Abstract
Background
The significance of immunosuppressants as an adjunct treatment with corticosteroids for IgA nephropathy (IgAN) has not been well demonstrated. This study was performed to compare two treatment regimens, steroid-pulse therapy or combined with mizoribine (MZR) in progressive IgAN.
Methods
Study design was a prospective randomized controlled trial of 40 patients with moderate to severe glomerular injuries who were randomly administered either pulse methylprednisolone followed by a 25-month course of oral prednisolone (P group, n = 20) or in combination with MZR (150 mg/day for 24 months, M + P group, n = 20). The primary endpoint was a reduction of proteinuria by ≥50 % of the baseline value. Secondary endpoints were increased serum creatinine (Cr) by ≥50 %, or a decrease in estimated glomerular filtration rate by ≤50 %.
Results
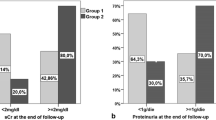
Twenty-five months after the initiation of treatment, urinary protein excretion significantly declined from the median of 0.98 to 0.17 g/gCr in the P group (P < 0.05) and from 1.01 to 0.38 g/gCr in the M + P group (P < 0.05). There was no statistical difference in the serial changes of proteinuria between two groups (P = 0.81). All patients reached the primary endpoint, and the cumulative incidence of the reduction of proteinuria was not significantly different (P = 0.76). No patient reached the secondary endpoint during the 25 months of treatment.
Conclusions
Both therapeutic regimens significantly reduced the levels of proteinuria. We could not find the additional effect of MZR in combination with steroid-pulses in this small-scale controlled trial. Steroid-pulse therapy with a 25-month course of oral steroids seems to be effective for progressive IgAN.



Similar content being viewed by others
References
D’Amico G. Natural history of idiopathic IgA nephropathy: role of clinical and histological prognostic factors. Am J Kidney Dis. 2000;36:227–37.
Goto M, Wakai K, Kawamura T, Ando M, Endoh M, Tomino Y. A scoring system to predict renal outcome in IgA nephropathy: a nationwide 10-year prospective cohort study. Nephrol Dial Transpl. 2009;24:3068–74.
Katafuchi R, Oh Y, Hori K, Komota T, Yanase T, Ikeda K, et al. An important role of glomerular segmental lesions on progression of IgA nephropathy: a multivariate analysis. Clin Nephrol. 1994;41:191–8.
Tanaka S, Ninomiya T, Katafuchi R, Masutani K, Tsuchimoto A, Noguchi H, et al. Development and validation of a prediction rule using the Oxford classification in IgA nephropathy. Clin J Am Soc Nephrol. 2013;8:2082–90.
Nagayama Y, Nishiwaki H, Hasegawa T, Komukai D, Kawashima E, Takayasu M, et al. Impact of the new risk stratification in the 2011 Japanese Society of Nephrology clinical guidelines for IgA nephropathy on incidence of early clinical remission with tonsillectomy plus steroid-pulse therapy. Clin Exp Nephrol. 2015;19:646–52.
Pozzi C, Andrulli S, Del Vecchio L, Melis P, Fogazzi GB, Altieri P, et al. Corticosteroid effectiveness in IgA nephropathy: long-term results of a randomized, controlled trial. J Am Soc Nephrol. 2004;5:157–63.
Katafuchi R, Ninomiya T, Mizumasa T, Ikeda K, Kumagai H, Nagata M, et al. The improvement of renal survival with steroid-pulse therapy in IgA nephropathy. Nephrol Dial Transplant. 2008;23:3915–20.
Balladie FW, Roberts IS. Controlled prospective trial of prednisolone and cytotoxics in progressive IgA nephropathy. J Am Soc Nephrol. 2002;13:142–8.
Harmankaya O, Oztürk Y, Baştürk T, Obek A, Kiliçarslan I. Efficacy of immunosuppressive therapy in IgA nephropathy presenting with isolated hematuria. Int Urol Nephrol. 2002;33:167–71.
