Abstract
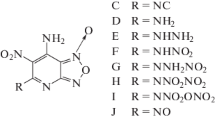
5-Nitro-3-trinitromethyl-1H-1,2,4-triazole (NTMT, A) and its substituted derivatives A–CH3, A–OCH3, A–NH2, A–OH, A–NO2, and A–ONO2 were studied using density functional theory (DFT). For all of the molecules except for A–ONO2, the C–NO2 bond in the trinitromethyl group was found to be the weakest, and no transition state occurred during the scission of this bond. The weakest C–NO2 of the trinitromethyl group bond dissociation energies for all of the molecules were all very similar. Most of the title molecules had similar frontier orbital distributions and comparable energy gaps between the frontier orbitals. The impact sensitivity (h 50, in cm), predicted at various levels of theory, decreased in the order A–NH2 (53.0–71.0) > A–CH3 (53.0) > A (36.7) > A–OCH3 (32.6–42.3) > A–OH (26.7–53.0) > A–NO2 (5.6–7.4) > A–ONO2 (4.6–6.1). Their detonation velocities (D), detonation pressures (P), and specific impulses (I s) were 8.02–8.82 km/s, 29.92–35.54 GPa, and 214–260 s, respectively. Composite explosives made from hexahydro-1,3,5-trinitro-1,3,5-triazine (RDX) and A, A–OH, A–NH2, A–NO2, or A–ONO2 as an oxidizer were found to possess much better detonation performance (D = 9.04–9.29 km/s, P = 37.25–39.26 GPa, and I s = 270–281 s). Thus, introducing –OCH3, –OH, and –NH2 groups into A produced new explosives with acceptable stability and good detonation performance. A–OH and A–NH2 appear to be promising candidates for oxidizers in composite explosives.







Similar content being viewed by others
References
Rahm M, Brinck T (2010) Kinetic stability and propellant performance of green energetic materials. Chem Eur J 16(22):6590–6600
Klapötke TM, Krumm B, Moll R, Rest SF (2011) CHNO based molecules containing 2,2,2-trinitroethoxy moieties as possible high energy dense oxidizers. Z Anorg Allg Chem 637(14–15):2103–2110
Badgujar DM, Talawar MB, Asthana SN, Mahulikar PP (2008) Advances in science and technology of modern energetic materials: an overview. J Hazard Mater 151(2):289–305
Sikder A, Sikder N (2004) A review of advanced high performance, insensitive and thermally stable energetic materials emerging for military and space applications. J Hazard Mater 112(1):1–15
Urbanski T (1964) Chemistry and technology of explosives, vol 6. Pergamon, New York
Agrawal JP (1998) Recent trends in high-energy materials. Prog Energ Combust 24(1):1–30
Singh G, Kapoor IPS, Mannan SM, Kaur J (2000) Studies on energetic compounds. Part 8: Thermolysis of Salts of HNO3 and HClO4. J Hazard Mater 79(1):1–18
Fried LE, Manaa MR, Pagoria PF, Simpson RL (2001) Design and synthesis of energetic materials 1. Ann Rev Mater Res 31(1):291–321
Shlyapochnikov V, Tafipolsky M, Tokmakov I, Baskir E, Anikin O, Strelenko YA, Luk'yanov O, Tartakovsky V (2001) On the structure and spectra of dinitramide salts. J Mol Struct 559(1):147–166
Pagoria PF, Lee GS, Mitchell AR, Schmidt RD (2002) A review of energetic materials synthesis. Thermochim Acta 384(1):187–204
An C, Li H, Geng X, Li J, Wang J (2013) Preparation and properties of 2,6-diamino-3,5-dinitropyrazine-1-oxide based nanocomposites. Propell Explos Pyrot 38(2):172–175
Zhang J, Wu P, Yang Z, Gao B, Zhang J, Wang P, Nie F, Liao L (2014) Preparation and properties of submicrometer-sized LLM-105 via spray-crystallization method. Propell Explos Pyrot 39(5):653–657
Bolton O, Simke LR, Pagoria PF, Matzger AJ (2012) High power explosive with good sensitivity: a 2:1 cocrystal of CL-20: HMX. Cryst Growth Des 12(9):4311–4314
Bayat Y, Zeynali V (2011) Preparation and characterization of nano-CL-20 explosive. J Energ Mater 29(4):281–291
Mandal AK, Thanigaivelan U, Pandey RK, Asthana S, Khomane RB, Kulkarni BD (2012) Preparation of spherical particles of 1,1-diamino-2,2-dinitroethene (FOX-7) using a micellar nanoreactor. Org Process Res Dev 16(11):1711–1716
