Abstract
Background
The present study evaluated the efficacy and safety of oxaliplatin, UFT, and leucovorin in metastatic gastric cancer.
Methods
Patients received intravenous oxaliplatin 130 mg/m2 on day 1, followed by oral UFT capsules (350 mg/m2 per day) and leucovorin tablets (90 mg/day), every 8 h, for 14 days, in a 3-week cycle.
Results
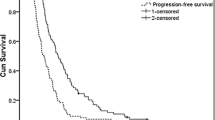
Twenty-three patients (61% with ≥2 metastatic sites), median age of 60 years (range, 39–69 years) were entered. Based on intention-to-treat analysis, one complete response and seven partial responses were found, resulting in an overall response rate (RR) of 35% (95% confidence interval [CI], 16–54), a median time to progression of 4 months (95% CI, 0.5–7.5), and a median overall survival (OS) of 8 months (95% CI, 4.5–11.5). The 1-year survival rate was 26%. Three patients did not complete the first course of 2 weeks; 1 died suddenly on day 16 with fatal lung embolism; 1 had rapid progressive disease and 1 experienced gastric hemorrhage on day 15 — both these patients withdrew. In the 20 patients assessable for toxicity no grade 4 toxicity occurred, grade 3 toxicity consisted of anemia in 1, diarrhea in 2, and neurotoxicity in 3 patients. No hand-foot syndrome (HFS) occurred.
Conclusion
Oxaliplatin is an effective drug in gastric cancer, but, as previously reported, its feasibility in combination with capecitabine is hampered due to combined hand-foot-based toxicity. The present phase II study of a combination of oxaliplatin with UFT and leucovorin appears to have efficacy and tolerability comparable to two other drug regimens used in gastric cancer, without the HFS problem.
Article PDF
Similar content being viewed by others

References
Wilke H, Preusser P, Fink U, Achterrath W, Mayer HJ, Stahl M, et al. New developments in the treatment of gastric carcinoma. Cancer Treat Res 1991;55:363–373.
Rivera F, Vega-Villegas ME, Lopez-Brea MF. Chemotherapy of advanced gastric cancer. Cancer Treat Rev 2007;33:315–324.
Glimelius B, Ekstrom K, Hoffman K, Graf W, Sjoden PO, Haglund U, et al. Randomized comparison between chemotherapy plus best supportive care with best supportive care in advanced gastric cancer. Ann Oncol 1997;8:163–168.
Murad AM, Santiago FF, Petroianu A, Rocha PR, Rodrigues MA, Rausch M. Modified therapy with 5-fluorouracil, doxorubicin, and methotrexate in advanced gastric cancer. Cancer 1993;72:37–41.
Pyrhonen S, Kuitunen T, Nyandoto P, Kouri M. Randomised comparison of fluorouracil, epidoxorubicin and methotrexate (FEMTX) plus supportive care with supportive care alone in patients with non-resectable gastric cancer. Br J Cancer 1995;71:587–591.
Wohrer SS, Raderer M, Hejna M. Palliative chemotherapy for advanced gastric cancer. Ann Oncol 2004;15:1585–1595.
Cunningham D, Starling N, Rao S, Iveson T, Nicolson M, Coxon F, et al. Capecitabine and oxaliplatin for advanced esophagogastric cancer. N Engl J Med 2008;358:36–46.
Ross P, Nicolson M, Cunningham D, Valle J, Seymour M, Harper P, et al. Prospective randomized trial comparing mitomycin, cisplatin, and protracted venous-infusion fluorouracil (PVI 5-FU) with epirubicin, cisplatin, and PVI 5-FU in advanced esophagogastric cancer. J Clin Oncol 2002;20:1996–2004.
Webb A, Cunningham D, Scarffe JH, Harper P, Norman A, Joffe JK, et al. Randomized trial comparing epirubicin, cisplatin, and fluorouracil versus fluorouracil, doxorubicin, and methotrexate in advanced esophagogastric cancer. J Clin Oncol 1997;15:261–267.
Wagner AD, Grothe W, Haerting J, Kleber G, Grothey A, Fleig WE. Chemotherapy in advanced gastric cancer: a systematic review and meta-analysis based on aggregate data. J Clin Oncol 2006;24:2903–2909.
