Abstract
Background
The caspase family of enzymes is grouped into two major sub-families, namely apoptotic and inflammatory caspases, which play central roles in the induction of apoptosis, regulation of inflammation and immunity, and cellular differentiation.
Methods
The role of caspase activation in tubular epithelium and interstitial cells of 3 lines of transgenic mice with obstructed nephropathy was examined: p35 mice bearing the pan-caspase inhibitor protein expressed by the p35 gene separated from the universal CAG promoter by a floxed STOP sequence were crossed with γGT.Cre and FSP1.Cre mice that express Cre recombinase in the cortical tubular epithelium and FSP1+ interstitial cells, respectively. The γGT.Cre;p35, FSP1.Cre;p35 and p35 control mice were then challenged with unilateral ureter obstruction (UUO).
Results
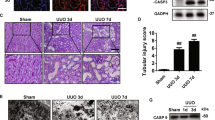
Proinflammatory parameters such as protein levels of active IL-1β subunit and mRNA levels of TNF-α and NOD-like receptor pyrin domain containing-3, and profibrogenic parameters such as interstitial matrix deposition and mRNA levels of fibronectin EIIIA isoform and α1 chain of procollagen type I in the kidneys were significantly increased at 7 days in the FSP1.Cre;p35- and p35-UUO mice, but not in the γGT.Cre;p35-UUO mice. These changes paralleled the numbers of apoptotic nuclei in tubules, but not in interstitial cells, and the protein levels of active caspase-3 subunit in the kidneys of FSP1.Cre;p35-, p35- and γGT.Cre;p35-UUO mice.
Conclusion
This study provides evidence of the critical role of caspase activation in the tubular epithelium, but not in FSP1+ interstitial cells, in apoptosis and inflammasome induction, leading to proinflammatory and profibrogenic processes in fibrous kidneys with UUO.




Similar content being viewed by others
References
Lavrik IN, Golks A, Krammer PH. Caspases: pharmacological manipulation of cell death. J Clin Invest. 2005;115:2665–72.
Sung JH, Zhao H, Roy M, Sapolsky RM, Steinberg GK. Viral caspase inhibitor p35, but not crmA, is neuroprotective in the ischemic penumbra following experimental stroke. Neuroscience. 2007;149:804–12.
Chandrashenkhar Y, Anway R, Shuros A, Anand I. Long-term caspase inhibition ameliorates apoptosis, reduces myocardial troponin-I cleavage, protects left ventricular function, and attenuates remodeling in rats with myocardial infarction. J Am Coll Cardiol. 2004;43:295–301.
Daemen MARC, van’t Veer C, Denecker G, Heemskerk VH, Wolfs TGAM, Clauss M, et al. Inhibition of apoptosis induced by ischemia-reperfusion prevents inflammation. J Clin Invest. 1999;104:541–9.
Sanz AB, Santamaria B, Ruiz-Ortega M, Egido J, Ortiz A. Mechanisms of renal apoptosis in health and disease. J Am Soc Nephrol. 2008;19:1634–42.
Bryant C, Fitzgerald KA. Molecular mechanisms involved in inflammasome activation. Trends Cell Biol. 2009;19:455–64.
Docherty NG, O’Sullivan OE, Healy DA, Fitzpatrick JM, Watson RWG. Evidence that inhibition of tubular cell apoptosis protects against renal damage and development of fibrosis following ureteric obstruction. Am J Physiol Renal Physiol. 2006;290:F4–13.
Tao Y, Kim J, Faubel S, Wu JC, Falk SA, Schrier RW, et al. Caspase inhibition reduces tubular apoptosis and proliferation and slows disease progression in polycystic kidney disease. PNAS. 2005;102:6954–9.
Seery JP, Cattell V, Watt FM. Cutting edge: amelioration of kidney disease in a transgenic mouse model of lupus nephritis by administration of the caspase inhibitor carbobenzoxy-valyl-ananyl-aspartyl-(beta-o-methyl)-fluoromethylketone. J Immunol. 2001;167:2452–5.
Yang B, Johnson TS, Haylor JL, Wagner B, Watson PF, El Kossi MMH, et al. Effects of caspase inhibition on the progression of experimental glomerulonephritis. Kidney Int. 2003;63:2050–64.
