Abstract
Background
A gender difference in survival has been documented in colorectal cancer (CRC) patients, although the underlying mechanism remains undefined. This study aimed to gain improved insight into this difference, with a special focus on improved cancer-specific survival.
Methods
The study population consisted of 82,402 patients with invasive CRC who had undergone surgery in Japan between 1985 and 2004. To estimate improved survival, multivariate adjustment using patient demographics and tumor characteristics was performed.
Results
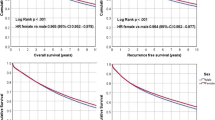
Patient characteristics changed over time. The 5-year survival rates increased from 66.5 to 76.3 % during the study period. Higher survival rates persisted in women over time (multivariate-adjustment model—hazard ratio [HR] 0.87, 95 % confidence interval [CI] 0.85–0.90). Patients who received surgery during the period 2000–2004 had significantly longer survival than those during the period 1985–1989 (men: HR 0.70, 95 % CI 0.67–0.74; women: HR 0.72, 95 % CI 0.67–0.76). However, there was no gender difference regarding improved survival.
Conclusions
A reduced risk of cancer-specific death for women relative to men persisted over time; however, enhancement of survival was equally observed in both genders. Identification of factors associated with gender differences and changes over time in CRC survival may serve as targets for further improvement.




Similar content being viewed by others
References
Ferlay J, Soerjomataram I, Ervik M et al (2013) GLOBOCAN 2012 v1.0, Cancer Incidence and Mortality Worldwide: IARC CancerBase No. 11 [Internet]. Lyon, France: International Agency for Research on Cancer. http://globocan.iarc.fr. Accessed on 27 Apr 2015
Gao RN, Neutel CI, Wai E (2008) Gender differences in colorectal cancer incidence, mortality, hospitalizations and surgical procedures in Canada. J Public Health (Oxf) 30(2):194–201
Cheng X, Chen VW, Steele B et al (2001) Subsite-specific incidence rate and stage of disease in colorectal cancer by race, gender, and age group in the United States, 1992–1997. Cancer 92(10):2547–2554
Allemani C, Weir HK, Carreira H, CONCORD Working Group et al (2015) Global surveillance of cancer survival 1995–2009: analysis of individual data for 25,676,887 patients from 279 population-based registries in 67 countries (CONCORD-2). Lancet 385(9972):977–1010
Zeng C, Wen W, Morgans A et al (2015) Disparities by race, age, and sex in the improvement of survival for major cancers: results from the National Cancer Institute Surveillance, Epidemiology, and End Results (SEER) Program in the United States, 1990 to 2010. JAMA Oncol 1(1):88–96
Matsuda T, Ajiki W, Marugame T et al, Research Group of Population-Based Cancer Registries of Japan: monitoring of Cancer Incidence in Japan—Survival 2003–2005 Report (Center for Cancer Control and Information Services, National Cancer Center, 2013) (2011) Population-based survival of cancer patients diagnosed between 1993 and 1999 in Japan: a chronological and international comparative study. Jpn J Clin Oncol 41:40–51
André T, Boni C, Mounedji-Boudiaf L et al (2004) Multicenter International Study of Oxaliplatin/5-Fluorouracil/Leucovorin in the Adjuvant Treatment of Colon Cancer (MOSAIC) Investigators: oxaliplatin, fluorouracil, and leucovorin as adjuvant treatment for colon cancer. N Engl J Med 350(23):2343–2351
Kopetz S, Chang GJ, Overman MJ et al (2009) Improved survival in metastatic colorectal cancer is associated with adoption of hepatic resection and improved chemotherapy. J Clin Oncol 27(22):3677–3683
Matsuda A, Matsuda T, Shibata A et al (2013) Cancer Incidence and Incidence Rates in Japan in 2008: a Study of 25 Population-based Cancer Registries for the Monitoring of Cancer Incidence in Japan (MCIJ) Project. Jpn J Clin Oncol 44(4):388–396