Aykan NF, Idelevich E. The role of UFT in advanced gastric cancer. Ann Oncol 2008;19:1045–1452.
Pazdur R, Hoff PM, Medgyesy D, Royce M, Brito R. The oral fluorouracil prodrugs. Oncology (Williston Park) 1998;12:48–51.
Ravaud A, Borner M, Schellens JH, Geoffrois L, Schoffski BP, Kroon K, et al. UFT and leucovorin in first-line chemotherapy for patients with metastatic gastric cancer. An Early Clinical Studies Group (ECSG)/European Organization for Research Treatment of Cancer (EORTC) phase II trial. Eur J Cancer 2001;37:1642–1647.
Takiuchi H, Ajani JA. Uracil-tegafur in gastric carcinoma: a comprehensive review. J Clin Oncol 1998;16:2877–2885.
Kopec JA, Yothers G, Ganz PA, Land SR, Cecchini RS, Wieand HS, et al. Quality of life in operable colon cancer patients receiving oral compared with intravenous chemotherapy: results from National Surgical Adjuvant Breast and Bowel Project Trial C-06. J Clin Oncol 2007;25:424–430.
Kim YH, Cheong SK, Lee JD, Park JS, Shin SW, Kim JS. Phase II trial of oral UFT and leucovorin in advanced gastric carcinoma. Am J Clin Oncol 1996;19:212–216.
Carmichael J, Popiela T, Radstone D, Falk S, Borner M, Oza A, et al. Randomized comparative study of tegafur/uracil and oral leucovorin versus parenteral fluorouracil and leucovorin in patients with previously untreated metastatic colorectal cancer. J Clin Oncol 2002;20:3617–3627.
Douillard JY, Hoff PM, Skillings JR, Eisenberg P, Davidson N, Harper P, et al. Multicenter phase III study of uracil/tegafur and oral leucovorin versus fluorouracil and leucovorin in patients with previously untreated metastatic colorectal cancer. J Clin Oncol 2002;20:3605–3616.
Twelves C, Wong A, Nowacki MP, Abt M, Burris H 3rd, Carrato A, et al. Capecitabine as adjuvant treatment for stage III colon cancer. N Engl J Med 2005;352:2696–2704.
Lembersky BC, Wieand HS, Petrelli NJ, O’Connell MJ, Colangelo LH, Smith RE, et al. Oral uracil and tegafur plus leucovorin compared with intravenous fluorouracil and leucovorin in stage II and III carcinoma of the colon: results from National Surgical Adjuvant Breast and Bowel Project Protocol C-06. J Clin Oncol 2006;24:2059–2064.
Al-Batran SE, Atmaca A, Hegewisch-Becker S, Jaeger D, Hahnfeld S, Rummel MJ, et al. Phase II trial of biweekly infusional fluorouracil, folinic acid, and oxaliplatin in patients with advanced gastric cancer. J Clin Oncol 2004;22:658–663.
Cassidy J, Tabernero J, Twelves C, Brunet R, Butts C, Conroy T, et al. XELOX (capecitabine plus oxaliplatin): active first-line therapy for patients with metastatic colorectal cancer. J Clin Oncol 2004;22:2084–2091.
Park YH, Kim BS, Ryoo BY, Yang SH. A phase II study of capecitabine plus 3-weekly oxaliplatin as first-line therapy for patients with advanced gastric cancer. Br J Cancer 2006;94:959–963.
Woynarowski JM, Faivre S, Herzig MC, Arnett B, Chapman WG, Trevino AV, et al. Oxaliplatin-induced damage of cellular DNA. Mol Pharmacol 2000;58:920–927.
André T, Boni C, Mounedji-Boudiaf L, Navarro M, Tabernero J, Hickish T, et al. Multicenter International Study of Oxaliplatin/5-Fluorouracil/Leucovorin in the Adjuvant Treatment of Colon Cancer (MOSAIC) Investigators. Oxaliplatin, fluorouracil, and leucovorin as adjuvant treatment for colon cancer. N Engl J Med 2004;350:2343–2351.
Borner MM, Dietrich D, Stupp R, Morant R, Honegger H, Wernli M, et al. Phase II study of capecitabine and oxaliplatin in first- and second-line treatment of advanced or metastatic colorectal cancer. J Clin Oncol. 2002;20:1759–1766.