Yoshikawa N, Honda M, Iijima K, Awazu M, Hattori S, Nakanishi K, et al. Steroid treatment for severe childhood IgA nephropathy: a randomized, controlled trial. Clin J Am Soc Nephrol. 2006;1:511–7.
Pozzi C, Andrulli S, Pani A, Scaini P, Del Vecchio L, Fogazzi G, et al. Addition of azathioprine to corticosteroids does not benefit patients with IgA nephropathy. J Am Soc Nephrol. 2010;21:1783–90.
Takahashi S, Wakui H, Gustafsson JA, Zilliacus J, Itoh H. Functional interaction of the immunosuppressant mizoribine with the 14-3-3 protein. Biochem Biophys Res Commun. 2000;274:87–92.
Aihara Y, Miyamae T, Ito S, Kobayashi S, Imagawa T, Mori M, et al. Mizoribine as an effective combined maintenance therapy with prednisolone in child-onset systemic lupus erythematosus. Pediatr Int. 2002;44:199–204.
Tanaka H, Tsugawa K, Suzuki K, Nakahata T, Ito E. Long-term mizoribine intermittent pulse therapy for young patients with flare of lupus nephritis. Pediatr Nephrol. 2006;21:962–6.
Yoshioka K, Ohashi Y, Sakai T, Ito H, Yoshikawa N, Nakamura H, et al. A multicenter trial of mizoribine compared with placebo in children with frequently relapsing nephritic syndrome. Kidney Int. 2000;58:317–24.
Xie Y, Huang S, Wang L, Miao L, Zhang A, Li Y, et al. Efficacy and safety of mizoribine combined with losartan in the treatment of IgA nephropathy: a multicenter, randomized, controlled study. Am J Med Sci. 2011;341:367–72.
Katafuchi R, Kiyoshi Y, Oh Y, Uesugi N, Ikeda K, Yanase T, et al. Glomerular score as a prognosticator in IgA nephropathy: its usefulness and limitation. Clin Nephrol. 1998;49:1–8.
Working Group of the International IgA Nephropathy Network, the Renal Pathology Society, Roberts IS, Cook HT, Troyanov S, Alpers CE, Amore A, Barratt J, et al. The Oxford classification of IgA nephropathy: pathology definitions, correlations, and reproducibility. Kidney Int. 2009;76:546–56.
Working Group of the International IgA Nephropathy Network, the Renal Pathology Society, Cattran DC, Coppo R, Cook HT, Feehally J, Roberts IS, Troyanov S, et al. The Oxford classification of IgA nephropathy: rationale, clinicopathological correlations, and classification. Kidney Int. 2009;76:534–45.
Hotta O, Furuta T, Chiba S, Yusa N, Taguma Y. Immunosuppressive effect of deoxyspergualin in proliferative glomerulonephritis. Am J Kidney Dis. 1999;34:894–901.
Matsuo S, Imai E, Horio M, Yasuda Y, Tomita K, Nitta K, et al. Collaborators developing the Japanese equation for estimated GFR: revised equations for estimated GFR from serum creatinine in Japan. Am J Kidney Dis. 2009;58:982–92.
Katafuchi R, Ikeda K, Mizumasa T, Tanaka H, Ando T, Yanase T, et al. Controlled, prospective trial of steroid treatment in IgA nephropathy: a limitation of low-dose prednisolone therapy. Am J Kidney Dis. 2003;41:972–83.
Yoshikawa N, Nakanishi K, Ishikura K, Hataya H, Iijima K, Honda M. Combination therapy with mizoribine for severe childhood IgA nephropathy: a pilot study. Pediatr Nephrol. 2008;23:757–63.
Frisch G, Lin J, Rosenstock J, Markowitz G, D’Agati V, Radhakrishnan J, et al. Mycophenolate mofetil (MMF) vs placebo in patients with moderately advanced IgA nephropathy: a double-blind randomized controlled trial. Nephrol Dial Transpl. 2005;20:2139–45.
Kim YC, Chin HJ, Koo HS, Kim S. Tacrolimus decreases albuminuria in patients with IgA nephropathy and normal blood pressure: a double-blind randomized controlled trial of efficacy of tacrolimus on IgA nephropathy. PLoS One. 2013;8:e71545.
