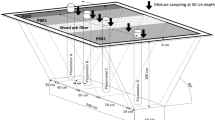
Abstract
Thermally modified pyrite (MPy) has submicron-sized particles and a porous polycrystalline structure. The ability of MPy to remove copper from simulated and actual acid mine drainage (AMD) was studied. Batch experiments showed good Cu2+ removal in the pH range of 4–6 in simulated AMD. Copper removal from actual AMD was investigated in an experiment in which column A was filled with limestone as the neutralizer and column B was filled with MPy. The MPy column had a removal capacity of 21.93 mg of Cu per g of MPy; the Cu content was as high as 9.23 % in the spent MPy particles. This indicates that MPy has the potential to be a novel cost-effective material for Cu removal and recovery from AMD.
Graphical Abstract

热力学改性黄铁矿去除酸性废水中铜:批次和柱试验
热力学改性的黄铁矿(MPy)具有亚微米级颗粒和多晶孔隙结构。利用人工合成酸性废水和实际酸性废水研究了MPy的铜去除能力。批次试验表明,人工合成酸性废水的pH值为4-6时, MPy的Cu2+去除效果较好。利用两个试验柱研究了实际酸性废水的铜去除效果,柱体A充填石灰石中和剂,柱体B充填MPy。MPy柱体的铜去除能力为每克MPy去除21.93毫克铜, 试验后的MPy颗粒铜含量高达9.23%。研究表明,MPy是一种能够有效去除和回收酸性废水中铜的新型材料。
Zusammenfassung
Thermisch modifizierter Pyrit (MPy) weist Partikelgrößen im Submikronbereich auf und besitzt eine poröse, polykristalline Struktur. Die Wirksamkeit von MPy, Kupfer aus synthetischem und realem Sauerwasser zu entfernen, wurde untersucht. Batchexperimente zeigten im synthetischen Sauerwasser eine gute Cu2+-Entfernung im pH-Bereich von 4-6. In einem Experiment mit realem Sauerwasser, bei dem die Kolonne A mit Kalkstein als Neutralisationsagent und die Kolonne B mit MPy gefüllt waren, wurde die Kupferentfernung untersucht. Die MPy Säule wies eine Entfernungskapazität von 21,93 mg Cu pro g MPy auf, und der Cu-Gehalt in den verbrauchten MPy-Partikeln betrug 9,23%. Dies ist ein Hinweis darauf, dass MPy ein neues potentielles und kostengünstiges Material für die Cu-Entfernung und -Rückgewinnung aus Sauerwasser zu sein scheint.
Resumen
La pirita modificada térmicamente (MPy) tiene partículas de tamaño sub-microscópico y una estructura policristalina porosa. Se estudió la capacidad de MPy para remover Cu desde drenaje ácido de mina (AMD) artificial y real. Los experimentos en batch mostraron una buena remoción de Cu2+ de AMD artificial, en el rango de 4-6. Se investigó la remoción de Cu desde AMD real en un experimento en el cuál la columna A se llenó con caliza como neutralizante y la columna B se llenó con MPy. La columna MPy tuvo una capacidad de remoción de 21,93 mg de Cu por g de MPy; el contenido de Cu fue 9,23% en las partículas agotadas de MPy. Esto sugiere que MPy tiene el potencial para ser un nuevo material económico y efectivo para la remoción y la recuperación de Cu desde AMD.





