Abstract
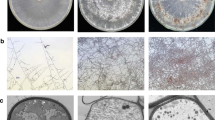
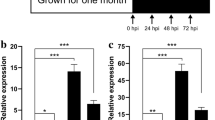
Sheath blight of rice, caused by Rhizoctonia solani Kühn AG-1 IA [teleomorph: Thanatephorus cucumeris (Frank) Donk], is one of the major diseases of rice (Oryza sativa L.) worldwide. Sclerotia produced by R. solani AG-1 IA are crucial for their survival in adverse environments and further dissemination when environmental conditions become conducive. Differentially expressed genes during three stages of sclerotial metamorphosis of R. solani AG-1 IA were investigated by utilizing complementary DNA amplified fragment length polymorphism (cDNA-AFLP) technique. A total of 258 transcript derived fragments (TDFs) were obtained and sequenced, among which 253 TDFs were annotated with known functions through BLASTX by searching the GenBank database and 19 annotated TDFs were assigned into 19 secondary metabolic pathways through searching the Kyoto Encyclopedia of Genes and Genomes (KEGG) PATHWAY database. Moreover, the results of quantitative real-time PCR (qRT-PCR) analysis showed that the expression patterns of eight representative annotated TDFs were positively correlated with sclerotial metamorphosis. Sequence annotation of TDFs showed homology similarities to several genes encoding for proteins belonging to the glycosyltransferases B (GTB) and RNA recognition motif (RRM) superfamily and to other development-related proteins. Taken together, it is concluded that the members of the GTB and RRM superfamilies and several new genes involved in proteolytic process identified in this study might serve as the scavengers of free radicals and reactive oxygen species (ROS) and thus play an important role in the sclerotial metamorphosis process of R. solani AG-1 IA.





Similar content being viewed by others
References
Aldaghi M, Bertaccini A, Lepoivre P (2012) cDNA-AFLP analysis of gene expression changes in apple trees induced by phytoplasma infection during compatible interaction. Eur J Plant Pathol 134:117–130. doi:10.1007/s10658-012-9970-z
Ambrosone A, Di Giacomo M, Leone A, Grillo MS, Costa A (2013) Identification of early induced genes upon water deficit in potato cell cultures by cDNA-AFLP. J Plant Res 126:169–178. doi:10.1007/s10265-012-0505-7
Arvas M et al (2006) Common features and interesting differences in transcriptional responses to secretion stress in the fungi Trichoderma reesei and Saccharomyces cerevisiae. BMC Genomics 7:32. doi:10.1186/1471-2164-7-32
Asakawa H, Kitamura K, Shimoda C (2001) A novel Cdc20-related WD-repeat protein, Fzr1, is required for spore formation in Schizosaccharomyces pombe. Mol Gen Genomics 265:424–435
Bachem CWB, Van Der Hoeven RS, De Bruijn SM, Vreugdenhil D, Zabeau M, Visser RGF (1996) Visualization of differential gene expression using a novel method of RNA fingerprinting based on AFLP: analysis of gene expression during potato tuber development. Plant J 9:745–753. doi:10.1046/j.1365-313X.1996.9050745.x
Bojovi Cveti D, Vuji IR (1988) Polysaccharide cytochemistry in maturing Aspergillus flavus sclerotia. Trans Br Mycol Soc 91:619–624
Botton A, Galla G, Conesa A, Bachem C, Ramina A, Barcaccia G (2008) Large-scale Gene Ontology analysis of plant transcriptome-derived sequences retrieved by AFLP technology. BMC Genomics 9:347. doi:10.1186/1471-2164-9-347
Chen CB, Harel A, Gorovoits R, Yarden O, Dickman MB (2004) MAPK regulation of sclerotial development in Sclerotinia sclerotiorum is linked with pH and cAMP sensing. Mol Plant-Microbe Interact 17:404–413. doi:10.1094/mpmi.2004.17.4.404
