Kuroki G, Warren G (1996) Cold responses of Arabidopsis mutants impaired in freezing tolerance. J of Exp Botany 47:1919–1925
Meier U (ed) (2001) Growth stages of mono- and dicotyledonous plants. BBCH Monograph. Federal Biology Research Centre for Agriculture and Forestry. 2nd edn
Merkys A, Novickienė L, Darginavičienė J, Maksimov G (2007) Advantages of auxin analogues of plant growth and productivity regulators. Int J Environ Pollut 29:443–456
Miliuvienė L, Novickienė L, Gavelienė V (2003) Effect of 17-DMC on phytohormone level and growth of rapeseed plants (Brassica napus). Russ J Plant Physiol 50:656–660
Novickienė L, Jurevičius J (1997) The problem of plant growth regulation and prospects of its solution 1. Physiological analogues of auxin and peculiarities of their activity. Biology 3:61–68
Novickienė L, Darginavičienė J, Maksimov G (2004) Root initiation and development by auxin physiological analogue TA-12. Acta Univer Latviensis Biol 676:201–206. http://eeb.lu.lv/EEB/2004/Novickiene.pdf
Novickienė L, Gavelienė V, Miliuvienė L, Kazlauskienė D, Pakalniškytė L (2010) Analysis of lateral root growth in Arabidopsis in response to physiologically active auxin analogues. Acta Agronom Hungarica 58:1–10
Rademacher W (2000) Growth retardants: effect on Gibberellin Biosynthesis and other Metabolic Pathways. Annu Rev Plant Physiol Plant Mol Biol 51:501–531
Sakalauskaitė J, Brazaitytė A, Urbonavičiūtė A, Samuolienė G, Šabajevienė G, Sakalauskienė S, Duchovskis P (2010) Radish plant behaviour under short-term elevated ozone fumigation. Cent Eur J Biol 5:674–681. doi:10.2478/s11535-010-0057-6
Shibasaki K, Uemura M, Tsurumi S, Rahman A (2009) Auxin response in Arabidopsis under cold stress: underlying molecular mechanisms. [C], [W]. Plant Cell 21:3823–3838
Shinozaki K, Yamaguchi-Shinozaki K, Seki M (2003) Regulatory network of gene expression in the drought and cold stress responses. Curr Opin Plant Biol 6:410–417
Skuodienė L, Gradeckas A (2004) Skirtingomis sąlygomis augančių gluosnių fiziologinė būklė (Physiological state of willow species depending on the site conditions). Ekologija (Ecology) 2:1–7 (in Lithuanian)
Szalai G, Pap M, Janda T (2009) Light-induced frost tolerance differs in winter and spring wheat plants. J Plant Physiol 166:1826–1831
Thelen JJ, Ohlorogge JB (2002) Metabolic engineering of fatty acid biosynthesis in plants. Metab Eng 4:12–21
Thomashow MF (1999) Plant cold acclimation: freezing tolerance genes and regulatory mechanisms. Ann Rev Plant Biol 50:571–599
Uemura M, Gilmour SJ, Thomashow MF, Steponkus PL (1996) Effects of COR6.6 and COR15am polypeptides encoded by COR (cold-regulated) genes of Arabidopsis thaliana on the freeze-induced fusion and leakage of liposomes. Plant Physiol 111:313–327
Velička R, Rimkevičienė M, Novickienė L, Anisimovienė N, Brazauskienė I (2005) Improvement of oil rape hardening and frost tolerance. Russ J Plant Physiol 52:532–553
Velička R, Marcinkeviciene A, Raudonius S, Rimkeviciene M (2006) Integrated evoluation of rape readiness for overwintering. Acta Agric Scandinavica Soil Plant Sci 56:110–116
Verbruggen N, Hermans Ch (2008) Proline accumulation in plants: a review. Amino Acids 35:753–759
Veselov DS, Sabirzhanova I, Akhiyarova G, Veselova SV, Farkhutdinov RG, Mustafina AR, Mitrichenko AN, Dedov AV, Veselov SYu, Kudoyarova GR (2002) The role of hormones in fast growth responses of wheat plants to osmotic and cold shocks. Russ J Plant Physiol 49:513–517
Wanner LA, Junttila O (1999) Cold-induced freezing tolerance in Arabidopsis. Plant Physiol 120:391–400
Webb MS, Uemura M, Steponkus PL (1994) A comparison of freezing injury in oat and rye: two cereals at the extremes of freezing tolerance. Plant Physiol 104:467–478
Xiong L, Schumaker KS, Zhu JK (2002) Cell signalling during cold, draught, and salt stress. Plant Cell (suppl) 14:165–183
Zhang JZ, Creelman RA, Zhu JK (2004) From laboratory to field. Information from Arabidopsis to engineer salt, cold, and drought tolerance in crops. Plant Physiol 135:615–621
Acknowledgments
The study partly was supported by the Lithuanian Research Council under the project ‘Biokuras’ in 2008–2010.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gavelienė, V., Novickienė, L. & Pakalniškytė, L. Effect of auxin physiological analogues on rapeseed (Brassica napus) cold hardening, seed yield and quality. J Plant Res 126, 283–292 (2013). https://doi.org/10.1007/s10265-012-0525-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10265-012-0525-3