Abstract
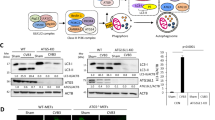
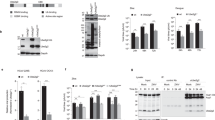
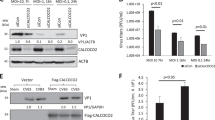
Autophagy is an intrinsic cellular process that can degrade cytoplasmic components. It has been reported that several pathogens hijack this process to facilitate their replication. Coxsackievirus B3 (CVB3), a member of the family Picornaviridae, induces autophagy upon infection. However, the details of CVB3-induced autophagy remain a subject of debate. This study applied a combination of multiple assays for the measurement of autophagy and demonstrated that CVB3 induces a complete autophagic flux. Experiments with infected HEK293A cells revealed that autophagosomes were induced upon CVB3 infection. Most of these autophagosomes were mCherry positive in mCherry-GFP-LC3 cells. Conversely, mCherry-positive autophagosomes were rescued to green positive when treated with the acidification inhibitors chloroquine (CQ) and bafilomycin A1 (BAF), suggesting that autophagosomes fused with late endosomes or lysosomes. The co-localization of LC3-positive puncta with lysosome-associated membrane protein 1 (LAMP1) or LysoTracker confirmed that the autophagosomes fused primarily with lysosomes. Interestingly, the disruption of autophagosome formation by 3-methyladenine (3-MA) or ATG5 siRNA treatment during viral infection significantly decreased CVB3 replication. However, inhibitors of lysosomal acidification, fusion, or degradation did not affect viral replication. Therefore, autolysosomes may not be critical for viral replication in vitro.




Similar content being viewed by others
References
Alirezaei M, Flynn CT, Wood MR, Whitton JL (2012) Pancreatic acinar cell-specific autophagy disruption reduces coxsackievirus replication and pathogenesis in vivo. Cell Host Microbe 11:298–305
Cornell CT, Kiosses WB, Harkins S, Whitton JL (2006) Inhibition of protein trafficking by coxsackievirus b3: multiple viral proteins target a single organelle. J Virol 80:6637–6647
Feldman AM, McNamara D (2000) Myocarditis. N Engl J Med 343:1388–1398
Kemball CC, Alirezaei M, Flynn CT, Wood MR, Harkins S, Kiosses WB, Whitton JL (2010) Coxsackievirus infection induces autophagy-like vesicles and megaphagosomes in pancreatic acinar cells in vivo. J Virol 84:12110–12124
Kimura S, Noda T, Yoshimori T (2007) Dissection of the autophagosome maturation process by a novel reporter protein, tandem fluorescent-tagged LC3. Autophagy 3:452–460
Kirkegaard K, Taylor MP, Jackson WT (2004) Cellular autophagy: surrender, avoidance and subversion by microorganisms. Nat Rev Microbiol 2:301–314
Klionsky DJ, Abdalla FC, Abeliovich H, Abraham RT, Acevedo-Arozena A, Adeli K, Zschocke J, Zuckerbraun B (2012) Guidelines for the use and interpretation of assays for monitoring autophagy. Autophagy 8:445–544
Krausslich HG, Nicklin MJ, Lee CK, Wimmer E (1988) Polyprotein processing in picornavirus replication. Biochimie 70:119–130
Kudchodkar SB, Levine B (2009) Viruses and autophagy. Rev Med Virol 19:359–378
Kyei GB, Dinkins C, Davis AS, Roberts E, Singh SB, Dong C, Wu L, Kominami E, Ueno T, Yamamoto A, Federico M, Panganiban A, Vergne I, Deretic V (2009) Autophagy pathway intersects with HIV-1 biosynthesis and regulates viral yields in macrophages. J Cell Biol 186:255–268
Lee YR, Hu HY, Kuo SH, Lei HY, Lin YS, Yeh TM, Liu CC, Liu HS (2013) Dengue virus infection induces autophagy: an in vivo study. J Biomed Sci 20:65
Lindberg AM, Stalhandske PO, Pettersson U (1987) Genome of coxsackievirus B3. Virology 156:50–63
Liu B, Fang M, Hu Y, Huang B, Li N, Chang C, Huang R, Xu X, Yang Z, Chen Z, Liu W (2014) Hepatitis B virus X protein inhibits autophagic degradation by impairing lysosomal maturation. Autophagy 10:416–430
Mizushima N, Komatsu M (2011) Autophagy: renovation of cells and tissues. Cell 147:728–741
Orvedahl A, Alexander D, Talloczy Z, Sun Q, Wei Y, Zhang W, Burns D, Leib DA, Levine B (2007) HSV-1 ICP34.5 confers neurovirulence by targeting the Beclin 1 autophagy protein. Cell Host Microbe 1:23–35