Coley-Smith JR, Cooke RC (1971) Survival and germination of fungal sclerotia. Annu Rev Phytopathol 9:65–92. doi:10.1146/annurev.py.09.090171.000433
Creste S, Neto AT, Figueira A (2001) Detection of single sequence repeat polymorphisms in denaturing polyacrylamide sequencing gels by silver staining. Plant Mol Biol Rep 19:299–306. doi:10.1007/BF02772828
Dilger M, Felsenstein FG, Schwarz G (2003) Identification and quantitative expression analysis of genes that are differentially expressed during conidial germination in Pyrenophora teres. Mol Gen Genomics 270:147–155. doi:10.1007/s00438-003-0910-7
Erental A, Harel A, Yarden O (2007) Type 2A phosphoprotein phosphatase is required for asexual development and pathogenesis of Sclerotinia sclerotiorum. Mol Plant-Microbe Interact 20:944–954. doi:10.1094/mpmi-20-8-0944
Erental A, Dickman MB, Yarden O (2008) Sclerotial development in Sclerotinia sclerotiorum: awakening molecular analysis of a “Dormant” structure. Fungal Biol Rev 22:6–16. doi:10.1016/j.fbr.2007.10.001
Georgiou CD, Cokic P, Carter K, Webster DA, Gennis RB (1988) Relationships between membrane-bound cytochrome o from Vitreoscilla and that of Escherichia coli. Biochim Biophys Acta 933:179–183. doi:10.1016/0005-2728(88)90068-0
Georgiou CD, Zervoudakis G, Petropoulou KP (2003) Ascorbic acid might play a role in the sclerotial differentiation of Sclerotium rolfsii. Mycologia 95:308–316. doi:10.2307/3762041
Georgiou CD, Patsoukis N, Papapostolou I, Zervoudakis G (2006) Sclerotial metamorphosis in filamentous fungi is induced by oxidative stress. Integr Comp Biol 46:691–712. doi:10.1093/icb/icj034
Harel A, Gorovits R, Yarden O (2005) Changes in protein kinase A activity accompany sclerotial development in Sclerotinia sclerotiorum. Phytopathology 95:397–404. doi:10.1094/phyto-95-0397
Hicks J, Lockington RA, Strauss J, Dieringer D, Kubicek CP, Kelly J, Keller N (2001) RcoA has pleiotropic effects on Aspergillus nidulans cellular development. Mol Microbiol 39:1482–1493. doi:10.1046/j.1365-2958.2001.02332.x
Hoeberichts FA et al (2008) A temperature-sensitive mutation in the Arabidopsis thaliana phosphomannomutase gene disrupts protein glycosylation and triggers cell death. J Biol Chem 283:5708–5718
Iordachescu M, Imai R (2008) Trehalose biosynthesis in response to abiotic stresses. J Integr Plant Biol 50:1223–1229. doi:10.1111/j.1744-7909.2008.00736.x
Jeney A, Beki E, Mule G, Hornok L (2004) Identification of growth stage specific transcript profiles in Fusarium proliferatum (Gibberella fujikuroi, mating population D) by cDNA-AFLP analysis. Eur J Plant Pathol 110:619–625. doi:10.1023/B:EJPP.0000032401.78288.d2
Jurick WM II, Rollins JA (2007) Deletion of the adenylate cyclase (sac1) gene affects multiple developmental pathways and pathogenicity in Sclerotinia sclerotiorum. Fungal Genet Biol 44:521–530. doi:10.1016/j.fgb.2006.11.005
Kimiharu I, Qingyuan G, Masao A (2004) Overwintering of rice sclerotial disease fungi, Rhizoctonia and Sclerotium spp. in paddy fields in Japan. Plant Pathol J 3:81–87
Kimura S et al (2003) Functional characterization of two flap endonuclease-1 homologues in rice. Gene 314:63–71. doi:10.1016/s0378-1119(03)00694-2
Lerouge P, Cabanes-Macheteau M, Rayon C, Fischette-Laine A-C, Gomord V, Faye L (1998) N-glycoprotein biosynthesis in plants: recent developments and future trends. Plant Mol Biol 38:31–48. doi:10.1023/a:1006012005654
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2 − ΔΔCT method. Methods 25:402–408. doi:10.1006/meth.2001.1262
Puttikamonkul S et al (2010) Trehalose 6-phosphate phosphatase is required for cell wall integrity and fungal virulence but not trehalose biosynthesis in the human fungal pathogen Aspergillus fumigatus. Mol Microbiol 77:891–911. doi:10.1111/j.1365-2958.2010.07254.x
