Abstract
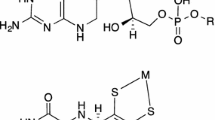
Molybdenum is found in the active site of enzymes usually coordinated by one or two pyranopterin molecules. Here, we mimic an enzyme with a mononuclear molybdenum-bis pyranopterin center by incorporating molybdenum in rubredoxin. In the molybdenum-substituted rubredoxin, the metal ion is coordinated by four sulfurs from conserved cysteine residues of the apo-rubredoxin and two other exogenous ligands, oxygen and thiol, forming a Mo(VI)-(S-Cys)4(O)(X) complex, where X represents –OH or –SR. The rubredoxin molybdenum center is stabilized in a Mo(VI) oxidation state, but can be reduced to Mo(IV) via Mo(V) by dithionite, being a suitable model for the spectroscopic properties of resting and reduced forms of molybdenum-bis pyranopterin-containing enzymes. Preliminary experiments indicate that the molybdenum site built in rubredoxin can promote oxo transfer reactions, as exemplified with the oxidation of arsenite to arsenate.






Similar content being viewed by others
Abbreviations
- apo-Rd:
-
Apo-rubredoxin
- β-ME:
-
β-Mercaptoethanol
- BSO:
-
Biotin sulfoxide
- CT:
-
Charge transfer
- CV:
-
Cyclic voltammetry
- DMSOR:
-
Dimethylsulfoxide reductase
- DTT:
-
Dithiothreitol
- DPV:
-
Differential pulsed voltammetry
- Fe-Rd:
-
Iron-containing rubredoxin
- GC:
-
Glassy carbon
- hSO:
-
Human sulfite oxidase
- M:
-
Metal
- Mo-bis PGD:
-
Molybdenum-bis pyranopterin guanosine dinucleotide-containing enzyme
- Mo-Rd:
-
Molybdenum-substituted rubredoxin
- PGE:
-
Pyrolytic graphite electrode
- NHE:
-
Normal hydrogen electrode
- RR:
-
Resonance Raman
- Rd:
-
Rubredoxin
- TCA:
-
Trichloroacetic acid
- TFET:
-
2,2,2-Trifluoroethanethiol
References
Lu Y, Berry SM, Pfister TD (2001) Chem Rev 101:3047–3080
Liu J, Meier KK, Tian S, Zhang J, Guo H, Schulz CE, Robinson H, Nilges MJ, Münck E, Lu Y (2014) J Am Chem Soc 136:12337–12344
Yu F, Cangelosi VM, Zastrow ML, Tegoni M, Plegaria JS, Tebo AG, Mocny CS, Ruckthong L, Qayyum H, Pecoraro VL (2014) Chem Rev 114:3495–3578
Adman ET, Sieker LC, Jensen LH, Bruschi M, Le Gall J (1977) J Mol Biol 112:113–120
Chen CJ, Liu MY, Chen YT, LeGall J (2003) Biochem Biophys Res Commun 308:684–688
Moura I, Bruschi M, LeGall J, Moura JJG, Xavier AV (1977) Biochem Biophys Res Commun 75:1037–1044
Bruschi M, Moura I, Le Gall J, Xavier AV, Sieker LC, Couchoud P (1979) Biochem Biophys Res Commun 90:596–605
Czaja C, Litwiller R, Tomlinson AJ, Naylor S, Tavares P, LeGall J, Moura JJG, Moura I, Rusnak F (1995) J Biol Chem 270:20273–20277
Yu L, Kennedy M, Czaja C, Tavares P, Moura JJ, Moura I (1997) Rusnak FBiochem Biophys Res Commun 231:679–682
Dauter Z, Wilson KS, Sieker LC, Moulis JM, Meyer J (1996) Proc Natl Acad Sci USA 93:8836–8840
Archer M, Carvalho AL, Teixeira S, Moura I, Moura JJG, Rusnak F, Romao MJ (1999) Protein Sci 8:1536–1545
Ascenso C, Rusnak F, Cabrito I, Lima MJ, Naylor S, Moura I, Moura JJG (2000) J Bio Inorg Chem 5:720–729
