Abstract
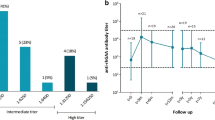
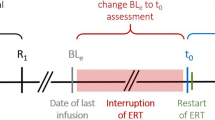
Late-onset Pompe disease (LOPD) is an autosomal recessive disorder caused by deficiency of the enzyme acid glucosidase alfa (GAA). Recently, enzyme replacement therapy (ERT) using recombinant human GAA (rhGAA) became clinically available, and is expected to modify the clinical course in LOPD of various stages. In this study, we evaluated the efficacy and adverse events of ERT for 48 weeks in Korean LOPD patients. Five Korean LOPD patients were included in the study. At baseline, clinical and laboratory features, including muscular and pulmonary function, were assessed, and rhGAA was infused every 2 weeks. Then, patients were examined at every 12-week interval for evaluation of changes in motor and pulmonary function for 48 weeks along with adverse reactions to ERT. The muscular and pulmonary function of the patients varied depending on the baseline condition of the patient after 48 weeks of ERT. However, the overall function of the patients showed stabilization of the disease rather than the improvement seen in infantile-onset Pompe disease. This is the first clinical study on ERT of Korean LOPD patients. Results of our study showed stabilization of muscular and pulmonary function in LOPD patients for 48 weeks with a favorable prognosis for patients who received early diagnosis and ambulatory patients. One of our patients developed a serious anaphylactic reaction, which necessitated the cessation of further ERT. Therefore, our study shows that early diagnosis and ERT are important in preventing further deterioration of the disease.

Similar content being viewed by others
References
van der Ploeg AT, Reuser A (2008) Lysosomal storage disease 2: Pompe’s disease. Lancet 372:1342–1353
Herzog A, Hartung R, Reuser AJ, Hermanns P, Runz H, Krabul N et al (2012) A cross-sectional single-centre study on the spectrum of Pompe disease, german patients: molecular analysis of the GAA gene, manifestation and gentotype = phenotype correlations. Orphanet J Rare Dis 7(7):35
Laforet P, Doppler V, Caillaud C, Laloui K, Claeys KG, Richard P et al (2010) Rigid spine syndrome revealing late-onset Pompe disease. Neuromuscul Disord 20:128–130
Hagemans ML, Winkel LP, Van Doorn PA, Hop WJ, Loonen MC, Reuser AJ et al (2005) Clinical manifestation and natural course of late-onset Pompe’s disease in 54 Dutch patients. Brain 128:671–677
Toscano A, Schoser B (2013) Enzyme replacement therapy in late-onset Pompe disease: a systematic literature review. J Neurol 260:951–959
Park YE, Park KH, Lee CH, Kim CM, Kim DS (2006) Two new missense mutations of GAA in late onset glycogen storage disease type II. J Neurol Sci 251:113–117
Kleyweg RP, van der Meche FG, Meulstee J (1988) Treatment of Guillain Barre syndrome with high-dose gammaglobulin. Neurology 38:1639–1641
Brooks D, Solway S, Gibbons WJ (2003) ATS statement on six-minute walk test. Am J Respir Crit Care Med 167:1287
Hobson-Webb LD, Dearmey S, Kishnani PS (2011) The clinical and electrodiagnostic characteristics of Pompe disease with post-enzyme replacement therapy findings. Clin Neurophysiol 122:2312–2317
Hobson-Webb LD, Kishnani PS (2012) How common is misdiagnosis in late-onset Pompe disease? Muscle Nerve 45:301–302
Kishnani PS, Amartino HM, Lindberg C, Miller TM, Wilson A, Keutzer J (2013) Timing of diagnosis of patents with Pompe disease: data from the Pompe registry. Am J Med Genet A 161:2431–2443
Forsha D, Li JS, Smith PB, van der Ploeg AT, Kishnani P, Pasquali SK (2011) Cardiovascular abnormalities in late-onset Pompe disease and response to enzyme replacement therapy. Genet Med 13:625–631
Soliman OI, van der Beek NA, van Doorn PA, Vietter WB, Nemes A, Van Dalen BM et al (2008) Cardiac involvement in adults with Pompe disease. J Intern Med 264:333–339
Roberts M, Kishnani PS, van der Ploeg AT, Muller-Felber W, Merlini L, Prasad S et al (2011) The prevalence and impact of scoliosis in Pompe disease: lessons learned from the Pompe Registry. Mol Genet Metab 104:574–582
Van der Ploeg AT, Clemens PR, Corzo D, Escolar DM, Florence J, Groeneveld GJ et al (2010) A randomized study of alglucosidase alfa in late-onset Pompe’s disease. N Engl J Med 362:1396–1406
van der Ploeg AT, Barohn R, Carlson L, Charrow J, Clemens PR, Hopkin RJ et al (2012) Open-label extension study following the Late-Onset Treatment Study (LOTS) of alglucosidase alfa. Mol Genet Metab 107:456–461
Hagemans ML, Winkel LP, Hop WC, Reuser AJ, Van Doorn PA, Van der Ploeg AT (2005) Disease severity in children and adults with Pompe disease related to age and duration. Neurology 64:2139–2141
Malicdan MC, Noguchi S, Nonaka I, Saftig P, Nishino I (2008) Lysosomal myopathies: an excessive build-up in autophagosomes is too much to handle. Neuromuscul Disord 18:521–529
Furusawa Y, Mori-Yoshimura M, Yamamoto T, Sakamoto C, Wakita M, Kobayashi Y, Fukumoto Y et al (2012) Effects of enzyme replacement therapy on five patients with advanced late-onset glycogen storage disease type II: 2-year follow-up study. J Inherit Metab Dis 35:301–310
Fukuda T, Ahearn M, Roberts A, Mattaliano RJ, Zaal K, Ralston E et al (2006) Autophagy and mistargeting of the therapeutic enzyme in skeletal muscle in Pompe disease. Mol Ther 14:831–839
Myozyme Prescribing information. Genzyme Corporation. http://www.Myozyme.com
Banugaria SG, Prater SN, Ng YK, Kobori JA, Finkel RS, Ladda RL (2011) The impact of antibodies on clinical outcomes in diseases treated with therapeutic protein: lessons learned from infantile Pompe disease. Genet Med 13:729–736
Lacana E, Yao LP, Pariser AR, Rosenberg AS (2012) The role of immune tolerance induction in restoration of the efficacy of ERT in Pompe disease. Am J Med Genet C Semin Med Genet 160:30–39
Patel TT, Banugaria SG, Case LE, Wenninger S, Schoser B, Kishnani PS (2012) The impact of antibodies in late-onset Pompe disease: a case series and literature review. Mol Genet Metab 106:301–309
Acknowledgments
This work was supported by 2-year research grant from Pusan National University.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Park, JS., Kim, HG., Shin, JH. et al. Effect of enzyme replacement therapy in late onset Pompe disease: open pilot study of 48 weeks follow-up. Neurol Sci 36, 599–605 (2015). https://doi.org/10.1007/s10072-014-2000-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-014-2000-5