Seifert GJ (2004) Nucleotide sugar interconversions and cell wall biosynthesis: how to bring the inside to the outside. Curr Opin Plant Biol 7:277–284. doi:10.1016/j.pbi.2004.03.004
Shi C, Chaudhary S, Yu K, Park SJ, Navabi A, McClean PE (2011) Identification of candidate genes associated with CBB resistance in common bean HR45 (Phaseolus vulgaris L.) using cDNA-AFLP. Mol Biol Rep 38:75–81. doi:10.1007/s11033-010-0079-1
Spiro RG (2002) Protein glycosylation: nature, distribution, enzymatic formation, and disease implications of glycopeptide bonds. Glycobiology 12:43R–56R. doi:10.1093/glycob/12.4.43R
Trembley ML, Ringli C, Honegger R (2002) Morphological and molecular analysis of early stages in the resynthesis of the lichen Baeomyces rufus. Mycol Res 106:768–776. doi:10.1017/s0953756202006081
Viaud MC, Balhadere PV, Talbot NJ (2002) A Magnaporthe grisea cyclophilin acts as a virulence determinant during plant infection. Plant Cell 14:917–930. doi:10.1105/tpc.010389
Vuylsteke M, Peleman JD, van Eijk MJT (2007) AFLP-based transcript profiling (cDNA-AFLP) for genome-wide expression analysis. Nat Protoc 2:1399–1413. doi:10.1038/nprot.2007.174
Wang P, Cardenas ME, Cox GM, Perfect JR, Heitman J (2001) Two cyclophilin A homologs with shared and distinct functions important for growth and virulence of Cryptococcus neoformans. EMBO Rep 2:511–518
Wang L, Liu LM, Wang ZG, Huang SW (2013) Genetic structure and aggressiveness of Rhizoctonia solani AG1-IA, the cause of sheath blight of rice in southern China. J Phytopathol 161:753–762. doi:10.1111/jph.12127
Wheeler GL, Jones MA, Smirnoff N (1998) The biosynthetic pathway of vitamin C in higher plants. Nature 393:365–369
Willetts HJ (1969) Structure of the outer surfaces of sclerotia of certain fungi. Arch Mikrobiol 69:48–53. doi:10.1007/bf00408562
Willetts HJ (1971) The survival of fungal sclerotia under adverse environmental conditions. Biol Rev 46:387–407. doi:10.1111/j.1469-185X.1971.tb01050.x
Yamashiro CT, Ebbole DJ, Lee B-U, Brown RE, Bourland C, Madi L, Yanofsky C (1996) Characterization of rco-1 of Neurospora crassa, a pleiotropic gene affecting growth and development that encodes a homolog of Tup1 of Saccharomyces cerevisiae. Mol Cell Biol 16:6218–6228
Yu G, Duan J, Yan H, Song B, He Z (2011) cDNA-AFLP analysis of gene expression differences between the flower bud and sprout-shoot apical meristem of Angelica sinensis (Oliv.) Diels. Genet Mol Biol 34:274–279
Yu Y, Jiang D, Xie J, Cheng J, Li G, Yi X, Fu Y (2012) Ss-Sl2, a novel cell wall protein with PAN modules, is essential for sclerotial development and cellular integrity of Sclerotinia sclerotiorum. PLoS One 7:e34962. doi:10.1371/journal.pone.0034962
Zheng A, et al (2013) The evolution and pathogenic mechanisms of the rice sheath blight pathogen. Nat Commun 4:1424. doi:10.1038/ncomms2427
Acknowledgments
This work was supported by a grant from the National Natural Science Foundation of China (Grant No. 31271994) awarded to Erxun Zhou.
Author information
Authors and Affiliations
Corresponding author
Additional information
Canwei Shu and Jieling Chen contributed equally to this work.
Rights and permissions
About this article
Cite this article
Shu, C., Chen, J., Sun, S. et al. Two distinct classes of protein related to GTB and RRM are critical in the sclerotial metamorphosis process of Rhizoctonia solani AG-1 IA. Funct Integr Genomics 15, 449–459 (2015). https://doi.org/10.1007/s10142-015-0435-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10142-015-0435-2