Abstract
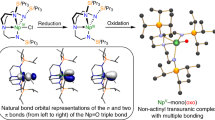
The structures, stabilities, nature of bonding, and spectroscopic properties of the new actinide imine molecules, neptunimine (HN=NpH2) and plutonimine (HN=PuH2), in the gas phase have been systematically explored at different levels of theory. Our calculation indicates that HN=AnH2 (An=Np, Pu) should be nonplanar and have a quartet (\( {\tilde{\mathrm{X}}}^4\mathrm{A} \)) and quintet (\( {\tilde{\mathrm{X}}}^5\mathrm{A} \)) ground state, respectively. The nature of the chemical bonding in these molecules were investigated by employing topological methods including electron localization function (ELF), atoms in molecules (AIM) as well as natural bond orbital analysis (NBO). The results showed that these actinide complexes possess relatively strong An=N multiple bonds between the An 6d-5f hybrid orbitals with N 2s-2p orbitals. The charge decomposition analysis (CDA) diagram demonstrated that the transition of electrons mainly happened inside the AnH2 of HN=AnH2. Total and partial density of state (TDOS and PDOS) and also overlap population density of state (OPDOS) diagrams analysis were implemented. The IR and Raman spectra were theoretically simulated as a convenient way to confirm the existence of the actinide imine complexes in further experiments.







Similar content being viewed by others
References
Brennan JG, Andersen RA (1985) J Am Chem Soc 107:514–516
Stevens RC, Bau R, Cramer RE, Afzal D, Gilje JW, Koetzle TF (1990) Organometallics 9:694–697
Gagliardi L, Pyykkö P (2004) Angew Chem Int Ed 43:1573–1576
Pyykkö P, Riedel S, Patzschke M (2005) Chem Eur J 11:3511–3520
Burns CJ (2005) Science 309:1823–1824
Evans WJ, Kozimor SA, Ziller JW (2005) Science 309:1835–1838
Hayton TW, Boncella JM, Scott BL, Palmer PD, Batista ER, Hay PJ (2005) Science 310:1941–1943
Hayton TW, Boncella JM, Scott BL, Batista ER, Hay PJ (2006) J Am Chem Soc 128:10549–10559
Frenking G, Tonner R (2007) Nature 446:276–277
Lyon JT, Hu HS, Andrews L, Li J (2007) PNAS 104:18919–18924
Graves CR, Yang P, Kozimor SA, Vaughn AE, Clark DL, Conradson SD, Schelter EJ, Scott BL, Thompson JD, Hay PJ, Morris DE, Kiplinger JL (2008) J Am Chem Soc 130:5272–5285
Cantat T, Arliguie T, Noel A, Thuery P, Ephritikhine M, Le Floch P, Mezailles N (2009) J Am Chem Soc 131:963–972
Thomson RK, Cantat T, Scott BL, Morris DL, Batista ER, Kiplinger JL (2010) Nat Chem 2:723–729
Fox AR, Arnold PL, Cummins CC (2010) J Am Chem Soc 132:3250–3251
Wang XF, Andrews L, Vlaisavljevich B, Gagliardi L (2011) Inorg Chem 50:3826–3831
King DM, Tuna F, McInnes EL, McMaster J, Lewis W, Blake AJ, Liddle ST (2013) Nat Chem 5:482–488
Fox AR, Bart SC, Meyer K, Cummins CC (2008) Nature 455:341–349
Wang XF, Andrews L, Marsden CJ (2007) Chem Eur J 13:5601–5606
Wang XF, Andrews L, Marsden CJ (2008) Chem Eur J 14:9192–9201
Li P, Niu WX, Gao T (2014) RSC Adv 4:29806–29817
Lee C, Yang W, Parr RG (1988) Phys Rev B 37:785–789
Becke AD (1993) J Chem Phys 98:5648–5652
Perdew JP, Burke K, Wang Y (1996) Phys Rev B 54:16533–16539
Becke AD (1998) Phys Rev A 38:3098–3100
Adamo C, Barone V (1999) J Chem Phys 110:6158–6170
Kuchle W, Dolg M, Stoll H, Preuss H (1994) J Chem Phys 100:7535–7542
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Montgomery JA Jr, Vreven T, Kudin KN et al. (2004) Gaussian 03, revision E.01. Gaussian, Inc, Wallingford
Michelini MC, Russo N, Sicilia E (2006) Angew Chem Int Ed 45:1095–1099
Alikhani ME, Michelini MC, Russo N, Silvi B (2008) J Phys Chem A 112:12966–12974
Zhou J, Schlegel HB (2010) J Phys Chem A 114:8613–8617
Li P, Niu WX, Gao T, Wang HY (2014) Int J Quantum Chem 114:760–768
De Almeida KJ, Duarte HA (2010) Organometallics 29:3735–3745
Lu T, Chen F (2012) J Comput Chem 33:580–592
Becke AD, Edgecombe KE (1990) J Chem Phys 92:5397–5403
Savin A, Nesper R, Wengert S, Fassler TR (1997) Angew Chem Int Ed Engl 36:1808–1832
Bader RFW (1990) Atoms in molecules. A quantum theory. Clarendon, Oxford
Hoffmann R (1988) Solids and surfaces: a chemist’s view of bonding in extended structures. VCH, New York
Malecki JG (2010) Polyhedron 29:1973–1979
Dapprich S, Frenking G (1995) J Phys Chem 99:9352–9362
Sousa SF, Fernandes PA, Ramos MJ (2007) J Phys Chem A 111:10439–10452
Bakken V, Millam JM, Schlegel HB (1999) J Chem Phys 111:8773–8777
Michelini MC, Russo N, Sicilis E (2007) J Am Chem Soc 129:4229–4239
Di Santo E, Michelini MC, Russo N (2009) Organometallics 28:3716–3726
Palusiak M, Krygowski TM (2007) Chem Eur J 13:7996–8006
Farrugia LJ, Senn HM (2010) J Phys Chem A 114:13418–13433
De Almeida KJ, Ramalho TC, Neto JL, Santiago RT, Felicíssimo VC, Duarte HA (2013) Organometallics 32:989–999
Cremer D, Kraka E (1984) Angew Chem Int Ed Engl 23:627–628
Du J, Sun X, Chen J, Zhang L, Jiang G (2014) Dalton Trans 43:5574–5579
Vyboishchikov SF, Sierraalta A, Frenking G (1996) J Comput Chem 18:416–429
Weinhold F, NBO 5.0 Program Manual
Acknowledgments
We are very grateful to Dr. Sobereva for many helpful discussions and providing us with the Multiwfn package. Computer time made available by the Center of High Performance Computing at Physics discipline of Sichuan University is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, P., Niu, W. & Gao, T. Systematic analysis of structural and spectroscopic properties of neptunimine (HN=NpH2) and plutonimine (HN=PuH2). J Mol Model 21, 316 (2015). https://doi.org/10.1007/s00894-015-2856-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00894-015-2856-1