Sun N, Dey A, Xiao Z, Wedd AG, Hodgson KO, Hedman B, Solomon EI (2010) J Am Chem Soc 132:12639–12647
May SW, Kuo JY (1978) Biochemistry 17:3333–3338
Kowal AT, Zambrano IC, Moura I, Moura JJG, LeGall J, Johnson MK (1988) Inorg Chem 27:1162–1166
Saint-Martin P, Lespinat PA, Fauque G, Berlier Y, Legall J, Moura I, Teixeira M, Xavier AV, Moura JJG (1988) Proc Natl Acad Sci USA 85:9378–9380
Moura I, Teixeira M, LeGall J, Moura JJG (1991) J Inorg Biochem 44:127–139
Henehan CJ, Pountney DL, Zerbe O, Vasak M (1993) Protein Sci 2:1756–1764
Ravi N, Prickril BC, Kurtz DM Jr, Huynh BH (1993) Biochemistry 32:8487–8491
Faller P, Ctortecka B, Tröger W, Butz T, Vasák M (2000) J Biol Inorg Chem 5:393–401
Maher M, Cross M, Wilce MCJ, Guss JM, Wedd AG (2004) Acta Cryst D 60:298–303
Mathies G, Almeida RM, Gast P, Moura JJG, Groenen EJJ (2012) J Phys Chem B 116:7122–7128
Thapper A, Rizzi AC, Brondino CD, Wedd AG, Pais RJ, Maiti BK, Moura I, Pauleta SR, Moura JJG (2013) J Inorg Biochem 127:232–237
Hille R (1996) Chem Rev 96:2757–2816
Brondino CD, Romao MJ, Moura I, Moura JJG (2006) Curr Opin Chem Biol 10:109–114
Hille R, Hall J, Basu P (2014) Chem Rev 114:3963–4038
Rothery RA, Workun GJ, Weiner JH (2008) Biochim Biophys Acta 1778:1897–1929
Grimaldi S, Schoepp-Cothenet B, Ceccaldi P, Guigliarelli B, Magalon A (2013) Biochem Biophys Acta 1827:1048–1085
Rajagopalan KV (1980) Sulfite Oxidase. In: Coughlan MP (ed) Molybdenum and molybdenum containing enzymes, chap 7. Pergamon Press, Oxford, pp 241–272
Pushie MJ, George GN (2011) Coord Chem Rev 255:1055–1084
Stiefel EL (2002) The biogeochemistry of molybdenum and tungsten. In: Sigel A, Sigel H (eds) Molybdenum and tungsten: their roles in biological processes. Metals ions in biological system, chap 1, vol 39. CRC Press, pp 1–29
Mendel RR (2005) Dalton Trans 3404–3409
Almeida RM, Pauleta SR, Moura I, Moura JJG (2009) J Inorg Biochem 103:1245–1253
Bruschi M, Hatchikian CE, Golovleva LA, LeGall J (1977) J Bacteriol 129:30–38
Lode ET, Coon MJ (1971) J Biol Chem 246:791–802
Spence JT, Chang HYY (1963) Inorg Chem 2:319–323
Buchanan I, Minelli M, Ashby MT, King TJ, Enemark JH, Garner CD (1984) Inorg Chem 23:495–500
Bishop PT, Dilworth JR, Hutchinson JP, Zubieta JA (1990) Trans Met Chem 15:177–182
Huang TJ, Haight GP Jr (1970) J Am Chem Soc 92:2336–2342
Ueyama N, Okamura T, Nakamura A (1992) J Am Chem Soc 114:8129–8137
Murphy J, Riley JP (1962) Anal Chim Acta 27:31–36
Tsang S, Phu F, Baum MM, Poskrebyshev GA (2007) Talanta 71:1560–1568
Yang J, Rothery R, Sempombe J, Weiner JH, Kirk ML (2009) J Am Chem Soc 131(43):15612–15614
Liu S, Sun X, Zubieta JA (1988) J Am Chem Soc 110:3324–3326
Brondino CD, Rivas MG, Romao MJ, Moura JJG, Moura I (2006) Acc Chem Res 29:7887–7896
Klein EL, Astashkin AV, Raitsimring AM, Enemark JH (2013) Coord Chem Rev 257:110–118
Izumi Y, Glaser T, Rose K, McMaster J, Basu P, Enemark JH, Hedman B, Hodgson KO, Solomon EI (1999) J Am Chem Soc 121:10035–10046
