Abstract
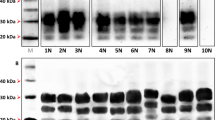
Prion diseases are characterized by a long incubation period. In scrapie, sheep may incubate and spread the infection for several years before clinical signs evolve. We have previously studied the occurrence of subclincal infection in the brain. Now, we have studied the occurrence of subclinical infection in the brain and several lymphoid tissues in two scrapie-affected Icelandic sheep flocks by immunohistochemistry for PrPSc, a molecular marker for infectivity, and correlated this with results of PrP genotyping. At culling, one flock had one confirmed scrapie case, while the other flock had two. Analysis of 106 asymptomatic sheep by immunostaining for PrPSc revealed that the incidence of subclinical infection was 58.3% in one flock and 42.5% in the other. PrPSc was only detected in lymphoid tissues. The youngest positive sheep were 4 months old. PrP genotyping showed that over 90% of the sheep were of a genotype which is moderately sensitive to infection and may delay neuroinvasion. Our results show that asymptomatic sheep may spread the infection during the long incubation period of several years, which constitutes an important obstacle in the eradication of scrapie. Our findings indicate that contamination of the environment plays an important part in sustaining the infection.


Similar content being viewed by others
References
Andréoletti O, Berthon P, Marc D, Sarradin P, Grosclaude J, van Keulen L, Schelcher F, Elsen J-M, Lantier F (2000) Early accumulation of PrPSc in gut-associated lymphoid and nervous tissue of susceptible sheep from a Romanov flock with natural scrapie. J Gen Virol 81:3115–3126
Andréoletti O, Lacroux C, Chabert A, Monnereau L, Tabouret G, Lantier F, Berthon P, Eychenne F, Lafond-Benestad S, Elsen J-M, Schelcher F (2002) PrPSc accumulation in placentas of ewes exposed to natural scrapie: influence of foetal PrP genotype and effect on ewe-to-lamb transmission. J Gen Virol 83:2607–2616
Belt PBGM, Muileman IH, Schreuder BEC, Bos-de Ruiter J, Gielkens ALJ, Smits MA (1995) Identification of five allelic variants of the sheep PrP gene and their association with natural scrapie. J Gen Virol 76:509–517
Bolton DC, McKinley MP, Prusiner SB (1982) Identification of a protein that purifies with the scrapie prion. Science 218:1309–1311
Brown KL, Stewart K, Ritchie DL, Mabbott NA, Williams A, Fraser H, Morrison WI, Bruce ME (1999) Scrapie replication in lymphoid tissues depends on prion protein-expressing follicular dendritic cells. Nat Med 5:1308–1312
Bruce ME, Brown KL, Mabbott NA, Farquhar CF, Jeffrey M (2000) Follicular dendritic cells in TSE pathogenesis. Immunol Today 21:442–446
Dawson G, Hoinville LJ, Hosie BD, Hunter N (1998) Guidance on the use of PrP genotyping as an aid to the control of clinical scrapie. Vet Rec 142:623–5
Elsen J-M, Amigues Y, Schelcher F, Ducrocq V, Andreoletti O, Eychenne F, Tien Khang JV, Poivey J-P, Lantier F, Laplance J-L (1999) Genetic susceptibility and transmission factors in scrapie:detailed analysis of an epidemic in a closed flock of Romanov. Arch Virol 144:431–445
Ersdal C, Ulvund MJ, Benestad SL, Tranulis MA (2003) Accumulation of pathogenic prion protein (PrPSc) in nervous and lymphoid tissues of sheep with subclinical scrapie. Vet Pathol 40:164–174
Foster J, McKenzie C, Parnham D, Drummond D, Goldmann W, Stevenson E, Hunter N (2006) Derivation of a scrapie-free sheep flock from the progeny of a flock affected by scrapie. Vet Rec 159:42–45
Fraser H, Dickinson AG (1978) Studies of the lymphoreticular system in the pathogenesis of scrapie: the role of spleen and thymus. J Comp Path 88:563–573
Georgsson G, Sigurdarson S, Brown P (2006) Infectious agent of sheep scrapie may persists in the environment for at least 16 years. J Gen Virol 87:3737–3740
