Abstract
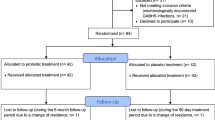
Probiotics are live microorganisms which, when administered in adequate amounts, confer a health benefit on the host. The probiotic Streptococcus salivarius has been shown to be effective in reducing the frequency of recurrent pharyngeal infections in children and adult populations. However, probiotics have not yet been evaluated in the treatment of acute pharyngotonsillitis in adults. We aimed to examine whether the addition of S. salivarius probiotics to the routine therapy of acute pharyngotonsillitis in adult patients may shorten disease duration and reduce symptom severity. This study was a prospective, randomized, placebo-controlled, double-blinded study comparing treatment with probiotics to placebo in addition to antibiotics in patients who were hospitalized with severe pharyngotonsillitis. Laboratory results, pain levels, body temperature, and daily volume of fluids consumed were recorded for both groups. Sixty participants were recruited, 30 for each group. No statistically significant differences between the two groups were observed regarding any of the major clinical and laboratory parameters examined. Supplement probiotic treatment with S. salivarius in patients with acute pharyngotonsillitis treated with penicillin is ineffective in relation to the parameters examined in this study and we cannot, therefore, recommend the use of S. salivarius during active pharyngotonsillar infection treated with penicillin.

Similar content being viewed by others
References
Shulman ST, Bisno AL, Clegg HW, Gerber MA, Kaplan EL, Lee G, Martin JM, Van Beneden C (2012) Clinical practice guideline for the diagnosis and management of group A streptococcal pharyngitis: 2012 update by the Infectious Diseases Society of America. Clin Infect Dis 55(10):1279–1282
Bisno AL (2001) Acute pharyngitis. N Engl J Med 344(3):205–211
Guarner F, Schaafsma GJ (1998) Probiotics. Int J Food Microbiol 39:237–238
Goldenberg JZ, Ma SS, Saxton JD, Martzen MR, Vandvik PO, Thorlund K, Guyatt GH, Johnston BC (2013) Probiotics for the prevention of Clostridium difficile-associated diarrhea in adults and children. Cochrane Database Syst Rev 5:CD006095
Patel RM, Lin PW (2010) Developmental biology of gut-probiotic interaction. Gut Microbes 1(3):186–195
Sazawal S, Dhingra U, Hiremath G, Sarkar A, Dhingra P, Dutta A, Verma P, Menon VP, Black RE (2010) Prebiotic and probiotic fortified milk in prevention of morbidities among children: community-based, randomized, double-blind, controlled trial. PLoS One 5(8):e12164
Pregliasco F, Anselmi G, Fonte L, Giussani F, Schieppati S, Soletti L (2008) A new chance of preventing winter diseases by the administration of synbiotic formulations. J Clin Gastroenterol 42(Suppl 3 Pt 2):S224–S233
Nogueira JC, Gonçalves Mda C (2011) Probiotics in allergic rhinitis. Braz J Otorhinolaryngol 77(1):129–134
del Giudice MM, Leonardi S, Maiello N, Brunese FP (2010) Food allergy and probiotics in childhood. J Clin Gastroenterol 44(Suppl 1):S22–S25
Ozdemir O (2010) Various effects of different probiotic strains in allergic disorders: an update from laboratory and clinical data. Clin Exp Immunol 160(3):295–304
Zhu DL, Yang WX, Yang HM (2010) Meta analysis of lactic acid bacteria as probiotics for the primary prevention of infantile eczema. Zhongguo Dang Dai Er Ke Za Zhi 12(9):734–739
Giovannini M, Agostoni C, Riva E, Salvini F, Ruscitto A, Zuccotti GV, Radaelli G; Felicita Study Group (2007) A randomized prospective double blind controlled trial on effects of long-term consumption of fermented milk containing Lactobacillus casei in pre-school children with allergic asthma and/or rhinitis. Pediatr Res 62(2):215–220
Abrahamsson TR, Jakobsson T, Böttcher MF, Fredrikson M, Jenmalm MC, Björkstén B, Oldaeus G (2007) Probiotics in prevention of IgE-associated eczema: a double-blind, randomized, placebo-controlled trial. J Allergy Clin Immunol 119(5):1174–1180
Hao Q, Lu Z, Dong BR, Huang CQ, Wu T (2011) Probiotics for preventing acute upper respiratory tract infections. Cochrane Database Syst Rev 9:CD006895
Kang JG, Kim SH, Ahn TY (2006) Bacterial diversity in the human saliva from different ages. J Microbiol 44:572–576
Guglielmetti S, Taverniti V, Minuzzo M, Arioli S, Stuknyte M, Karp M, Mora D (2010) Oral bacteria as potential probiotics for the pharyngeal mucosa. Appl Environ Microbiol 76:3948–3958
