Abstract
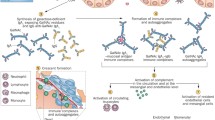
Henoch–Schönlein purpura (HSP) is a systemic disorder characterized by leukocytoclastic vasculitis involving the capillaries and the deposition of IgA immune complexes. Renal involvement is the principal cause of morbidity and mortality in children with HSP. Thus, it is important to clarify the onset mechanism of Henoch–Schönlein purpura nephritis (HSPN) and to identify the most appropriate treatment. We herein review the pathogenesis and treatment of HSPN. As to the pathogenesis, several studies suggest that galactose-deficient IgA1 is recognized by anti-glycan antibodies, leading to the formation of circulating immune complexes and their mesangial deposition, thereby inducing renal injury. Aggressive therapies for the treatment of severe HSPN, including multiple drug combination therapy and plasmapheresis, have been shown to be effective in ameliorating proteinuria and histological severity. Nevertheless, detailed investigations of the pathogenesis of HSPN and double-blind randomized control studies on children with HSPN are still necessary.



Similar content being viewed by others
References
Mills JA, Michel BA, Bloch DA, Calabrese LH, Hunder GG, Arend WP, et al. The American College of Rheumatology 1990 criteria for the classification of Henoch–Schonlein purpura. Arthritis Rheum. 1990;33(8):1114–21.
Saulsbury FT. Epidemiology of Henoch-Schonlein purpura. Cleve Clin J Med. 2002;69:187–9.
Bunchman TE, Mauer SM, Sibley RK, Vernier RL. Anaphylactoid purpura: characteristics of 16 patients who progressed to renal failure. Pediatr Nephrol. 1988;2:393–7.
Counahan R, Winterborn MH, White RH, Heaton JM, Meadow SR, Bluett NH, Swetschin H, Cameron JS, Chantler C. Prognosis of Henoch–Schönlein nephritis in children. Br Med J. 1977;2:11–4.
Yoshikawa N, Ito H, Yoshiya K, Nakahara C, Yoshiara S, Hasegawa O, Matsuyama S, Matsuo T. Henoch–Schoenlein nephritis and IgA nephropathy in children: a comparison of clinical course. Clin Nephrol. 1987;27:233–7.
Gardner-Medwin JM, Dolezalova P, Cummins C, Southwood TR. Incidence of Henoch–Schonlein purpura, Kawasaki disease, and rare vasculitides in children of different ethnic origins. Lancet. 2002;360:1197–202.
Stewart M, Savage JM, Bell B, McCord B. Long term renal prognosis of Henoch–Schönlein purpura in unselected childhood population. Eur J Pediatr. 1988;147:113–5.
Nielsen HE. Epidemiology of Schonlein–Henoch purpura. Acta Paediatr Scand. 1988;77:125–31.
Kawasaki Y, Suyama K, Yugeta E, Katayose M, Suzuki S, Sakuma H, et al. The incidence and severity of Henoch–Schönlein purpura nephritis over a 22-year period in Fukushima Prefecture, Japan. Int Urol Nephrol. 2010;42:1023–9.
Vogler C, Eliason SC, Wood EG. Glomerular membranopathy in children with IgA nephropathy and Henoch–Schoenlein purpura. Pediatr Dev Pathol. 1999;2:227–35.
Kerr MA. The structure and function of human IgA. Biochem J. 1990;271:285–96.
Yoo EM, Morrison SL. IgA: an immune glycoprotein. Clin Immunol. 2005;116:3–10.
Jones CL, Powell HR, Kincaid-Smith P, Roberton DM. Polymeric IgA and immune complex concentrations in IgA-related renal disease. Kidney Int. 1990;38:323–31.
Allen AC, Willis FR, Beattie TJ, Feehally J. Abnormal IgA glycosylation in Henoch–Schoenlein purpura restricted to patients with clinical nephritis. Nephrol Dial Transplant. 1998;13:930–4.
Egmond M, Damen CA, Spriel AB, Vidarsson G, Garderen E, Winkel JGJ. IgA and IgA Fc receptor. Trends Immunol. 2001;22:205–11.
Moja P, Quesnel A, Resseguier V, Lambert C, Freycon F, Berthoux F, et al. Is there IgA from gut mucosal origin in the serum of children with Henoch–Schoenlein purpura? Clin Immunol Immunopathol. 1998;86:290–7.
Silva FG. IgA nephropathy and Henoch–Schoenlein syndrome. In: Jennette JC, Olson JL, Schwartz MM, Silva FG, editors. Heptinstall’s pathology of the kidney. 15th ed. Philadelphia: Lippincott-Raven; 1998. p. 479–540.
Allen A, Harper S, Feehally J. Origin and structure of pathogenic IgA in IgA nephropathy. Biochem Soc Trans. 1997;25:486–90.
Coppo R, Basolo B, Bulzomi MR, Roccatello D, Carbonara AO, Barbiano di Belgiojoso G, et al. IgA1 and IgA2 immune complexes in primary IgA nephropathy and Henoch–Schoenlein purpura nephritis. Clin Exp Immunol. 1984;57:583–90.