Vo TT, Zhang J, Parrish DA, Twamley B, Shreeve JM (2013) New roles for 1,1-diamino-2,2-dinitroethene (FOX-7): halogenated FOX-7 and azo-bis (diahaloFOX) as energetic materials and oxidizers. J Am Chem Soc 135(32):11787–11790
Behrens R Jr, Bulusu S (2013) Thermal decomposition studies of a new, gem-dinitroalkyl nitramine 1,3,3-trinitroazetidine (TNAZ). Defence Sci J 46(5):361–369
Ma H, Feng X, Zhu T, Miao C, Ma Y, Feng M (2012) Research progress in high energy density material 1,3,3-trinitroazetidine. Chem Propell Ploym Mater 10(4):20–24
Zhang C (2009) Review of the establishment of nitro group charge method and its applications. J Hazard Mater 161(1):21–28
Katritzky A, Sommen G, Gromova A, Witek R, Steel P, Damavarapu R (2005) Synthetic routes towards tetrazolium and triazolium dinitromethylides. Chem Heterocycl Com 41(1):111–118
Göbel M, Klapötke TM (2009) Development and testing of energetic materials: the concept of high densities based on the trinitroethyl functionality. Adv Funct Mater 19(3):347–365
Puchala A, Belaj F, Bergman J, Kappe CO (2001) On the reaction of 3, 4-dihydropyrimidones with nitric acid. Preparation and X-ray structure analysis of a stable nitrolic acid. J Heterocyclic Chem 38(6):1345–1352
Bellamy AJ, Latypov NV, Goede P (2002) Transamination reactions of 1,1-diamino-2,2-dinitroethene (FOX-7). J Chem Res 2002(6):257–257
Zeng Z, Gao H, Twamley B, Shreeve JM (2007) Energetic mono and dibasic 5-dinitromethyltetrazolates: synthesis, properties, and particle processing. J Mater Chem 17(36):3819–3826
Oh CH, Park DI, Ryu JH, Cho JH, Han J (2007) Syntheses and characterization of cyclopropane-fused hydrocarbons as new high energetic materials. B Korean Chem Soc 28(2):322
Bulusu S, Damavarapu R, Autera J, Behrens R Jr, Minier L, Villanueva J, Jayasuriya K, Axenrod T (1995) Thermal rearrangement of 1,4-dinitroimidazole to 2,4-dinitroimidazole: characterization and investigation of the mechanism by mass spectrometry and isotope labeling. J Phys Chem 99(14):5009–5015
Minier L, Behrens R, Bulusu S (1996) Mass spectra of 2,4‐dinitroimidazole and its isotopomers using simultaneous thermogravimetric modulated beam mass spectrometry. J Mass Spectrom 31(1):25–30
Bracuti A (1995) Crystal structure of 2,4-dinitroimidazole (24DNI). J Chem Crystallogr 25(10):625–627
Cho SG, Park BS, Cho JR (1999) Theoretical studies on the structure of 1,2,4,5-tetranitroimidazole. Propell Explos Pyrot 24(6):343–348
Rice BM, Hare JJ (2002) A quantum mechanical investigation of the relation between impact sensitivity and the charge distribution in energetic molecules. J Phys Chem A 106(9):1770–1783
Cho JR, Kim KJ, Cho SG, Kim JK (2002) Synthesis and characterization of 1-methyl-2,4,5-trinitroimidazole (MTNI). J Heterocyclic Chem 39(1):141–148
Thottempudi V, Gao H, Shreeve JM (2011) Trinitromethyl-substituted 5-nitro-or 3-azo-1,2,4-triazoles: synthesis, characterization, and energetic properties. J Am Chem Soc 133(16):6464–6471
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Montgomery JA, Vreven T, Kudin KN, Burant JC, Millam JM, Iyengar SS, Tomasi J, Barone V, Mennucci B, Cossi M, Scalmani G, Rega N, Petersson GA, Nakatsuji H, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Klene M, Li X, Knox JE, Hratchian HP, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Ayala PY, Morokuma K, Voth GA, Salvador P, Dannenberg JJ, Zakrzewski VG, Dapprich S, Daniels AD, Strain MC, Farkas O, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Ortiz JV, Cui Q, Baboul AG, Clifford S, Cioslowski J, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Gonzalez C, Pople JA (2004) Gaussian 03, revision C02. Gaussian, Inc., Wallingford
Benson SW (1976) Thermochemical kinetics: methods for the estimation of thermochemical data and rate parameters. Wiley, New York
Yao XQ, Hou XJ, Wu GS, Xu YY, Xiang HW, Jiao H, Li YW (2002) Estimation of CC bond dissociation enthalpies of large aromatic hydrocarbon compounds using DFT methods. J Phys Chem A 106(31):7184–7189