Tagg JR, Dierksen KP (2003) Bacterial replacement therapy: adapting ‘germ warfare’ to infection prevention. Trends Biotechnol 21(5):217–223
Walls T, Power D, Tagg J (2003) Bacteriocin-like inhibitory substance (BLIS) production by the normal flora of the nasopharynx: potential to protect against otitis media? J Med Microbiol 52:829–833
Barbour A, Philip K (2014) Variable characteristics of bacteriocin-producing Streptococcus salivarius strains isolated from Malaysian subjects. PLoS One 9(6):e100541
Di Pierro F, Donato G, Fomia F, Adami T, Careddu D, Cassandro C, Albera R (2012) Preliminary pediatric clinical evaluation of the oral probiotic Streptococcus salivarius K12 in preventing recurrent pharyngitis and/or tonsillitis caused by Streptococcus pyogenes and recurrent acute otitis media. Int J Gen Med 5:991–997
Di Pierro F, Adami T, Rapacioli G, Giardini N, Streitberger C (2013) Clinical evaluation of the oral probiotic Streptococcus salivarius K12 in the prevention of recurrent pharyngitis and/or tonsillitis caused by Streptococcus pyogenes in adults. Expert Opin Biol Ther 13(3):339–343
Burton JP, Chilcott CN, Tagg JR (2005) The rationale and potential for the reduction of oral malodour using Streptococcus salivarius probiotics. Oral Dis 11(Suppl 1):29–31
Burton JP, Wescombe PA, Moore CJ, Chilcott CN, Tagg JR (2006) Safety assessment of the oral cavity probiotic Streptococcus salivarius K12. Appl Environ Microbiol 72(4):3050–3053
Di Pierro F, Colombo M, Zanvit A, Risso P, Rottoli AS (2014) Use of Streptococcus salivarius K12 in the prevention of streptococcal and viral pharyngotonsillitis in children. Drug Healthc Patient Saf 6:15–20
Roos K, Holm S (2002) The use of probiotics in head and neck infections. Curr Infect Dis Rep 4(3):211–216
Taverniti V, Minuzzo M, Arioli S, Junttila I, Hämäläinen S, Turpeinen H, Mora D, Karp M, Pesu M, Guglielmetti S (2012) In vitro functional and immunomodulatory properties of the Lactobacillus helveticus MIMLh5-Streptococcus salivarius ST3 association that are relevant to the development of a pharyngeal probiotic product. Appl Environ Microbiol 78:4209–4216
Fiedler T, Riani C, Koczan D, Standar K, Kreikemeyer B, Podbielski A (2013) Protective mechanisms of respiratory tract Streptococci against Streptococcus pyogenes biofilm formation and epithelial cell infection. Appl Environ Microbiol 79(4):1265–1276
Burton JP, Wescombe PA, Macklaim JM, Chai MH, Macdonald K, Hale JD, Tagg J, Reid G, Gloor GB, Cadieux PA (2013) Persistence of the oral probiotic Streptococcus salivarius M18 is dose dependent and megaplasmid transfer can augment their bacteriocin production and adhesion characteristics. PLoS One 8(6):e65991
Cosseau C, Devine DA, Dullaghan E, Gardy JL, Chikatamarla A, Gellatly S, Yu LL, Pistolic J, Falsafi R, Tagg J, Hancock RE (2008) The commensal Streptococcus salivarius K12 downregulates the innate immune responses of human epithelial cells and promotes host-microbe homeostasis. Infect Immun 76:4163–4175
Klaenhammer TR, Kullen MJ (1999) Selection and design of probiotics. Int J Food Microbiol 50:45–57
Mohseni MJ, Aryan Z, Emamzadeh-Fard S, Paydary K, Mofid V, Joudaki H, Kajbafzadeh AM (2013) Combination of probiotics and antibiotics in the prevention of recurrent urinary tract infection in children. Iran J Pediatr 23(4):430–438
King S, Glanville J, Sanders ME, Fitzgerald A, Varley D (2014) Effectiveness of probiotics on the duration of illness in healthy children and adults who develop common acute respiratory infectious conditions: a systematic review and meta-analysis. Br J Nutr 112(1):41–54
Acknowledgments
We are grateful to SupHerb, Israel and their representative Dr. Carmela Rotem PhD, for the supply of the probiotics and the placebo.
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest. No financial support was received for this work. Probiotics and placebo were supplied by the manufacturer: SupHerb, Israel.
Ethical standards
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study.
Author information
Authors and Affiliations
Corresponding author
Additional information
NIH ClinicalTrials.gov identifier: NCT01679366.
Rights and permissions
About this article
Cite this article
Gilbey, P., Livshits, L., Sharabi-Nov, A. et al. Probiotics in addition to antibiotics for the treatment of acute tonsillitis: a randomized, placebo-controlled study. Eur J Clin Microbiol Infect Dis 34, 1011–1015 (2015). https://doi.org/10.1007/s10096-015-2315-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-015-2315-z