Levinsky RJ, Barratt TM. IgA immune complexes in Henoch–Schoenlein purpura. Lancet. 1979;24:1100–3.
Leu KK, Wyatt RJ, Moldoveanu Z, Tomana M, Julian BA, Hogg RJ, et al. Serum levels of galactose-deficient IgA in children with IgA nephropathy and Henoch–Schoenlein purpura. Pediatr Nephrol. 2007;22:2067–72.
Oortwijn BD, Eijgenraam JW, Rastaldi MP, Roos A, Daha MR, van Kooten C. The role of secretary IgA and complement in IgA nephropathy. Semin Nephrol. 2008;28:58–65.
Endo M, Ohi H, Ohsawa I, Fujita T, Matsushita M. Complement activation through the lectin pathway in patients with Henoch–Schönlein purpura nephritis. Am J Kidney Dis. 2000;35:401–7.
Hisano S, Matsushita M, Fujita T, Iwasaki H. Activation of the lectin complement pathway in Henoch–Schönlein purpura nephritis. Am J Kidney Dis. 2005;45:295–302.
Moura IC, Benhamou M, Launay P, Vrtovsnik F, Blank U, Monteiro RC. The glomerular response to IgA deposition in IgA nephropathy. Semin Nephrol. 2008;28:88–95.
Kawasaki Y, Suzuki J, Nemoto K, Nozawa R, Suzuki S, Suzuki H. Clinical and pathological features of children with Henoch–Schoenlein purpura nephritis: risk factors associated with poor prognosis. Clin Nephrol. 2003;60:153–60.
Fujieda M, Oishi N, Naruse K, Hashizume M, Nishiya K, Kurashige T, Ito K. Soluble thrombomodulin and antibodies to bovine glomerular endothelial cells in patients with Henoch–Schoenlein purpura. Arch Dis Child. 1998;78:240–4.
Kawasaki Y, Suzuki J, Nozawa R, Sakai N, Tannji M, Isome M, et al. FB21, a monoclonal antibody that reacts with a sialic-acid-dependent carbohydrate epitope, is a marker for glomerular endothelial cell injury. Am J Kidney Dis. 2004;44:239–49.
Masuda M, Nakanishi K, Yoshizawa N, Iijima K, Yoshikawa N. Group A streptococcal antigen in the glomeruli of children with Henoch–Schönlein nephritis. Am J Kidney Dis. 2003;41:366–70.
Davin JC, Pierard G, Dechenne C, Grossman D, Nagy J, Quacoe M, et al. Possible pathogenic role of IgE in Henoch–Schönlein purpura. Pediatr Nephrol. 1994;8:169–71.
Ehara T, Shigematsu H. Mast cells in the kidney. Nephrology (Carlton). 2003;8:130–8.
Namgoong MK, Lim BK, Kim JS. Eosinophil cationic protein in Henoch–Schönlein purpura and in IgA nephropathy. Pediatr Nephrol. 1997;11:703–6.
Kawasaki Y, Hosoya M, Suzuki H. Possible pathologenic role of interleukin-5 and eosino cationic protein in Henoch–Schönlein purpura nephritis. Pediatr Int. 2005;47:51251–7.
Kawasaki Y, Imaizumi T, Matsuura H, Ohara S, Takano K, Suyama K, et al. Renal expression of alpha-smooth muscle actin and c-Met in children with Henoch–Schönlein purpura nephritis. Pediatr Nephrol. 2008;23:913–9.
Algoet C, Proesmans W. Renal biopsy 2–9 years after Henoch–Schönlein purpura. Pediatr Nephrol. 2003;18:471–3.
Goldstein AR, White RH, Akuse R, Chantler C. Long-term follow-up of childhood Henoch–Schoenlein nephritis. Lancet. 1992;339:280–2.
Meadow SR, Glasgow EF, White RH, Moncrieff MW, Cameron JS, Ogg CS. Schoenlein–Henoch nephritis. Q J Med. 1972;41:241–5.
Reif S, Jain A, Santiago J, Rossi T. Protein-losing enteropathy as a manifestation of Henoch–Schoenlein purpura. Acta Paediatr Scand. 1991;80:482–5.
Cull DL, Rosario V, Lally KP, Ratner I, Mahour GH. Surgical implications of Henoch–Schoenlein purpura. J Pediatr Surg. 1990;25:741–3.
Kawasaki Y, Suzuki H. Henoch–Schoenlein nephritis. In: Geary DF, Schaefer F, editors. Comprehensive pediatric nephrology. Mosby: Elsevier; 2008. p. 343–51.
Suthanthiran M, Strom TB. Immunoregulatory drugs: mechanistic basis for use in organ transplantation. Pediatr Nephrol. 1997;11:651–7.
Taube D, Brown Z, Williams DG. Long-term impairment of suppressor-cell function by cyclophosphamide in minimal-change nephropathy and its association with therapeutic response. Lancet. 1981;1:235–8.