Maindrault-Goebel F, de Gramont A, Louvet C, André T, Carola E, Mabro M, et al. High-dose intensity oxaliplatin added to the simplified bimonthly leucovorin and 5-fluorouracil regimen as second-line therapy for metastatic colorectal cancer (FOLFOX 7). Eur J Cancer 2001;37:1000–1005.
Gehan EA. The determination of the number of patients required in a preliminary and a follow-up trial of a new chemotherapeutic agent. J Chronic Dis 1961;13:346–353.
De VF, Orditura M, Matano E, Bianco R, Carlomagno C, Infusino S, et al. A phase II study of biweekly oxaliplatin plus infusional 5-fluorouracil and folinic acid (FOLFOX-4) as first-line treatment of advanced gastric cancer patients. Br J Cancer 2005;92:1644–1649.
Jatoi A, Murphy BR, Foster NR, Nikcevich DA, Alberts SR, Knost JA, et al. Oxaliplatin and capecitabine in patients with metastatic adenocarcinoma of the esophagus, gastroesophageal junction and gastric cardia: a phase II study from the North Central Cancer Treatment Group. Ann Oncol 2006;17:29–34.
Lordick F, Lorenzen S, Stollfuss J, Vehling-Kaiser U, Kullmann F, Hentrich M, et al. Phase II study of weekly oxaliplatin plus infusional fluorouracil and folinic acid (FUFOX regimen) as first-line treatment in metastatic gastric cancer. Br J Cancer 2005;93:190–194.
Park YH, Lee JL, Ryoo BY, Ryu MH, Yang SH, Kim BS, et al. Capecitabine in combination with oxaliplatin (XELOX) as a first-line therapy for advanced gastric cancer. Cancer Chemother Pharmacol 2008;61:623–629.
Al-Batran SE, Hartmann JT, Probst S, Schmalenberg H, Hollerbach S, Hofheinz R, et al. Phase III trial in metastatic gastroesophageal adenocarcinoma with fluorouracil, leucovorin plus either oxaliplatin or cisplatin: a study of the Arbeitsgemeinschaft Internistische Onkologie. J Clin Oncol 2008;26:1435–1442.
Feliu J, Gonzalez BM, Garcia-Giron C, Espinosa E, Garcia-Alfonso P, Belon J, et al. Treatment of patients with advanced gastric carcinoma with the combination of etoposide plus oral tegafur modulated by uracil and leucovorin. A phase II study of the ONCOPAZ Cooperative Group. Cancer 1996;78:211–216.
Suga S, Iwase H, Shimada M, Nishio Y, Ichihara T, Ichihara S, et al. Neoadjuvant chemotherapy in scirrhous cancer of the stomach using uracil and tegafur and cisplatin. Intern Med 1996;5:930–936.
Chao Y, Li CP, Chao TY, Su WC, Hsieh RK, Wu MF, et al. An open, multi-centre, phase II clinical trial to evaluate the efficacy and safety of paclitaxel, UFT, and leucovorin in patients with advanced gastric cancer. Br J Cancer 2006;95:159–163.
Okines AF, Norman AR, McCloud P, Kang YK, Cunningham D. Meta-analysis of the REAL-2 and ML17032 trials: evaluating capecitabine-based combination chemotherapy and infused 5-fluorouracil-based combination chemotherapy for the treatment of advanced oesophago-gastric cancer. Ann Oncol 2009;20:1529–1534.
Pazdur R, Lassere Y, Rhodes V, Ajani JA, Sugarman SM, Patt YZ, et al. Phase II trial of uracil and tegafur plus oral leucovorin: an effective oral regimen in the treatment of metastatic colorectal carcinoma. J Clin Oncol 1994;12:2296–2300.
Eng C. Toxic effects and their management: daily clinical challenges in the treatment of colorectal cancer. Nat Rev Clin Oncol 2009;6:207–218.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Siemerink, E.J.M., Drenth, A.F.J., Mulder, N.H. et al. Phase II study of oxaliplatin, UFT, and leucovorin in patients with metastatic gastric cancer. Gastric Cancer 13, 95–100 (2010). https://doi.org/10.1007/s10120-010-0545-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10120-010-0545-4