Inoue T, Suzuki H, Okada H. Targeted expression of a pan-caspase inhibitor in tubular epithelium attenuates interstitial inflammation and fibrogenesis in nephritic but not nephrotic mice. Kidney Int. 2012;82:980–9.
Iwano M, Plieth D, Danoff TM, Xue C, Okada H, Neilson EG. Evidence that fibroblasts derive from epithelium during tissue fibrosis. J Clin Invest. 2002;110:341–50.
Inoue T, Takenaka T, Hayashi M, Monkawa T, Yoshino J, Shimoda K, et al. Fibroblast expression of an IkB dominant-negative transgene attenuates renal fibrosis. J Am Soc Nephrol. 2010;21:2047–52.
Okada H, Inoue T, Kikuta T, Watanabe T, Kanno Y, Ban S, et al. A possible anti-inflammatory role of angiotensin II type 2 receptor in immune-mediated glomerulonephritis during type 1 receptor blockade. Am J Pathol. 2006;169:1577–89.
Strutz F, Okada H, Lo CW, Danoff TM, Carone RL, Tomaszewski JE, et al. Identification and characterization of a fibroblast marker: fSP1. J Cell Biol. 1995;130:393–405.
Okada H, Inoue T, Kikuta T, Kato N, Kanno Y, Hirosawa N, et al. Poly(ADP-ribose) polymerase-1 (PARP-1) and PARP-1 binding element enhance murine CCN2 gene transcription in renal tubular epithelial cells. J Am Soc Nephrol. 2008;19:933–42.
Savill J. Regulation of glomerular cell number by apoptosis. Kidney Int. 1999;56:1216–22.
Yang B, Johnson TS, Thomas GL, Watson PF, Wagner B, El Nahas AM. Apoptosis and caspase-3 in experimental anti-glomerular basement membrane nephritis. J Am Soc Nephrol. 2001;12:485–95.
Misseri R, Meldrum DR, Dinarello CA, Dagher P, Hile KL, Rink RC, et al. TNF-a mediates obstruction-induced renal tubular cell apoptosis and proapoptotic signaling. Am J Physiol Renal Physiol. 2005;288:F406–11.
Meldrum KK, Misseri R, Metcalfe P, Dinarello CA, Hille KL, Meldrum DR. TNF-a neutralization ameliorates obstruction-induced renal fibrosis and dysfunction. Am J Physiol Integr Comp Physiol. 2007;292:R1456–64.
Zarjou A, Yang S, Abraham E, Agarwal A, Liu G. Identification of a microRNA signature in renal fibrosis: role of miR-21. Am J Physiol Renal Physiol. 2011;301:F793–801.
Sanz AB, Justo P, Sanchez-Nino MD, Blanco-Colio LM, Winkles JA, Kreztler M, et al. The cytokine TWEAK modulates renal tubulointerstitial inflammation. J Am Soc Nephrol. 2008;19:695–703.
Jin Y, Ratnam K, Chuang PY, Fan Y, Zhong Y, Dai Y, et al. A systems approach identifies HIPK2 as a key regulator of kidney fibrosis. Nat Med. 2012;18:580–9.
Jones LK, O’Sullivan KM, Semple T, Kuligowski MP, Fukami K, Ma FY, et al. IL-1RI deficiency ameliorates early experimental renal interstitial fibrosis. Nephrol Dial Transpl. 2009;24:3024–32.
Vilaysane A, Chun J, Seamone ME, Wnag W, Chin R, Hirota S, et al. The NLRP3 inflammasome promotes renal inflammation and contributes to CKD. J Am Soc Nephrol. 2010;21:1732–44.
Chowdhury P, Sacks SH, Sheerin NS. Endogenous ligands for TLR2 and TLR4 are not involved in renal injury following ureteric obstruction. Nephrol Exp Nephrol. 2010;115:e122–30.
Campbell MT, Hile KL, Zhang H, Asanuma H, Vanderbrink BA, Rink RR, et al. Toll-like receptor 4: a novel signaling pathway during renal fibrogenesis. J Surg Res. 2011;168:e61–9.
Bani-Hani AH, Leslie JA, Asanuma H, Dinarello CA, Campbell MT, Meldrum DR, et al. IL-18 neutralization ameliorates obstruction-induced epithelial-mesenchymal transition and renal fibrosis. Kidney Int. 2009;76:500–11.