Similar content being viewed by others
References
Barr G, Defreyne J, Jones D, Mean R (2005) On-site processing vs. sale of copper concentrates. CESL, Perth, Australia. http://www.teck.com/media/CESL-Publication-Copper-On-Site-vs-Cu-Con-Sales-Alta-2005.pdf
Belzile N, Chen Y-W, Cai M-F, Li Y (2004) A review on pyrrhotite oxidation. J Geochem Explor 84:65–76
Bigham JM (1994) Mineralogy of ochre deposits formed by sulfide oxidation. In: Jambor JL, Blowes DW (eds), Short course handbook on environmental geochemistry of sulfide mine-wastes, vol 22. Mineralogical Assoc of Canada, Canada, pp 103–132
Burgos WD et al (2012) Schwertmannite and Fe oxides formed by biological low-pH Fe(II) oxidation versus abiotic neutralization: impact on trace metal sequestration. Geochim Cosmochim Acta 76:29–44
Chen TH, Yang Y, Chen D, Li P, Shi YD, Zhu X (2013) The structural evolution of heat-treated colloidal pyrite under inert atmosphere and its application for the purification of Cu(II) ion from wastewater. Environ Eng Manag J 12:1411–1416
Chiriţă P, Rimstidt JD (2014) Pyrrhotite dissolution in acidic media. Appl Geochem 41:1–10
Hughes TA, Gray NF, Sánchez Guillamón O (2013) Removal of metals and acidity from acid mine drainage using liquid and dried digested sewage sludge and cattle slurry. Mine Water Environ 32:108–120
Kalin M, Fyson A, Wheeler WN (2006) The chemistry of conventional and alternative treatment systems for the neutralization of acid mine drainage. Sci Total Environ 366:395–408
Kameda T, Yabuuchi F, Yoshioka T, Uchida M, Okuwaki A (2003) New method of treating dilute mineral acids using magnesium–aluminum oxide. Water Res 37:1545–1550
Maree JP, Mujuru M, Bologo V, Daniels N, Mpholoane D (2013) Neutralisation treatment of AMD at affordable cost. Water SA. doi:10.4314/wsa.v39i2.7
Motsi T, Rowson NA, Simmons MJH (2009) Adsorption of heavy metals from acid mine drainage by natural zeolite. Int J Miner Process 92:42–48
Ozverdi A, Erdem M (2006) Cu2+, Cd2+ and Pb2+ adsorption from aqueous solutions by pyrite and synthetic iron sulphide. J Hazard Mater 137:626–632
Romero FM, Nunez L, Gutierrez ME, Armienta MA, Ceniceros-Gomez AE (2011) Evaluation of the potential of indigenous calcareous shale for neutralization and removal of arsenic and heavy metals from acid mine drainage in the Taxco mining area, Mexico. Arch Environ Con Tox 60:191–203
Sahinkaya E, Gunes FM, Ucar D, Kaksonen AH (2011) Sulfidogenic fluidized bed treatment of real acid mine drainage water. Bioresource Technol 102:683–689
Sahoo PK, Tripathy S, Panigrahi MK, Equeenuddin SM (2013) Inhibition of acid mine drainage from a pyrite-rich mining waste using industrial by-products: role of neo-formed phases. Water Air Soil Pollut 224:1757–1767
Sánchez EJ, López PE, Santofimia PE, Reyes AJ, Martín RJA (2006) The removal of dissolved metals by hydroxysulphate precipitates during oxidation and neutralization of acid mine waters, Iberian Pyrite Belt. Aquat Geochem 12:269–298
Shi YD, Chen TH, Wang YM, Wang J (2010) The evolution of crystal structure and arsenic content of pyrite calcined under N2 atmosphere. Acta Miner Sin S 1:231–231
Stookey LL (1970) Ferrozine—a new spectrophotometric reagent for iron. Analyt Chem 42:779–781
Tabak HH, Scharp R, Burckle J, Kawahara FK, Govind R (2003) Advances in biotreatment of acid mine drainage and biorecovery of metals: 1. Metal precipitation for recovery and recycle. Biodegradation 14:423–436
Thomas JE, Smart RSC, Skinner WM (2000) Kinetic factors for oxidative and non-oxidative dissolution of iron sulfides. Miner Eng 13:1149–1159
Utgikara V, Chena B-Y, Tabaka HH, Bishopa DF, Govindb R (2000) Treatment of acid mine drainage: I. Equilibrium biosorption of zinc and copper on non-viable activated sludge. Int Biodeter Biodegr 46:19–28
Wei FS, Qi WQ (2002) Monitoring and analysis method of water and waste water. China Environmental Science Press, China
Widler AM, Seward TM (2002) The adsorption of gold(I) hydrosulphide complexes by iron sulphide surfaces. Geochim Cosmochim Acta 66:383–402
Yang Y, Chen T, Li P, Liu H, Xie J, Xie Q, Zhan X (2014) Removal and recovery of Cu and Pb from single-metal and Cu–Pb–Cd–Zn multimetal solutions by modified pyrite: fixed-bed columns. Ind Eng Chem Res 53:18180–18188
Acknowledgments
This study was financially supported by the Natural Science Foundation of China (No. 41130206, 41402029, and 41472047). Y. Yang is grateful for funding provided by the China Scholarship Council (No. 201306690001) and European Cooperation in Science and Technology (COST Action Programs 1205 and 1302). The authors declare no competing financial interest.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yang, Y., Chen, T., Li, P. et al. Cu Removal from Acid Mine Drainage by Modified Pyrite: Batch and Column Experiments. Mine Water Environ 36, 371–378 (2017). https://doi.org/10.1007/s10230-016-0421-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10230-016-0421-5