Mizuno K, Tsujino M, Takada M, Hayashi M, Atsumi K. Studies on bredinin. I. Isolation, characterization and biological properties. J Antibiot. 1974;27:775–82.
Ichikawa Y, Ihara H, Takahara S. The immunosuppressive mode of action of mizoribine. Transplantation. 1984;38:262–7.
Ishikawa H. Mizoribine and mycophenolate mofetil. Curr Med Chem. 1999;6:575–97.
Kawasaki Y. Mizoribine: a new approach in the treatment of renal disease. Clin Dev Immunol. 2009;681482.
Diasio RB, Lo Buglio AF. Immunomodulators: immunosuppressive agents and immunostimulants. In: Hardman JG, editor. Goodman and Gilman’s: The pharmacological basis of therapeutics. 9th ed, vol. 52. New York: McGraw-Hill; 1996. p. 1291–308.
Niaudet P, Habib R. Methylprednisolone pulse therapy in the treatment of severe forms of Schoenlein–Henoch purpura nephritis. Pediatr Nephrol. 1998;12:238–43.
Mollica F, Li Volti S, Garozzo R, Russo G. Effectiveness of early prednisone treatment in preventing the development of nephropathy in anaphylactoid purpura. Eur J Pediatr. 1992;151:140–4.
Kawasaki Y, Suzuki J, Nozawa R, Suzuki S, Suzuki H. Efficacy of methylprednisolone and urokinase pulse therapy for severe Henoch–Schoenlein nephritis. Pediatrics. 2003;111:785–9.
Oner A, Tinaztepe K, Erdogan O. The effect of triple therapy on rapidly progressive type of Henoch–Schönlein nephritis. Pediatr Nephrol. 1995;9(1):6–10.
Bergstein J, Leiser J, Andreoli SP. Response of crescentic Henoch–Schoenlein purpura nephritis to corticosteroid and azathioprine therapy. Clin Nephrol. 1998;49:9–14.
Iijima K, Ito-Kariya S, Nakamura H, Yoshikawa N. Multiple combined therapy for severe Henoch–Schoenlein nephritis in children. Pediatr Nephrol. 1998;12:244–8.
Flynn JT, Smoyer WE, Bunchman TE, Kershaw DB, Sedman A. Treatment of Henoch–Schoenlein purpura glomerulonephritis in children with high-dose corticosteroids plus oral cyclophosphamide. Am J Nephrol. 2001;21:128–33.
Kawasaki Y, Suzuki J, Suzuki H. Efficacy of methylprednisolone and urokinase pulse therapy combined with or without cyclophosphamide in severe Henoch–Schoenlein nephritis: a clinical and histopathological study. Nephrol Dial Transplant. 2004;19:858–64.
Shin JI, Park JM, Shin YH, Kim JH, Lee JS, Jeong HJ. Henoch–Schönlein purpura nephritis with nephrotic-range proteinuria: histological regression possibly associated with cyclosporin A and steroid treatment. Scand J Rheumatol. 2005;34:392–5.
Kawasaki Y, Suyama K, Hashimoto K, Hosoya M. Methylprednisolone pulse plus mizoribine in children with Henoch–Schoenlein purpura nephritis. Clin Rheumatol. 2011;30:529–35.
Hattori M, Ito K, Konomoto T, Kawaguchi H, Yoshioka T, Khono M. Plasmapheresis as the sole therapy for rapidly progressive Schoenlein–Henoch purpura nephritis in children. Am J Kidney Dis. 1999;33:427–33.
Kawasaki Y, Suzuki J, Murai M, Takahashi A, Isome M, Nozawa R, et al. Plasmapheresis therapy for rapidly progressive Henoch–Schoenlein nephritis. Pediatr Nephrol. 2004;19:920–3.
Heldrich FJ, Minkin S, Gatdula CI. Intravenous immunoglobulin in Henoch–Schoenlein purpura: a case study. Md Med J. 1993;42:577–9.
Suguyama H, Watanabe N, Onoda T, Kikumoto Y, Yamamoto M, Maeta M, et al. Successful treatment of progressive Henoch–Schoenlein purpura nephritis with tonsillectomy and steroid pulse therapy. Intern Med. 2005;44:611–5.
Kawasaki Y, Suyama K, Matsumoto A, Takano K, Hashimoto K, Suzuki S, et al. Efficacy of tonsillectomy plus methylprednisolone pulse therapy for a child with Henoch–Schoenlein purpura nephritis. Tohoku J Exp Med. 2007;211:291–5.
Meulders Q, Pirson Y, Cosyns JP, Squifflet JP, de Strihou C. Course of Henoch–Schoenlein nephritis after renal transplantation: report on ten patients and review of the literature. Transplantation. 1994;58:1179–86.
Ramos EL. Recurrent diseases in the renal allograft. J Am Soc Nephrol. 1991;2:109–21.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Kawasaki, Y. The pathogenesis and treatment of pediatric Henoch–Schönlein purpura nephritis. Clin Exp Nephrol 15, 648–657 (2011). https://doi.org/10.1007/s10157-011-0478-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10157-011-0478-1