Watanabe A, Sohail MA, Gomes DA, Hashmi A, Nagata J, Sutterwala FS, et al. Inflammasome-mediated regulation of hepatic stellate cells. Am J Physiol Gastrointest Liver Physiol. 2009;296:G1248–57.
Artlett CM, Sassi-Gaha S, Rieger JL, Boesteanu AC, Feghali-Bostwick CA, Katsikis PD. The inflammasome activating caspase 1 mediates fibrosis and myofibroblast differentiation in systemic sclerosis. Arthritis Rheumatol. 2011;63:3563–74.
Shigeoka AA, Mueller JL, Kambo A, Mathison JC, King AJ, Hall WF, et al. An inflammasome-independent role for epithelial-expressed Nlrp3 in renal ischemia-reperfusion injury. J Immunol. 2010;185:6277–85.
Wang W, Wang X, Chun J, Vilaysane A, Clark S, French G, et al. Inflammasome-independent NLRP3 augments TGF-b signaling in kidney epithelium. J Immunol. 2013;190:1239–49.
Cailhier JF, Laplante P, Hebert MJ. Endothelial apoptosis and chronic transplant vasculopathy: recent results, novel mechanisms. Am J Transpl. 2006;6(2):247–53.
Laplante P, Sirois I, Raymond M, Kokta V, Beliveau A, Prat A, et al. Caspase-3-mediated secretion of connective tissue growth factor by apoptotic endothelial cells promotes fibrosis. Cell Death Differ. 2010;17:291–303.
Wang L, Scabilloni JF, Antonini JM, Rojanasakul Y, Castranova V, Mercer RR. Induction of secondary apoptosis, inflammation, and lung fibrosis after intratracheal instillation of apoptotic cells in rats. Am J Physiol Lung Cell Mol Physiol. 2006;290(4):L695–702.
Mathai SK, Gulati M, Peng X, Russell TR, Shaw AC, Rubinowtiz AN, et al. Circulating monocytes from systemic sclerosis patients with interstitial lung disease show an enhanced profibrotic phenotype. Lab Invest. 2010;90:812–23.
Canbay A, Taimr P, Torok N, Higuchi H, Friedman S, Gores GJ. Apoptotic body engulfment by a human stellate cell line is profibrogenic. Lab Invest. 2003;83:655–63.
Kodama T, Takehara T, Hikita H, Shimizu S, Shigekawa M, Tsunematsu H, et al. Increases in p53 expression induce CTGF synthesis by mouse and human hepatocytes and result in liver fibrosis in mice. J Clin Invest. 2011;121:3343–56.
Ichimura T, Asseldonk EJ, Humphreys BD, Gunaratnam L, Duffield JS, Bonventre JV. Kidney injury molecule-1 is a phosphatidylserine receptor that confers a phagocytic phenotype on epithelial cells. J Clin Invest. 2008;118:1657–68.
Inoue T, Plieth D, Venkov CD, Xu C, Neilson EG. Antibodies against macrophages that overlap in specificity with fibroblasts. Kidney Int. 2005;67:2488–93.
Oesterreicher CH, Penz-Oesterreicher M, Grivennikov SI, Guma M, Koltsova EK, Datz C, et al. Fibroblast-specific protein 1 identifies an inflammatory subpopulation of macrophages in the liver. PNAS. 2011;108:308–13.
Iwano M, Fischer A, Okada H, Pleith D, Xue C, Danoff TM, et al. Conditional abatement of tissue fibrosis using nucleotide analogs to corrupt DNA replication selectively in transgenic fibroblasts. Mol Ther. 2001;3:149–59.
Linkermann A, Hackl MJ, Kunzendorf U, Walczak H, Krautwald S, Jevnikar AM. Necroptosis in immunity and ischemia-reperfusion injury. Am J Transpl. 2013;13:2797–804.
Acknowledgments
We thank M. Fukuoka and T. Iso for their technical assistance. This research was supported in part by research grants from the Japanese Ministry of Education, Culture, Sports, Science and Technology, and Asubio Pharma Co. Ltd.
Conflict of interest
All of the authors declared no conflicts of interests.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Inoue, T., Kusano, T., Tomori, K. et al. Effects of cell-type-specific expression of a pan-caspase inhibitor on renal fibrogenesis. Clin Exp Nephrol 19, 350–358 (2015). https://doi.org/10.1007/s10157-014-1011-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10157-014-1011-0