Cruzado JM, Poveda R, Ibernón M, Díaz M, Fulladosa X, Carrera M, et al. Low-dose sirolimus combined with angiotensin-converting enzyme inhibitor and statin stabilizes renal function and reduces glomerular proliferation in poor prognosis IgA nephropathy. Nephrol Dial Transpl. 2011;26:3596–602.
Stangou M, Ekonomidou D, Giamalis P, Liakou H, Tsiantoulas A, Pantzaki A, et al. Steroids and azathioprine in the treatment of IgA nephropathy. Clin Exp Nephrol. 2011;15:373–80.
Pozzi C, Andrulli S, Pani A, Scaini P, Roccatello D, Fogazzi G, et al. IgA nephropathy with severe chronic renal failure: a randomized controlled trial of corticosteroids and azathioprine. J Nephrol. 2013;26:86–93.
Liu H, Xu X, Fang Y, Ji J, Zhang X, Yuan M, et al. Comparison of glucocorticoids alone and combined with cyclosporine A in patients with IgA nephropathy: a prospective randomized controlled trial. Intern Med. 2014;53:675–81.
Disease Kidney. Improving global outcome (KDIGO) Glomerulonephritis Work Group: KDIGO Clinical Practice Guideline for Glomerulonephritis. Kidney Int Suppl. 2012;2:209–17.
Sonda K, Takahashi K, Tanabe K, Funchinoue S, Hayasaka Y, Kawaguchi H, et al. Clinical pharmacokinetic study of mizoribine in renal transplant patients. Transpl Proc. 1996;28:3643–8.
Kawamura T, Yoshimura M, Miyazaki Y, Okamoto H, Kimura K, Hirano K, et al. A multicenter randomized controlled trial of tonsillectomy combined with steroid pulse therapy in patients with immunoglobulin A nephropathy. Nephrol Dial Transpl. 2014;29:1546–53.
Acknowledgments
We thank Drs. Kei Hori, Kazuhito Takeda, and Tetsuhiko Yoshida for their help designing the early version of this study. We also thank Drs. Takashi Inenaga, Akinori Nagashima, Masaru Nakayama, Hidetoshi Kanai, Hiroshi Tsuruta, Harumichi Higashi, Tadashi Hirano, Hiroshi Tanaka, and Atsumi Harada for treatment of the patients, and data collection. This study was funded by The Kidney Foundation, Japan. We are deeply grateful to Asahi Kasei Pharma for providing MZR, because the use of MZR for IgAN is not covered by health insurance in Japan.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of interest
Honoraria: Takanari Kitazono (Bayer Pharmaceutical Co., Bristol-Myers Squibb Co., Daiichi-Sankyo Co.), Kazuhiko Tsuruya (Chugai Pharmaceutical Co., Kyowa Hakko Kirin Co.)
Funding
This study was funded by The Kidney Foundation, Japan.
Research funding
Takanari Kitazono (Astellas Pharma Inc., Daiichi-Sankyo Co., Eisai Co., Kyowa Hakko Kirin Co., Mitsubishi Tanabe Pharma Co., MSD K.K., Ono Pharmaceutical Co., Otsuka Pharmaceutical Co., Sanofi-Aventis Pharmaceutical Co., Takeda Pharmaceutical Co.), Kazuhiko Tsuruya (Chugai Pharmaceutical Co., Kyowa Hakko Kirin Co., Otsuka Pharmaceutical Co., Takeda Pharmaceutical Co.)
Endowed department: Kazuhiko Tsuruya (Baxter).
Additional information
K. Masutani and A. Tsuchimoto equally contributed to this study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
About this article
Cite this article
Masutani, K., Tsuchimoto, A., Yamada, T. et al. Comparison of steroid-pulse therapy and combined with mizoribine in IgA nephropathy: a randomized controlled trial. Clin Exp Nephrol 20, 896–903 (2016). https://doi.org/10.1007/s10157-016-1226-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10157-016-1226-3