Abstract
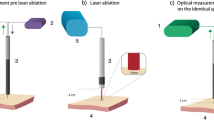
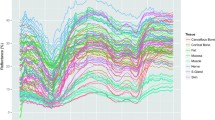
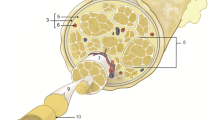
Facial nerve function may be hampered by iatrogenic damage during head and neck laser surgery procedures. Optical techniques can serve as a basis for feedback-controlled tissue-specific laser surgery on the jaw bone and the parotid gland. In order to preserve nerve tissue during laser surgery, the alteration of optical tissue properties through laser-tissue interactions have to be taken into account. It was the aim of this study to evaluate the viability of optical tissue differentiation through diffuse reflectance spectroscopy after exposure to laser light as a basis for a feedback system for tissue-specific laser surgery. Spectra of diffuse reflectance (wavelength, 350–650 nm) of nerves, salivary glands, and cortical and cancellous bone of the midfacial region (ex vivo domestic pig heads) were acquired before/after Er:YAG laser (wavelength, 2.94 μm) ablation (each 16,800 spectra). Principal component analysis was computed followed by quadratic discriminant analysis. The tissue classification performance as well as area under the curve (AUC) sensitivity and specificity for tissue differentiation was assessed before and after laser-tissue exposure. A high classification performance was observed before laser ablation (total error, 7.74 %). Nerve tissue was differentiated from bone and salivary glands with results greater than 0.96 in AUC, sensitivity and specificity. After laser exposure, a total classification error of 18.61 % was observed. The differentiation of nerve tissue was reduced with an AUC of >0.94, sensitivity of >0.95, and specificity >0.87. Er:YAG laser ablation only slightly reduces the differentiation performance through diffuse reflectance in the investigated tissue types. The results show the general viability of diffuse reflectance spectroscopy in identifying neural structures in the vicinity of salivary glands and bone as a basis for nerve preservation during feedback-controlled laser surgery.





Similar content being viewed by others
References
Minton JP (1986) The laser in surgery. A 23 year perspective. Am J Surg 151:725–729
Kuttenberger JJ, Stubinger S, Waibel A, Werner M, Klasing M, Ivanenko M, Hering P, Von Rechenberg B, Sader R, Zeilhofer HF (2008) Computer-guided CO2-laser osteotomy of the sheep tibia: technical prerequisites and first results. Photomed Laser Surg 26:129–136
Stopp S, Svejdar D, von Kienlin E, Deppe H, Lueth TC (2008) A new approach for creating defined geometries by navigated laser ablation based on volumetric 3-D data. IEEE Trans Biomed Eng 55:1872–1880
Spinelli P, Calarco G, Mancini A, Ni XG (2006) Operative colonoscopy in cancer patients. Minim Invasive Ther Allied Technol 15:339–347
Colella G, Cannavale R, Vicidomini A, Lanza A (2007) Neurosensory disturbance of the inferior alveolar nerve after bilateral sagittal split osteotomy: a systematic review. J Oral Maxillofac Surg 65:1707–1715
Yoshida T, Nagamine T, Kobayashi T, Michimi N, Nakajima T, Sasakura H, Hanada K (1989) Impairment of the inferior alveolar nerve after sagittal split osteotomy. J Craniomaxillofac Surg 17:271–277
Marchesi M, Biffoni M, Trinchi S, Turriziani V, Campana FP (2006) Facial nerve function after parotidectomy for neoplasms with deep localization. Surg Today 36:308–311
Baxter GD, Walsh DM, Allen JM, Lowe AS, Bell AJ (1994) Effects of low intensity infrared laser irradiation upon conduction in the human median nerve in vivo. Exp Physiol 79:227–234
Menovsky T, van den Bergh WM, Beek JF (1996) Effect of CO2 milliwatt laser on peripheral nerves: part I. A dose-response study. Microsurgery 17:562–567
Menovsky T, Van Den Bergh Weerman M, Beek JF (2000) Effect of CO(2)-milliwatt laser on peripheral nerves: part II. A histological and functional study. Microsurgery 20:150–155
Taroni P, Pifferi A, Torricelli A, Comelli D, Cubeddu R (2003) In vivo absorption and scattering spectroscopy of biological tissues. Photochem Photobiol Sci 2:124–129
Marchesini R, Pignoli E, Tomatis S, Fumagalli S, Sichirollo AE, Di Palma S, Dal Fante M, Spinelli P, Croce AC, Bottiroli G (1994) Ex vivo optical properties of human colon tissue. Lasers Surg Med 15:351–357
Bashkatov AN, Genina EA, Kochubey VI, Tuchin VV (2005) Optical properties of human skin, subcutaneous and mucous tissues in the wavelength range from 400 to 2000 nm. J Phys D Appl Phys 38:2543
Ebert DW, Roberts C, Farrar SK, Johnston WM, Litsky AS, Bertone AL (1998) Articular cartilage optical properties in the spectral range 300–850 nm. J Biomed Opt 3:326–333
Stelzle F, Tangermann-Gerk K, Adler W, Zam A, Schmidt M, Douplik A, Nkenke E (2010) Diffuse reflectance spectroscopy for optical soft tissue differentiation as remote feedback control for tissue-specific laser surgery. Lasers Surg Med 42:319–325
Stelzle F, Zam A, Adler W, Tangermann-Gerk K, Douplik A, Nkenke E, Schmidt M (2011) Optical nerve detection by diffuse reflectance spectroscopy for feedback controlled oral and maxillofacial laser surgery. J Transl Med 9:20