Ministry of Health, Labour and Welfare (2015) Vital Statistics Japan. http://www.mhlw.go.jp/english/database/db-hw/index.html. Accessed on 27 Apr 2015
Kotake K, Honjo S, Sugihara K et al (2003) Changes in colorectal cancer during a 20-year period: an extended report from the multi-institutional registry of large bowel cancer, Japan. Dis Colon Rectum 46(10 Suppl):S32–S43
Rhodes JB, Holmes FF, Clark GM (1997) Changing distribution of primary cancers in the large bowel. JAMA 238(15):1641–1643
Ji BT, Devesa SS, Chow WH et al (1998) Colorectal cancer incidence trends by subsite in urban Shanghai, 1972–1994. Cancer Epidemiol Biomark Prev 7(8):661–666
Wichmann MW, Müller C, Hornung HM et al (2001) Gender differences in long-term survival of patients with colorectal cancer. Br J Surg 88(8):1092–1098
McArdle CS, McMillan DC, Hole DJ (2003) Male gender adversely affects survival following surgery for colorectal cancer. Br J Surg 90(6):711–715
Paulson EC, Wirtalla C, Armstrong K et al (2009) Gender influences treatment and survival in colorectal cancer surgery. Dis Colon Rectum 52(12):1982–1991
Siegel R, DeSantis C (2014) Colorectal cancer statistics, 2014. CA Cancer J Clin 64(2):104–117
Verschueren RC, Mulder NH, Van Loon AJ et al (1997) The anatomical substrate for a differences in surgical approach to rectal cancer in male and female patients. Anticancer Res 17(1B):637–641
Fietkau R, Rödel C, Hohenberger W et al (2007) Rectal cancer delivery of radiotherapy in adequate time and with adequate dose is influenced by treatment center, treatment schedule, and gender and is prognostic parameter for local control: results of study CAO/ARO/AIO-94. Int J Radiat Oncol Biol Phys 67(4):1008–1019
Slattery ML, Friedman GD, Potter JD et al (1996) A description of age, sex, and site distributions of colon carcinoma in three geographic areas. Cancer 78(8):1666–1670
Hendifar A, Yang D, Lenz F et al (2009) Gender disparities in metastatic colorectal cancer survival. Clin Cancer Res 15(20):6391–6397
McMillan DC, Wotherspoon HA, Fearon KC et al (1995) A prospective study of tumor recurrence and the acute-phase response after apparently cur active colorectal cancer surgery. Am J Surg 170(4):319–322
Ward R, Meagher A, Tomlinson I et al (2001) Microsatellite instability and the clinicopathological features of sporadic colorectal cancer. Gut 48(6):821–829
Hawkins N, Norrie M, Cheong K et al (2002) CpG island methylation in sporadic colorectal cancers and its relationship to microsatellite instability. Gastroenterology 122(5):1376–1387
Ogino S, Cantor M, Kawasaki T et al (2006) CpG island methylator phenotype (CIMP) of colorectal cancer is best characterised by quantitative DNA methylation analysis and prospective cohort studies. Gut 55(7):1000–1006
Martling A, Granath F, Cedermark B et al (2009) Gender differences in the treatment of rectal cancer: a population based study. Eur J Surg Oncol 35(4):427–433
Rayson D, Urquhart R, Cox M et al (2012) Adherence to clinical practice guidelines for adjuvant chemotherapy for colorectal cancer in a Canadian province: a population-based analysis. J Oncol Pract 8(4):253–259
Jessup JM, Stewart A, Greene FL et al (2005) Adjuvant chemotherapy for stage III colon cancer: implications of race/ethnicity, age, and differentiation. JAMA 294(21):2703–2711
Verschueren RC, Mulder NH, Van Loon AJ et al (1997) The anatomical substrate for a differences in surgical approach to rectal cancer in male and female patients. Anticancer Res 17(1B):637–641
Acknowledgments
This work was supported in part by the National Cancer Center Research and Development Fund (26-A-32).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
About this article
Cite this article
Kotake, K., Asano, M., Ozawa, H. et al. Gender differences in colorectal cancer survival in Japan. Int J Clin Oncol 21, 194–203 (2016). https://doi.org/10.1007/s10147-015-0868-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10147-015-0868-6