Abstract
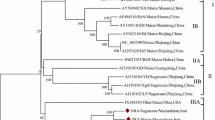
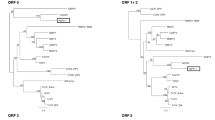
A novel curtovirus, spinach severe curly top virus (SSCTV), was associated with symptomatic spinach plants collected from a commercial field in south-central Arizona during 2009. In addition, a second viral molecule of about 2.9 kb from the same spinach plants was amplified, cloned and sequenced. The latter isolate, herein named spinach curly top Arizona virus (SCTAV), was found to share 77 % pairwise sequence identity with beet curly top Iran virus (BCTIV), a leafhopper-transmitted geminivirus that has been assigned to the new genus Becurtovirus. The SCTAV genome encodes three viral-sense genes, V1, V2, and V3, and two complementary-sense genes, C1 and C2. There was no evidence for the presence of either a C3 or C4 ORF in the genome sequence. The genome organization of SCTAV is not like that of New World curtoviruses but instead is similar to that of BCTIV, which, to date, is only known to be present in Iran. Consistent with this observation, SCTAV and BCTIV both contain the unusual nonanucleotide TAAGATT/CC and a replication-associated protein, Rep (or C1), that is more closely related to the mastrevirus Rep than to those of curtoviruses reported to date. Both SSCTV and SCTAV were found to have a recombinant genome containing sequences (AY548948) derived from ancestral SCTV sequences in the virion-sense portions of the genome. Agroinoculation of Nicotiana benthamiana (Domin) plants with the cloned genome of SCTAV resulted in infection of 95 % of the plants and the development of severe curling symptoms, whereas only 20 % of the SSCTV-inoculated plants were infected, developing only mild curling symptoms. When plants were co-inoculated with both viruses, the frequency of infection remained higher for SCTAV than for SSCTV (80 % vs. 20 %), indicating no evidence of synergistic effects between the two viruses with respect to efficiency of infection.





Similar content being viewed by others
References
Fauquet CM, Mayo MA, Maniloff J, Desselberger U, Ball LA (2011) Family Geminiviridae. In: Virus taxonomy: classification and nomenclature of viruses: Ninth Report of the International Committee on Taxonomy of Viruses. Elsevier, Inc., San Diego, pp 301–326
Soto MJ, Chen LF, Seo YS, Gilbertson RL (2005) Identification of regions of the Beet mild curly top virus (family Geminiviridae) capsid protein involved in systemic infection, virion formation and leafhopper transmission. Virology 341:257–270
Bennett CW (1971) The curly top disease of sugarbeet and other plants. Monograph 7. The American Phytopathological Society, St. Paul
Stanley J, Markham PG, Callis RJ, Pinner MS (1986) The nucleotide sequence of an infectious clone of the geminivirus beet curly top virus. EMBO J 5:1761–1767
Stenger DC (1994) Complete nucleotide sequence of the hypervirulent CFH strain of beet curly top virus. Mol Plant Microbe Interact 7:154–157
Stenger DC (1995) Genotypic variability and the occurrence of less than genome-length viral DNA forms in a field population of beet curly top geminivirus. Phytopathology 85:1316–1322
Stenger DC (1998) Replication specificity elements of the Worland strain of beet curly top virus are compatible with those of the CFH strain but not those Cal/Logan strain. Phytopathology 88:1174–1178
Stenger DC, Carbonaro D, Duffus JE (1990) Genomic characterization of phenotypic variants of beet curly top virus. J Gen Virol 71:2211–2215
Briddon RW, Stenger DC, Bedford ID, Stanley J, Izadpanah K, Markham PG (1998) Comparison of a beet curly top virus isolate originating from the old world with those from the New World. E J Plant Pathol 104:77–84
Chen LF, Vivoda E, Gilbertson RL (2011) Genetic diversity in curtoviruses: a highly divergent strain of Beet mild curly top virus associated with an outbreak of curly top disease in pepper in Mexico. Arch Virol 156:547–555
Klute KA, Nadler SA, Stenger DC (1996) Horseradish curly top virus is a distinct subgroup II geminivirus species with rep and C4 genes derived from a subgroup III ancestor. J Gen Virol 77:1369–1378
Lam N, Creamer R, Rascon J, Belfon R (2009) Characterization of a new curtovirus, Pepper yellow dwarf virus, from chile pepper and distribution in weed hosts in New Mexico. Arch Virol 154:429–436
Baliji S, Black MC, French R, Stenger DC, Sunter G (2004) Spinach curly top virus: a newly described curtovirus species from southwest Texas with incongruent gene phylogenies. Phytopathology 94:772–779
Heydarnejad J, Keyvani N, Razavinejad S, Massumi H, Varsani A (2012) Fulfilling Koch′s postulates for beet curly top Iran virus and proposal for consideration of new genus in the family Geminiviridae. Arch Virol. doi:10.1007/s00705-012-1485-6
Yazdi HR, Heydarnejad J, Massumi H (2008) Genome characterization and genetic diversity of beet curly top Iran virus: a geminivirus with a novel nonanucleotide. Virus Gen 36:539–545
