Abstract
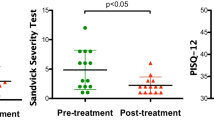
Vaginal atrophy occurring during menopause is closely related to the dramatic decrease in ovarian estrogens due to the loss of follicular activity. Particularly, significant changes occur in the structure of the vaginal mucosa, with consequent impairment of many physiological functions. In this study, carried out on bioptic vaginal mucosa samples from postmenopausal, nonestrogenized women, we present microscopic and ultrastructural modifications of vaginal mucosa following fractional carbon dioxide (CO2) laser treatment. We observed the restoration of the vaginal thick squamous stratified epithelium with a significant storage of glycogen in the epithelial cells and a high degree of glycogen-rich shedding cells at the epithelial surface. Moreover, in the connective tissue constituting the lamina propria, active fibroblasts synthesized new components of the extracellular matrix including collagen and ground substance (extrafibrillar matrix) molecules. Differently from atrophic mucosa, newly-formed papillae of connective tissue indented in the epithelium and typical blood capillaries penetrating inside the papillae, were also observed. Our morphological findings support the effectiveness of fractional CO2 laser application for the restoration of vaginal mucosa structure and related physiological trophism. These findings clearly coupled with striking clinical relief from symptoms suffered by the patients before treatment.




Similar content being viewed by others
References
Brincat M, Moniz CJ, Studd JW, Darby A, Magos A, Emburey G, Versi E (1985) Long-term effects of the menopause and sex hormones on skin thickness. Br J Obstet Gynaecol 92(3):256–259
Mac Bride MB, Rhodes DJ, Shuster LT (2010) Vulvovaginal atrophy. Mayo Clin Proc 85(1):87–94. doi:10.4065/mcp.2009.0413
Leiblum S, Bachmann G, Kemmann E, Colburn D, Swartzman L (1983) Vaginal atrophy in the postmenopausal woman. The importance of sexual activity and hormones. JAMA 249(16):2195–2198
WHO (2013) European Health Report 2012. Charting the way to well-being
Berlin AL, Hussain M, Phelps R, Goldberg DJ (2009) A prospective study of fractional scanned nonsequential carbon dioxide laser resurfacing: a clinical and histopathologic evaluation. Dermatol Surg 35(2):222–228. doi:10.1111/j.1524-4725.2008.34413.x
Lee CJ, Park JH, Ciesielski TE, Thomson JG, Persing JA (2008) Retinoids, 585-nm laser, and carbon dioxide laser: a numeric comparison of neocollagen formation in photoaged hairless mouse skin. Aesth Plast Surg 32(6):894–901. doi:10.1007/s00266-008-9121-2
Salvatore S, Nappi RE, Zerbinati N, Calligaro A, Ferrero S, Candiani M, Maggiore ULR (2014) A 12-week treatment with fractional CO2 laser for vulvovaginal atrophy: a pilot study. Climacteric. 17(4):363–369. doi:10.3109/13697137.2014.899347
Alberts B, Johnson A, Lewis Jea (2002) Transport from the ER through the Golgi apparatus. In: Molecular biology of the bell, 4th edition. Garland Science, New York
Stephens DJ (2012) Cell biology: collagen secretion explained. Nature 482(7386):474–475. doi:10.1038/482474a
Tierney EP, Kouba DJ, Hanke CW (2009) Review of fractional photothermolysis: treatment indications and efficacy. Dermatol Surg 35(10):1445–1461. doi:10.1111/j.1524-4725.2009.01258.x
Prignano F, Campolmi P, Bonan P, Ricceri F, Cannarozzo G, Troiano M, Lotti T (2009) Fractional CO2 laser: a novel therapeutic device upon photobiomodulation of tissue remodeling and cytokine pathway of tissue repair. Dermatol Ther 22(Suppl 1):S8–15. doi:10.1111/j.1529-8019.2009.01265.x
Nowak KC, McCormack M, Koch RJ (2000) The effect of superpulsed carbon dioxide laser energy on keloid and normal dermal fibroblast secretion of growth factors: a serum-free study. Plast Reconstr Surg 105(6):2039–2048
Manolis EN, Kaklamanos IG, Spanakis N et al (2007) Tissue concentration of transforming growth factor b1 and basic fibroblast growth factor in skin wounds created with a CO2 laser and scalpel: a comparative experimental study, using an animal model of skin resurfacing. Wound Repair Regen 15:252–257. doi:10.1111/j.1524-475X.2007.00212.x
Orringer JS, Kang S, Johnson TJ, Karimipour DJ, Hamilton T, Hammerberg C, Voorhees JJ, Fisher GJ (2004) Connective tissue remodeling induced by carbon dioxide laser resurfacing of photodamaged human skin. Arch Dermatol 140(11):1326–1332. doi:10.1001/archderm.140.11.1326
Hantash BM, Bedi VP, Kapadia B, Rahman Z, Jiang K, Tanner H, Chan KF, Zachary CB (2007) In vivo histological evaluation of a novel ablative fractional resurfacing device. Lasers Surg Med 39:96–107. doi:10.1002/lsm.20468
Ishida Y, Nagata K (2011) Hsp47 as a collagen-specific molecular chaperone. Methods Enzymol 499:167–182. doi:10.1016/B978-0-12-386471-0.00009-2
Tasab M, Batten MR, Bulleid NJ (2000) Hsp47: a molecular chaperone that interacts with and stabilizes correctly-folded procollagen. EMBO J 19(10):2204–2211. doi:10.1093/emboj/19.10.2204
Dafforn TR, Della M, Miller AD (2001) The molecular interactions of heat shock protein 47 (Hsp47) and their implications for collagen biosynthesis. J Biol Chem 276(52):49310–49319. doi:10.1074/jbc.M108896200
North American Menopause Society (2007) The role of local vaginal estrogen for treatment of vaginal atrophy in postmenopausal women: 2007 position statement of The North American Menopause Society. Menopause 14:355–369. doi:10.1097/gme.0b013e31805170eb
Farage MA, Maibach HI (2011) Morphology and physiological changes of genital skin and mucosa. Curr Probl Dermatol 40:9–19. doi:10.1159/000321042
Guerre-Millo M (2002) Adipose tissue hormones. J Endocrinol Invest 25(10):855–861
Acknowledgments
NZ, MS, MO, MC, TI, SS, FM and AC contributed equally to this work. Many thanks to Aurora Farina for her skillful technical contribution. This article was not supported by grants.
Conflict of interest
NZ, MS, MO, MC, TI, SS, FM and AC certify that there is no conflict of interest with any financial organization regarding the material discussed in the manuscript.
Statement of authorship
NZ, MS, MO, MC, TI, SS, FM and AC hereby certify that all work contained in this article is original. The authors claim full responsibility for the content of this article.
Informed consent
Written informed consent was obtained from the patients participating in this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zerbinati, N., Serati, M., Origoni, M. et al. Microscopic and ultrastructural modifications of postmenopausal atrophic vaginal mucosa after fractional carbon dioxide laser treatment. Lasers Med Sci 30, 429–436 (2015). https://doi.org/10.1007/s10103-014-1677-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10103-014-1677-2