González L, Jeffrey M, Sisó S, Martin S, Bellworthy SJ, Stack MJ, Chaplin MJ, Davis L, Dagleish MP, Reid HW (2005) Diagnosis of preclinical scrapie in samples of rectal mucosa. Vet Rec 156:846–847
Hadlow WJ, Kennedy RC, Race RE (1982) Natural infection of Suffolk sheep with scrapie virus. J Infect Dis 146:657–664
Heggebö R, Press CMcL,Gunnes G, González L, Jeffrey M (2002) Distribution and accumulation of PrP in gut-associated and peripheral lymphoid tissue of scrapie-affected Suffolk sheep. J Gen Virol 83:479–489
Heggebö R, Press CMcL, Gunnes G, Lie KI, Tranulis MA, Ulvund M, Groschup MH, Landsverk T (2000) Distribution of prion protein in the ileal Peyer’s patch of scrapie-free lambs and lambs naturally and experimentally exposed to the scrapie agent. J Gen Virol 81:2327–2337
Hourrigan J, Klinsporn A, Clark WW, de Camp M (1979) Epidemiology of scrapie in the United States. In: Prusiner SB, Hadlow WJ (eds) Slow transmissible diseases of the nervous system. vol 1. Academic Press, New York, pp 331–356
Huang F-P, Farquhar CF, Mabbott NA, Bruce ME, MacPherson GG (2002) Migrating intestinal dendritic cells transport PrPSc from the gut. J Gen Virol 83:267–271
Ikegami Y, Ito M, Isomura H, Momotani E, Sasaki K, Muramatsu Y, Ishiguro N, Shinagawa M (1991) Pre-clinical and clinical diagnosis of scrapie by detection of PrP protein in tissues of sheep. Vet Rec 128:271–275
Jeffrey M, McGovern G, Goodsir CM, Brown KL, Bruce ME (2000) Sites of prion protein accumulation in scrapie-infected mouse spleen revealed by immuno-electron microscopy. J Pathol 191:323–332
Johnson CJ, Phillips KE, Schramm PT, McKenzie D, Aiken JM, Pedersen JA (2006) Prions adhere to soil minerals and remain infectious. PloS Pathogens 2:1–7
Kimberlin RH, Walker CA (1968) Pathogenesis of scrapie (strain 263K) in hamsters infected intracerebrally, intraperiotoneally or intraocularly. J Gen Virol 67:255–263
Lasmézas CI, Cesbron J-Y, Deslys J-P, Demaimay R, Adjou KT, Rioux R, Lemaire C, Locht C, Dormont D (1996) Immune system-dependent and- independent replication of the scrapie agent. J Virol 70:1292–1295
Ligios C, Sigurdson CJ, Santucciu C, Carcassola G, Manco G, Basagni M, Maestrale C, Cancedda MG, Madau L, Aguzzi A (2005) PrPSc in mammary glands of sheep affected by scrapie and mastitis. Nat Med 11:1137–1138
Ligios C, Cancedda MG, Madau L, Santucciu C, Maestrale C, Agrimi U, Ru G, Di Guardo G (2006) PrPScdeposition in nervous tissues without lymphoid tissue involvement is frequently found in ARQ/ARQ Sarda sheep preclinically affected with natural scrapie. Arch Virol 151:2007–2020
Mabbott NA, Farquhar CF, Brown KL, Bruce ME (1998) Involvement of the immune system in TSE pathogenesis. Immunol Today 5:201–203
Mabbott NA, Mackay F, Minns F, Bruce ME (2000) Temporary inactivation of follicular dendritic cells delays neuroinvasion of scrapie. Nat Med 6:719–720
Mabbott NA, MacPherson GG (2006) Prions and their lethal journey to the brain. Nat Rev Microbiol 4:201–211
McBride PA, Eikelenboom P, Kraal G, Fraser H, Bruce ME (1992) PrP protein is associated with follicular dendritic cells of spleens and lymph nodes in uninfected and scrapie-infected mice. J Pathol 168:413–418
McGovern G, Brown KL, Bruce ME, Jeffrey M (2004) Murine scrapie infection causes an abnormal germinal centre reaction in the spleen. J Comp Path 130:181–194
Miller MW, Williams ES, Thompson Hobbs N, Wolfe LL (2004) Environmental sources of prion transmission in mule deer. Emerg Infect Dis 10:1003–1006
Montrasio F, Frigg R, Glatzel M, Klein MA, Mackay F, Aguzzi A, Weissmann C (2000) Impaired prion replication in spleens of mice lacking functional follicular dendritic cells. Science 288:1257–1259
Muramatsu Y, Onodera A, Horiuchi M, Ishiguro N, Shinagawa M (1993) Detection of PrPSc in sheep at the preclinical stage of scrapie and its siginficance for diagnosis of insidious infection. Arch Virol 134:427–432