Subramanian P, Burgmayer S, Richards S, Szalai V, Spiro TG (1990) Inorg Chem 29:3849–3853
Johnson MK (2004) Prog Inorg Chem 52:213–266
Ueyama N, Nakata M, Araki T, Nakamura A, Yamashita S, Yamashita T (1981) Inorg Chem 20:1934–1937
Garrett RM, Rajagopalan KV (1996) J Biol Chem 271:7387–7391
Czernuszewicz RS, LeGall J, Moura I, Spiro TG (1986) Inorg Chem 25:696–700
Garton SD, Garrett RM, Rajagopalan KV, Johnson MK (1997) J Am Chem Soc 119:2590–2591
Garton SD, Temple CA, Dhawan IK, Barber MJ, Rajagopalan KV, Johnson MK (2000) J Biol Chem 275:6798–6805
Ellis PJ, Conrads T, Hille R, Kuhn P (2001) Structure 9:125–132
Conrads T, Hemann C, George GN, Pickering IJ, Prince RC, Hille R (2002) J Am Chem Soc 124:11276–11277
Roy R, Adams MW (2002) Met Ions Biol Syst 39:673–697
Caradonna JP, Harlan EW, Holm RH (1986) J Am Chem Soc 108:7856–7858
Tierney DL, Gassner GT, Luchinat C, Bertini I, Ballou DP, Penner-Hahn JE (1999) Biochemistry 38:11051–11061
Bonaccio M, Ghaderi N, Borchardt D, Dunn MF (2005) Biochemistry 44:7656–7668
Bray RC, Adams B, Smith AT, Bennett B, Bailey S (2000) Biochemistry 39:11258–11269
Klein EL, Raitsimring AM, Astashkin AV, Rajapakshe A, Johnson-Winters K, Arnold AR, Potapov A, Goldfarb D, Enemark JH (2012) Inorg Chem 51:1408–1418
Burgrnayer SJN, Stlefel EI (1985) J Chem Edu 62:943–953
Harlan EE, Berg JM, Holm RH (1986) J Am Chem Soc 108:6992–7000
Arnoux P, Sabaty M, Alric J, Frangioni B, Guigliarelli B, Adriano JM, Pignol D (2003) Nat Struct Biol 10:928–934
Jepson BJN, Mohan S, Clarke TA, Gates AJ, Cole JA, Butler CS, Butt JN, Hemmings AM, Richardson DJ (2007) J Biol Chem 282:6425–6437
Andersong GL, Williamsll J, Hille R (1992) J Biol Chem 267:23674–23682
Cammack R, Barber MJ, Bray RC (1976) Biochem J 157:469–478
Moura JJ, Xavier AV, Cammack R, Hall DO, Bruschi M, Le Gall J (1978) Biochem J 173:419–425
Weber K, Creager SE (1994) Anal Chem 66:3164–3172
Enemark JH, Garner CD (1997) J Biol Inorg Chem 2:817–822
Schröder I, Rech S, Krafft T, Macy JM (1997) J Biol Chem 272:23765–23768
Watts CA, Ridley H, Dridge EJ, Leaver JT, Reilly AJ, Richardson DJ, Butler CS (2005) Biochem Soc Trans 33:173–175
Spuches AM, Kruszyna HG, Rich AM, Wilcox DE (2005) Inorg Chem 44:2964–2972
Hoke KR, Cobb N, Armstrong FA, Hille R (2004) Biochemistry 43:1667–1674
Acknowledgments
We acknowledge Fundação para a Ciência e Tecnologia (FCT) for the financial support Granted to REQUIMTE (PEst-C/EQB/LA0006/2013) and UCIBIO-REQUIMTE (UID/Multi/04378/2013), BKM (SFRH/BPD/63066/2009) and SRP (FCT-ANR/BBB-MET0023/2012). NMR data were collected on 400-MHz spectrometer that is part of the National NMR Network, also supported by FCT (RECI/BBB-BQB/0230/2012).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Maiti, B.K., Maia, L.B., Silveira, C.M. et al. Incorporation of molybdenum in rubredoxin: models for mononuclear molybdenum enzymes. J Biol Inorg Chem 20, 821–829 (2015). https://doi.org/10.1007/s00775-015-1268-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-015-1268-0