Briddon RW, Heydarnejad J, Khosrowfar F, Massumi H, Martin DP, Varsani A (2010) Turnip curly top virus, a highly divergent geminivirus infecting turnip in Iran. Virus Res 152:169–175
Creamer R, Hubble H, Lewis A (2005) Curtovirus infection of chile pepper in New Mexico. Plant Dis 89:480–486
Strausbaugh CA, Wintermantel WM, Gillen AM, Eujayl IA (2008) Curly top survey in the Western United States. Phytopathology 98:1212–1217
Stenger DC, McMahon CL (1997) Genotypic diversity of beet curly top virus populations in the western United States. Phytopathology 87:737–744
Correll JC, Morelock TE, Black MC, Koike ST, Brandenberger LP, Daniello FJ (1994) Economically important diseases of spinach. Plant Dis 78:653–660
Nischwithz C, Olsen MW (2010) First report of two curtoviruses in spinach and common beet in Arizona. Plant Health Progress. doi:10.1094/PHP-2010-0216-02-BR
Hernández-Zepeda C, Brown JK (2010) First report of a new curtovirus species, Spinach severe curly top virus, in commercial spinach plants (Spinacia oleracea) from south-central Arizona. Plant Dis 94:917
Haible D, Kober S, Jeske H (2006) Rolling circle amplification revolutionizes diagnosis and genomics of geminiviruses. J Virol Meth 135:9–16
Doyle JJ, Doyle JL (1990) A rapid total DNA preparation procedure for fresh plant tissue. Focus 12:13–15
Inoue-Nagata AK, Albuquerque LC, Rocha WB, Nagata T (2004) A simple method for cloning the complete begomovirus genome using the bacteriophage phi29 DNA polymerase. J Virol Methods 116:209–211
Oliveira-Ferreira PT, Oliveira-Lemos T, Nagata T, Inoue-Nagata AK (2008) One-step cloning approach for construction of agroinfectious begomovirus clones. J Virol Methods 147:351–354
Hellens RP, Edwards EA, Leyland NR, Bean S, Mullineaux PM (2000) pGreen: a versatile and flexible binary Ti vector for Agrobacterium-mediated plant transformation. Plant Mol Biol 42:819–832
Edgar RC (2004) MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res 32:1792–1797
Guindon S, Dufayard JF, Lefort V, Anisimova M, Hordijk W, Gascuel O (2010) New algorithms and methods to estimate maximum-likelihood phylogenies: assessing the performance of PhyML 3.0. Syst Biol 59(3):307–321
Martin DP, Lemey P, Lott M, Moulton V, Posada D, Lefeuvre P (2010) RDP3: a flexible and fast computer program for analyzing recombination. Bioinformatics 26:2462–2463
Muhire B, Martin DP, Brown JK, Navas-Castillo J, Moriones E, Zerbini FM, Rivera-Bustamante R, Malathi VG, Briddon RW, Varsani A (2013) A genome-wide pairwise-identity-based proposal for the classification of viruses in the genus Mastrevirus (Geminiviridae). Arch Virol. doi:10.1007/s00705-012-1601-7
Anisimova M, Gascuel O (2006) Approximate likelihood-ratio test for branches: a fast, accurate, and powerful alternative. Syst Biol 55:539–552
Arguello-Astorga GR, Ruíz-Medrano R (2001) An iteron-related domain is associated to Motif 1 in the replication proteins of geminiviruses: identification of potential interacting amino acid-base pairs by a comparative approach. Arch Virol 146:1465–1485
Andrade EC, Manhani GG, Alfenas PF, Calegario RF, Fontes EPB, Zerbini MF (2006) Tomato yellow spot virus, a tomato-infecting begomovirus from Brazil with a closer relationship to viruses from Sida sp., forms pseudorecombinants with begomoviruses from tomato but not from Sida. J Gen Virol 87:3687–3696
Briddon RW, Pinner MS, Stanley J, Markham PG (1990) Geminivirus coat protein gene replacement alters insect specificity. Virology 177:85–94
Fauquet CM, Briddon RW, Brown JK, Moriones E, Stanley J, Zerbini M, Zhou X (2008) Geminivirus strain demarcation and nomenclature. Arch Virol 153:783–821
Settlage S, See RG, Hanley-Bowdoin L (2005) Geminivirus C3 protein: replication enhancement and protein interactions. J Virol 79:9885–9895
Fondong VN, Pita JS, Rey MEC, Kochko A, Beachy RN, Fauquet CM (2000) Evidence of synergism between African cassava mosaic virus and a new double recombinant geminivirus infecting cassava in Cameroon. J Gen Virol 81:287–297
Rentería-Canett I, Xoconostle-Cázares B, Ruíz-Medrano R, Rivera-Bustamante RF (2011) Geminivirus mixed infection on pepper plants: synergistic interaction between PHYVV and PepGMV. Virol J 8:104–116
Méndez-Lozano J, Torres-Pacheco I, Fauquet CM, Rivera-Bustamante RF (2003) Interactions between geminiviruses in a naturally occurring mixture: Pepper huasteco virus and Pepper golden mosaic virus. Phytopathology 93:270–277
Acknowledgments
This study was supported by an Arizona Specialty Crops Block Grant (Grant No. SCBGP FB09-07).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hernández-Zepeda, C., Varsani, A. & Brown, J.K. Intergeneric recombination between a new, spinach-infecting curtovirus and a new geminivirus belonging to the genus Becurtovirus: first New World exemplar. Arch Virol 158, 2245–2254 (2013). https://doi.org/10.1007/s00705-013-1733-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00705-013-1733-4