Stelzle F, Adler W, Zam A, Tangermann-Gerk K, Knipfer C, Douplik A, Schmidt M, Nkenke E (2012) In vivo optical tissue differentiation by diffuse reflectance spectroscopy: preliminary results for tissue-specific laser surgery. Surg Innov 19:385–393
Schomacker KT, Walsh JT, Flotte TJ, Deutsch TF (1990) Thermal damage produced by high-lrradiance continuous wave CO2 laser cutting of tissue. Lasers Surg Med 10:74–84
Ritz JP, Roggan A, Germer CT, Isbert C, Muller G, Buhr HJ (2001) Continuous changes in the optical properties of liver tissue during laser-induced interstitial thermotherapy. Lasers Surg Med 28:307–312
Ross EV, McKinlay JR, Sajben FP, Miller CH, Barnette DJ, Meehan KJ, Chhieng NP, Deavers MJ, Zelickson BD (2002) Use of a novel erbium laser in a Yucatan minipig: a study of residual thermal damage, ablation, and wound healing as a function of pulse duration. Lasers Surg Med 30:93–100
Lukianova-Hleb EY, Oginsky AO, Olson JS, Lapotko DO (2011) Short laser pulse-induced irreversible photothermal effects in red blood cells. Lasers Surg Med 43:249–260
Choi JY, Tanenbaum BS, Milner TE, Dao XV, Nelson JS, Sobol EN, Wong BJ (2001) Theramal, mechanical, optical, and morphologic changes in bovine nucleus pulposus induced by Nd:YAG (lambda = 1.32 microm) laser irradiation. Lasers Surg Med 28:248–254
Ivanenko MM, Fahimi-Weber S, Mitra T, Wierich W, Hering P (2002) Bone tissue ablation with sub-microS pulses of a Q-switch CO(2) laser: histological examination of thermal side effects. Lasers Med Sci 17:258–264
Jiao J, Guo Z (2009) Thermal interaction of short-pulsed laser focused beams with skin tissues. Phys Med Biol 54:4225–4241
Stelzle F, Terwey I, Knipfer C, Adler W, Tangermann-Gerk K, Nkenke E, Schmidt M (2012) The impact of laser ablation on optical soft tissue differentiation for tissue specific laser surgery-an experimental ex vivo study. J Transl Med 10:123
Development Core Team R (2008) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, ISBN 3-900051-07-0
Andrea Peters, Hothorn T (2000) ipred: improved predictors. R package version 0.8-8. http://CRAN.R-project.org/package=ipred. Accessed 18 November 2013
Sergej Potapov, Werner Adler, Lausen B (2009) Daim: diagnostic accuracy of classification models. R package version 1.1.0. http://CRAN.R-project.org/package=Daim. Accessed 18 November 2013
Palmer GM, Marshek CL, Vrotsos KM, Ramanujam N (2002) Optimal methods for fluorescence and diffuse reflectance measurements of tissue biopsy samples. Lasers Surg Med 30:191–200
Boppart SA, Herrmann J, Pitris C, Stamper DL, Brezinski ME, Fujimoto JG (1999) High-resolution optical coherence tomography-guided laser ablation of surgical tissue. J Surg Res 82:275–284
Niemz MH (2007) Laser-tissue interactions: fundamentals and applications. Springer, Berlin Heidelberg
Kim BM, Feit MD, Rubenchik AM, Mammini BM, Da Silva LB (1998) Optical feedback signal for ultrashort laser pulse ablation of tissue. Appl Surf Sci 127–129:857–862
Robertson CW, Williams D (1971) Lambert absorption coefficients of water in the infrared. J Opt Soc Am 61:1316–1320
Hale GM, Querry MR (1973) Optical constants of water in the 200-nm to 200-μm wavelength region. Appl Opt 12:555–563
Romanos G, Ko HH, Froum S, Tarnow D (2009) The use of CO(2) laser in the treatment of peri-implantitis. Photomed Laser Surg 3:381–386
Luerssen K, Lubatschowski H, Ptok M (2007) Erbium:YAG laser surgery on vocal fold tissue. HNO 55:443–446
Ruderman S, Gomes AJ, Stoyneva V, Rogers JD, Fought AJ, Jovanovic BD, Backman V (2010) Analysis of pressure, angle and temporal effects on tissue optical properties from polarization-gated spectroscopic probe measurements. Biomed Opt Expr 1:489–499
Zhengmao Y, Auner G (2004) Principal component analysis approach for biomedical sample identification. IEEE International Conference on Systems, Man and Cybernetics 10-13 Oct 2:1348-1353
Salomatina E, Yaroslavsky AN (2008) Evaluation of the in vivo and ex vivo optical properties in a mouse ear model. Phys Med Biol 53:2797–2807
Wilson BC, Jeeves WP, Lowe DM (1985) In vivo and post mortem measurements of the attenuation spectra of light in mammalian tissues. Photochem Photobiol 42:153–162
Acknowledgments
The authors gratefully acknowledge the funding by the ELAN-Funds, University of Erlangen-Nuremberg (AZ:07.03.06.1) and the Erlangen Graduate School in Advanced Optical Technologies (SAOT) by the German National Science Foundation (DFG) (AZ: STE 1877/2-1) as part of the Excellence Initiative.
Conflict of interests
The authors declare that there is no conflict of interest.
Ethics approval
Ethics approval is not necessary. The experimental study was carried out on tissues that were provided by a slaughterhouse.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Stelzle, F., Knipfer, C., Bergauer, B. et al. Optical nerve identification in head and neck surgery after Er:YAG laser ablation. Lasers Med Sci 29, 1641–1648 (2014). https://doi.org/10.1007/s10103-014-1569-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10103-014-1569-5