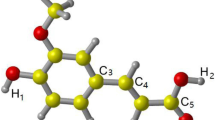
Abstract
Reaction enthalpies related to mechanisms of free radical scavenging activity of ellagic acid and its phenoxide anions were calculated by density functional theory and the semiempirical PM6 method. In addition to the gas phase, calculations are performed for water and benzene as the solvents, which may represent biological liquids and the membrane lipids, i.e., a natural environment for antiradical action. The thermodynamically favored mechanism depends on the polarity of reaction media, deprotonation degree of ellagic acid as well as the properties of scavenging radicals. The most acidic 3-OH group of ellagic acid is the active site for radical inactivation. The ellagate monoanions and dianions possess progressively better scavenging potency than unionized ellagic acid. The sequential proton loss electron transfer mechanism is the preferred reaction pathway for the ellagate monoanion and dianion in water. In benzene, ellagic acid inactivates free radicals by the hydrogen atom transfer mechanism. In the gas phase the latter mechanism is favored for all ellagic acid species.
Graphical Abstract

.




Similar content being viewed by others
References
Clifford MN, Scalbert A (2000) J Sci Food Agric 80:1118
Landete JM (2011) Food Res Int 44:1150
Larrosa M, Garcia-Conesa MT, Espin JC, Tomas-Barberan FA (2010) Mol Asp Med 31:513
Larrosa M, Tomas-Barberan FA, Espin JC (2006) J Nutr Biochem 17:611
Espin JC, Gonzalez-Barrio R, Cedra B, Lopez-Bote C, Rey AI, Tomas-Barberan FA (2007) J Agric Food Chem 55:10476
Quideau S, Deffieux D, Douat-Casassus C, Pouysegu L (2011) Angew Chem Int Ed 50:586
Vattem DA, Shetty K (2005) J Food Biochem 29:234
Kuo M-Y, Ou H-C, Lee W-J, Kuo W-W, Hwang L-L, Song T-Y, Huang C-Y, Chiu T-H, Tsai K-L, Tsai C-S, Sheu WH-H (2011) J Agric Food Chem 59:5100
Bala I, Bhardwaj V, Hariharan S, Ravi Kumar MNV (2006) J Pharm Biomed Anal 40:206
Ji H-F, Zhang H-Y, Shen L (2006) Bioorg Med Chem Lett 16:4095
Musialik M, Kuzmicz R, Pawlowski TS, Litwinienko G (2009) J Org Chem 74:2699
Priyadarsini KI, Khopde SM, Kumar SS, Mohan H (2002) J Agric Food Chem 50:2200
Hasegawa M, Terauchi M, Kikuchi Y, Nakao A, Okubo J, Yoshinaga T, Hiratsuka H, Kobayashi M, Hoshi T (2003) Monatsh Chem 134:811
Munoz-Munoz JL, Garcia-Molina F, Garcia-Molina M, Tudela J, Garcia-Canovas F, Rodriguez-Lopez JN (2009) IUBMB Life 61:171
Wright J, Johnson ER, DiLabio GA (2001) J Am Chem Soc 123:1173
Klein E, Lukeš V, Ilčin M (2007) Chem Phys 336:51
Litwinienko G, Ingold KU (2007) Acc Chem Res 40:222
Foti MC, Daquino C, Geraci C (2004) J Org Chem 69:2309
Zhang H-Y, Ji H-F (2006) New J Chem 30:503
Rimarčik J, Lukeš V, Klein E, Ilčin M (2010) J Mol Struct (Theochem) 952:25
Marković Z, Milenković D, Đorović J, Dimitrić Marković JM, Stepanić V, Lučić B, Amić D (2012) Food Chem 135:2070
Nenadis N, Tsimidou MZ (2012) Food Res Int 48:538
Leopoldini M, Russo N, Toscano M (2011) Food Chem 125:288
Marković Z, Milenković D, Đorović J, Dimitrić Marković JM, Stepanić V, Lučić B, Amić D (2012) Food Chem 134:1754
Frankel EN (2007) Antioxidants in food and biology: facts and fiction. chapter 3. The Oily Press, Bridgwater
Košinova P, Berka K, Wykes M, Otyepka M, Trouillas P (2012) J Phys Chem B 116:1309
Sato T, Kataoka M (1997) J Heterocyclic Chem 34:665
Huetz P, Mavaddat N, Mavri J (2005) J Chem Inf Model 45:1564
Kavuru P, Aboarayes D, Arora KK, Clarke HD, Kennedy A, Marshall L, Ong TT, Perman J, Pujari T, Wojtas L, Zaworotko MJ (2010) Cryst Growth Des 10:3568
