Abstract
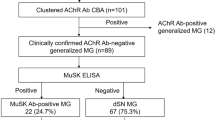
Anti-acetylcholine receptor antibodies (anti-AChR-Ab) are responsible for the failure of neuromuscular junction in myasthenia gravis (MG). Some anti-AChR-Ab-seronegative MG patients have anti-muscle-specific tyrosine kinase antibodies (anti-MuSk-Ab). Here, the anti-AChR-Ab was tested in 250 MG outpatients from Southern China. While anti-MuSk-Ab was tested in 66 patients who had no anti-AChR-Ab in blood serum, but none of them was positive. The antibodies were measured by a radioimmunoprecipitation assay. The frequency of anti-AChR-Ab was 51.2 %. The percentage of anti-AChR-Ab in ocular type was lower than generalized type (44.9 vs. 66.2 %, P = 0.002). Seronegative MG was characterized by a lower percentage of thymoma than seropositive patients (P = 0.013). It seemed to be less severe in seronegative MG than seropositive MG in these 250 patients. In ocular type, seronegative MG mainly manifesting blepharoptosis but seldom diplopia or eyeball fixation related to ocular movement disability (P = 0.016). While in generalized type, seronegative MG was characterized by a lower percentage of bulbar muscle involvements than seropositive patients (P = 0.005). Logistic regression analysis revealed that bulbar weakness was affected by the existence of anti-AChR antibodies (OR = 3.524, P = 0.015). Besides, seronegative MG tended to be characterized by a lower percentage of neck extensor involvement, but this did not reach significance. The percentage of anti-AChR antibodies was much lower than other countries. Seronegative MG has characteristic clinical features that are different from features of the remaining seropositive MG. This emphasises the predictive value of anti-AChR antibodies analysis in MG patients.


Similar content being viewed by others
References
Zhang X, Yang M, Xu J, Zhang M, Lang B, Wang W, Vincent A (2007) Clinical and serological study of myasthenia gravis in HuBei Province China. J Neurol Neurosurg Psychiatry 4:386–390
McConville J, Farrugia ME, Beeson D, Kishore U, Metcalfe R, Newsom-Davis J, Vincent A (2004) Detection and characterization of MuSk antibodies in seronegative myasthenia gravis. Ann Neurol 4:580–584
Chan KH, Lachance DH, Harper CM, Lennon VA (2007) Frequency of seronegativity in adult-acquired generalized myasthenia gravis. Muscle Nerve 5:651–658
Hoch W, McConville J, Helms S, Newsom-Davis J, Melms A, Vincent A (2001) Auto-antibodies to the receptor tyrosine kinase MuSk in patients with myasthenia gravis without acetylcholine receptor antibodies. Nat Med 3:365–368
Vincent A, Leite MI (2005) Neuromuscular junction autoimmune disease: muscle specific kinase antibodies and treatments for myasthenia gravis. Curr Opin Neurol 5:519–525
Liu WB, Xia Q, Men LN, Wu ZK, Huang RX (2007) Dysphonia as a primary manifestation in myasthenia gravis (MG): a retrospective review of 7 cases among 1,520 MG patients. J Neurol Sci 1–2:16–22
Yeh JH, Chen WH, Chiu HC, Vincent A (2004) Low frequency of MuSk antibody in generalized seronegative myasthenia gravis among Chinese. Neurology 11:2131–2132
Tsinzerling N, Lefvert AK, Matell G, Pirskanen-Matell R (2007) Myasthenia gravis: a long term follow-up study of Swedish patients with specific reference to thymic histology. J Neurol Neurosurg Psychiatry 10:1109–1112
Hewer R, Matthews I, Chen S, McGrath V, Evans M, Roberts E, Nute S, Sanders J, Furmaniak J, Smith BR (2006) A sensitive non-isotopic assay for acetylcholine receptor autoantibodies. Clin Chim Acta 1–2:159–166
Ohta M, Ohta K, Mori F, Itoh N, Nishitani H, Hayashi K (1990) Improved radioassay of anti-acetylcholine receptor antibody: application for the detection of extremely low antibody titers in sera from patients with myasthenia gravis. Clin Chem 6:911–913
Compston DA, Vincent A, Newsom-Davis J, Batchelor JR (1980) Clinical, pathological, HLA antigen and immunological evidence for disease heterogeneity in maysthenia gravis. Brain 3:579–601
Chiu HC, Vincent A, Newsom-Davis J, Hsieh KH, Hung T (1987) Myasthenia gravis: population differences in disease expression and acetylcholine receptor antibody titers between Chinese and Caucasians. Neurology 12:1854–1857
Somnier FE (2005) Increasing incidence of late-onset anti-AChR antibody seropositive myast henia gravis. Neurology 6:928–930
Uono MI (1980) Clinical statistics of myasthenia gravis in Japan. Int J Neurol 1:87–99
DeChiara TM, Bowen DC, Valenzuela DM, Simmons MV, Poueymirou WT, Thomas S, Kinetz E, Compton DL, Rojas E, Park JS, Smith C, DiStefano PS, Glass DJ, Burden SJ, Yancopoulos GD (1996) The receptor tyrosine kinase MuSk is required for neuromuscular junction formation in vivo. Cell 4:501–512
Ohta K, Shigemoto K, Kubo S, Maruyama N, Abe Y, Ueda N, Ohta M (2004) MuSk antibodies in AChR Ab-seropositive MG vs AChR Ab-seronegative MG. Neurology 11:2132–2133
Romi F, Aarli JA, Gilhus NE (2005) Seronegative myasthenia gravis: disease severity and prognosis. Eur J Neurol 6:413–418
Deymeer F, Gungor-Tuncer O, Yilmaz V, Parman Y, Serdaroglu P, Ozdemir C, Vincent A, Saruhan-Direskeneli G (2007) Clinical comparison of anti-MuSk- vs anti-AChR-positive and seronegative myasthenia gravis. Neurology 8:609–611
Lauriola L, Ranelletti F, Maggiano N, Guerriero M, Punzi C, Marsili F, Bartoccioni E, Evoli A (2005) Thymus changes in anti-MuSk-positive and negative myasthenia gravis. Neurology 3:536–538
De Baets M, Stassen MH (2002) The role of antibodies in myasthenia gravis. J Neurol Sci 1–2:5–11
Acknowledgments
The study was supported by the National Natural Science Foundation of China (30870850, 81071002) and Sun Yat-sen University Clinical Research 5010 Program (2010003).The work is attributed to the Department of Neurology, Affiliated first Hospital, Sun Yat-sen University, Guangzhou 510080, China.
Author information
Authors and Affiliations
Corresponding author
Additional information
H. Feng and H. Wang contributed equally to this work.
Rights and permissions
About this article
Cite this article
Feng, Hy., Wang, Hy., Liu, Wb. et al. The high frequency and clinical feature of seronegative myasthenia gravis in Southern China. Neurol Sci 34, 919–924 (2013). https://doi.org/10.1007/s10072-012-1159-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-012-1159-x