Shao J, Cheng X, Yang X (2005) Density functional calculations of bond dissociation energies for removal of the nitrogen dioxide moiety in some nitroaromatic molecules. J Mol Struc-Theochem 755(1):127–130
Fan XW, Ju XH, Xia QY, Xiao HM (2008) Strain energies of cubane derivatives with different substituent groups. J Hazard Mater 151(1):255–260
Politzer P, Martinez J, Murray JS, Concha MC, Toro-Labbe A (2009) An electrostatic interaction correction for improved crystal density prediction. Mol Phys 107(19):2095–2101
Chase MW Jr (1998) NIST-JANAF thermochemical tables, 4th edn. J Phys Chem Ref Data Monogr 9:1–1951
Rice BM, Pai SV, Hare J (1999) Predicting heats of formation of energetic materials using quantum mechanical calculations. Combust Flame 118(3):445–458
Politzer P, Lane P, Murray JS (2011) Computational characterization of a potential energetic compound: 1,3,5,7-tetranitro-2,4,6,8-tetraazacubane. Cent Eur J Energetic Mater 8(1):39–52
Kamlet MJ, Jacobs SJ (1986) A simple method for calculating detonation properties of CHNO explosives. J Chem Phys 48:23–25
Politzer P, Murray JS, Grice ME, Sjoberg P (eds) (1991) Computer-aided design of monopropellants. In: Olah GA, Squire DR (eds) Chemistry of energetic materials. Academic, San Diego
Benson SW (ed) (1976) Thermochemical kinetics, 2nd edn. Wiley, New York
Oxley JC, Smith JL, Ye H, McKenney RL, Bolduc PR (1995) Thermal stability studies on a homologous series of nitroarenes. J Phys Chem 99(23):9593–9602
Tsang W, Robaugh D, Mallard WG (1986) Single-pulse shock-tube studies on C–NO2 bond cleavage during the decomposition of some nitro aromatic compounds. J Phys Chem 90(22):5968–5973
Li J (2010) Relationships for the impact sensitivities of energetic C-nitro compounds based on bond dissociation energy. J Phys Chem B 114(6):2198–2202
Song X, Cheng X, Yang X, Li D, Linghu R (2008) Correlation between the bond dissociation energies and impact sensitivities in nitramine and polynitro benzoate molecules with polynitro alkyl groupings. J Hazard Mater 150(2):317–321
Chen G, Shi WY, Xia MZ, Lei W, Wang FY, Gong XD (2014) Theoretical study of solvent effects on RDX crystal quality and sensitivity using an implicit solvation model. J Mol Model 20(7):1–9
Murray JS, Concha MC, Politzer P (2009) Links between surface electrostatic potentials of energetic molecules, impact sensitivities and C–NO2/N–NO2 bond dissociation energies. Mol Phys 107(1):89–97
Politzer P, Murray JS (2014) Detonation performance and sensitivity: a quest for balance. Adv Quantum Chem 69:1–30
Ju XH, Wang ZY, Yan XF, Xiao HM (2007) Density functional theory studies on dioxygen difluoride and other fluorine/oxygen binary compounds: availability and shortcoming. J Mol Struc-Theochem 804(1):95–100
Zhou Z, Parr RG, Garst JF (1988) Absolute hardness as a measure of aromaticity. Tetrahedron Lett 29(38):4843–4846
Lu T, Chen F (2012) Multiwfn: a multifunctional wavefunction analyzer. J Comput Chem 33(5):580–592
Zhang XL, Gong XD (2014) Theoretical studies on the stability, detonation performance and possibility of synthesis of the nitro derivatives of epoxyethane. J Mol Model 20(8):1–11
Zhang XL, Yang JQ, Wang TY, Gong XD (2014) A theoretical study on the stability and etonation performance of 2,2,3,3-tetranitroaziridine (TNAD). J Phys Org Chem
Li ZX (2005) Study on the performance of several kinds of furazan energetic derivatives. Energ Mater 13(2):90–93
Chase MW, Force JANA (eds) (1998) NIST-JANAF thermochemical tables. American Chemical Society, Washington, DC
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 27 kb)
Rights and permissions
About this article
Cite this article
Zhang, X., Gong, X. Theoretical study of the stabilities and detonation performance of 5-nitro-3-trinitromethyl-1H-1,2,4-triazole and its derivatives. J Mol Model 21, 26 (2015). https://doi.org/10.1007/s00894-015-2581-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00894-015-2581-9