Rossi M, Erlebacher J, Zacharias DE, Carrell HL, Iannucci B (1991) Carcinogenesis 12:2227
Mathieson AMcL, Poppleton BJ (1968) Acta Cryst B 24:1456
Clarke HD, Arora KK, Bass H, Kavuru P, Ong TT, Pujari T, Wojtas L, Zaworotko MJ (2010) Cryst Growth Des 10:2152
Zhang J, Xiong Y, Peng B, Gao H, Zhou Z (2011) Comp Theor Chem 963:148
Glendening ED, Badenhoop JK, Reed AE, Carpenter JE, Bohmann JA, Morales CM, Weinhold F (2009) NBO 5.9. Theoretical Chemistry Institute, University of Wisconsin, Madison
Weinhold F, Landis CR (2005) Valency and bonding: a natural bond orbital donor-acceptor perspective. Cambridge University Press, Cambridge
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Zakrzewski VG, Montgomery JAJr, Stratmann RE, Burant JC, Dapprich S, Millam JM, Daniels AD, Kudin KN, Strain MC, Farkas O, Tomasi J, Barone V, Cossi M, Cammi R, Mennucci B, Pomelli C, Adamo C, Clifford S, Ochterski J, Petersson GA, Ayala PY, Cui Q, Morokuma K, Malick AD, Rabuck KD, Raghavachari K, Foresman JB, Cioslowski J, Ortiz JV, Baboul AG, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Gomperts R, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Andres JL, Gonzalez C, Head-Gordon M, Replogle ES, Pople JA (2009) Gaussian 09, revision A.1-SMP. Gaussian Inc., Wallingford
MOPAC2009™ (2009) Stewart Computational Chemistry, Version 11.366W. http://openmopac.net/MOPAC2009.html
Jurd L (1959) J Am Chem Soc 81:4610
Hewitt DG, Nelson PF (1965) Holzforschung 19:97
Vaganek A, Rimarčik J, Lukeš V, Klein E (2012) Comp Theor Chem 991:192
Shahidi F, Janitha PK, Wanasundara PD (1992) Crit Rev Food Sci Nutr 32:67
Leopoldini M, Marino T, Russo N, Toscano M (2004) J Phys Chem A 108:4916
Trouillas P, Marsal P, Siri D, Lazzaroni R, Duroux J-L (2006) Food Chem 97:679
Lemanska K, Szymusiak H, Tyrakowska B, Zielinski R, Soffers AEMF, Rietjens IMCM (2001) Free Radic Biol Med 31:869
Shen L, Zhang H-Y, Ji H-F (2005) Org Lett 7:243
Kozlowski D, Trouillas P, Calliste C, Marsal P, Lazzaroni R, Duroux J-L (2007) J Phys Chem A 111:1138
Estevez L, Otero N, Mosquera RA (2010) J Phys Chem B 114:9706
Kondo O, Benson SW (1984) J Phys Chem 88:6675
Zhao Y, Schultz NE, Truhlar DG (2006) J Chem Theory Comput 2:364
Black G, Simmie JM (2010) J Comput Chem 31:1236
Galano A, Alvarez-Idaboy JR (2009) Org Lett 11:5114
Galano A, Macias-Ruvalcaba NA, Medina-Campos ON, Pedraza-Chaverri J (2010) J Phys Chem B 114:6625
Marković Z, Dimitrić Marković JM, Doličanin ĆB (2010) Theor Chem Acc 127:69
Zavala-Oseguera C, Alvarez-Idaboy JR, Merino G, Galano A (2009) J Phys Chem A 113:13913
Vega-Rodriguez A, Alvarez-Idaboy JR (2009) Phys Chem Chem Phys 11:7649
Marenich AV, Cramer CJ, Truhlar DG (2009) J Phys Chem B 113:6378
Namazian M, Lin CY, Coote ML (2010) J Chem Theory Comput 6:2721
Carpenter JE, Weinhold F (1988) J Mol Struct (Theochem) 169:41
Reed AE, Curtiss LA, Weinhold F (1988) Chem Rev 88:899
Stewart JJP (2009) J Mol Model 15:765
Acknowledgments
This study is cofinanced by the Ministry of Science of the Republic of Serbia (Project No. 172015 and Serbia-Croatia Bilateral Agreement 2011-2012) as well as by the Ministry of Science, Education, and Sports of the Republic of Croatia (Projects Nos. 079-0000000-3211, 098-1770495-2919, and Croatia-Serbia Bilateral Agreement 2011-2012).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Marković, Z., Milenković, D., Đorović, J. et al. A DFT and PM6 study of free radical scavenging activity of ellagic acid. Monatsh Chem 144, 803–812 (2013). https://doi.org/10.1007/s00706-013-0949-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-013-0949-z