O’Rourke KI, Baszler TV, Miller JM, Spraker TR, Sadler-Riggleman I, Knowles DP (1998) Monoclonal antibody F89/160.1.5 defines a conserved epitope on the ruminant prion protein. J Clin Microbiol 36:1750–1755
Palsson PA (1979) Rida (scrapie) in Iceland and its epidemiology. In: Prusiner SB, Hadlow WJ (eds) Slow transmissible diseases of the nervous system. vol 1. Academic Press, New York, pp 357–366
Prusiner SB, Bolton DC, Groth DF, Bowman A, Cochran SP, McKinley MP (1982) Further purification and characterization of scrapie prions. Biochemistry 21:6942–6950
Race R, Jenny A, Sutton D (1998) Scrapie infectivity and proteinase K- resistant prion protein in sheep placenta, brain, spleen, and lymph node: implications for transmission and antemortem diagnosis. J Infect Dis 178:949–953
Roels S, Vanopdenbosch E, Langeveld JPM, Schreuder BEC (1999) Immunohistochemical evaluation of tonsillar tissue for preclinical screening of scrapie based on surveillance in Belgium. Vet Rec 145:524–525
Roels S, Renard C, De Bosschere H, Geeroms R, Van Poucke M, Peelman L, Vanopdenbosch E (2004) Detection of polymorphisms in the prion protein gene in the Belgian sheep population: Some preliminary data. Vet Q 26:3–11
Schreuder BEC, van Keulen LJM, Vromans MEW, Langeveld JPM, Smits MA (1998) Tonsillar biopsy and PrPSc detection in the preclinical diagnosis of scrapie. Vet Rec 142:564–568
Seeger H, Heikenwalder M, Zeller N, Kranich J, Schwarz P, Gaspert A, Seifert B, Miele G, Aguzzi A (2005) Coincident scrapie infection and nephritis lead to urinary prion excretion. Science 310:324–326
Skarphédinsson S, Jóhannsdóttir R, Gudmundsson P, Sigurdarson S, Georgsson G (1994) PrPSc in Icelandic sheep naturally infected with scrapie. Ann N Y Acad Sci 724:304–309
Somerville RA, Birkett CR, Farquhar CF, Hunter N, Goldmann W, Dornan J, Grover D, Hennion RM, Percy C, Foster J, Jeffrey M (1997) Immunodetection of PrPSc in spleens of some scrapie-affected sheep but not BSE-infected cows. J GenVirol 78:2389–2396
Thorgeirsdottir S, Sigurdarson S, Thorisson HM, Georgsson G, Palsdottir A (1999) PrP gene polymorphism and natural scrapie in Icelandic sheep. J Gen Virol 80:2527–2534
Thorgeirsdottir S, Georgsson G, Reynisson E, Sigurdarson S, Palsdottir A (2002) Search for healthy carriers of scrapie: an assessment of subclinical infection of sheep in an Icelandic scrapie flock by three diagnostic methods and correlation with genotypes. Arch Virol 147:709–722
van Keulen LJM, Schreuder BEC, Meloen RH, Mooij- Harkes G, Vromans MEW, Langeveld JPM (1996) Immunohistochemical detection of prion protein in lymphoid tissues of sheep with natural scrapie. J Clin Microbiol 34:1228–1231
van Keulen LJM, Schreuder BEC, Vromans MEW, Langeweld JPM, Smits MA (1999) Scrapie-associated prion protein in the gastro-intestinal tract of sheep with natural scrapie. J Comp Path 121:55–63
Vascellari M, Nonno R, Mutinelli F, Bigolaro M, Di Bari MA, Melchiotti E, Marcon S, D’Agostino C, Vaccari G, Conte M, De Grossi L, Rosone F, Giordani F, Agrimi U (2007) PrPScin salivary glands of scrapie-affected sheep. J Virol 81:4872–4876
Acknowledgments
We are indebted to Margret Jonsdottir, Steinunn Arnadottir MTA’s, Steinunn Johannsdóttir BSc for skilful technical help Hallgrimur Arnarson BS for statistical analysis and preparation of figures, Kristin Sveinsdottir librarian and Halldor Thorleifur Olafsson farmer, owner of flock A. The work was supported by grants from the Icelandic Research Fund and the University of Iceland Research Fund and the EU-project FAIR CT 987021.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Georgsson, G., Adolfsdottir, J.A., Palsdottir, A. et al. High incidence of subclinical infection of lymphoid tissues in scrapie-affected sheep flocks. Arch Virol 153, 637–644 (2008). https://doi.org/10.1007/s00705-008-0035-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00705-008-0035-8