Palmenberg AC (1990) Proteolytic processing of picornaviral polyprotein. Annu Rev Microbiol 44:603–623
Pauschinger M, Phan MD, Doerner A, Kuehl U, Schwimmbeck PL, Poller W, Kandolf R, Schultheiss HP (1999) Enteroviral RNA replication in the myocardium of patients with left ventricular dysfunction and clinically suspected myocarditis. Circulation 99:889–895
Racaniello VR (2007) Picornaviridae: the virus and their replication. In: Knipe DM, Howley PM, Griffin DE, Lamb RA, Straus SE, Martin MA, Roizman B (eds) Fields virology, 5th edn. Lippincott, Williams & Wilkins, Philadelphia
Reed L, Muench H (1938) A simple method of estimating fifty per cent endpoint. Am J Hyg 27:493–497
Richards AL, Jackson WT (2012) Intracellular vesicle acidification promotes maturation of infectious poliovirus particles. PLoS Pathog 8:e1003046
Richards AL, Jackson WT (2013) How positive-strand RNA viruses benefit from autophagosome maturation. J Virol 87:9966–9972
Rubinsztein DC, Shpilka T, Elazar Z (2012) Mechanisms of autophagosome biogenesis. Curr Biol 22:R29–R34
Shi J, Wong J, Piesik P, Fung G, Zhang J, Jagdeo J, Li X, Jan E, Luo H (2013) Cleavage of sequestosome 1/p62 by an enteroviral protease results in disrupted selective autophagy and impaired NFKB signaling. Autophagy 9:1591–1603
Singh R, Cuervo AM (2012) Lipophagy: connecting autophagy and lipid metabolism. Int J Cell Biol 2012:282041
Sun MX, Huang L, Wang R, Yu YL, Li C, Li PP, Hu XC, Hao HP, Ishag HA, Mao X (2012) Porcine reproductive and respiratory syndrome virus induces autophagy to promote virus replication. Autophagy 8:1434–1447
Tabor-Godwin JM, Tsueng G, Sayen MR, Gottlieb RA, Feuer R (2012) The role of autophagy during coxsackievirus infection of neural progenitor and stem cells. Autophagy 8:938–953
Tam PE (2006) Coxsackievirus myocarditis: interplay between virus and host in the pathogenesis of heart disease. Viral Immunol 19:133–146
Tong L, Lin L, Zhao W, Wang B, Wu S, Liu H, Zhong X, Cui Y, Gu H, Zhang F, Zhong Z (2011) Destabilization of coxsackievirus b3 genome integrated with enhanced green fluorescent protein gene. Intervirology 54:268–275
Wileman T (2007) Aggresomes and pericentriolar sites of virus assembly: cellular defense or viral design? Annu Rev Microbiol 61:149–167
Wong J, Zhang J, Si X, Gao G, Mao I, McManus BM, Luo H (2008) Autophagosome supports coxsackievirus B3 replication in host cells. J Virol 82:9143–9153
Acknowledgments
The authors would like to thank Prof. William Jackson at Medical College of Wisconsin for providing the mCherry-GFP-LC3 plasmid. The authors also thank Prof. Zhaohua Zhong at Harbin Medical University for providing the CVB3-eGFP plasmid. The anti-CVB3-VP1 polyclonal antibody was kindly provided by Dr. Fei Deng at the Wuhan Institute of Virology, Chinese Academy of Science.
This work was supported by grants from Major State Basic Research Development Program of China (2013CB531502, 2013CB530501), the National Natural Science Foundation of China (31270977, 81101257, 31470848), the Scientific Research Foundation for Returned Overseas Chinese Scholars from State Education Ministry to C.D., and Jiangsu Provincial Innovative Research Team. The authors declare that there are no conflicting financial interests.
Author information
Authors and Affiliations
Corresponding authors
Additional information
X. Shi and Z. Chen contributed equally to the work.
Electronic supplementary material
Below is the link to the electronic supplementary material.

705_2016_2896_MOESM1_ESM.jpg
Supplementary data 1 HEK293A mCherry-GFP-LC3 stable cells starved with or without 50 nM CQ treatment. At 8 h post-starvation, the cells were observed under confocal microscopy. The bar represents 10 μm. (JPEG 2103 kb)
Rights and permissions
About this article
Cite this article
Shi, X., Chen, Z., Tang, S. et al. Coxsackievirus B3 infection induces autophagic flux, and autophagosomes are critical for efficient viral replication. Arch Virol 161, 2197–2205 (2016). https://doi.org/10.1007/s00705-016-2896-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00705-016-2896-6