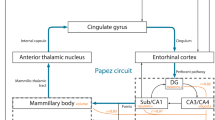
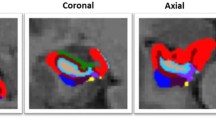
Abstract
Hippocampus volumes have been shown to be decreased in patients with major depression, but volume measurements are inconsistent in patients with bipolar disorder. Both disorders are associated with deficits in hippocampus-mediated cognitive functions. However, the underlying pathophysiology is widely unknown. In this post-mortem study, we used design-based stereology on Nissl-stained serial sections to investigate the number of neurons, oligodendrocytes and astrocytes in substructures of the posterior hippocampus in eight patients with major depression, eight patients with bipolar disorder and ten control patients without a neuropsychiatric disorder. Compared to controls, patients with bipolar disorder had significantly more neurons in the cornu ammonis subfield 1 (CA1) and the subiculum, while the number of oligodendrocytes was higher only in CA1. In patients with major depression, the density of oligodendrocytes was higher in CA2/3, CA4 and the subiculum. The dose of antidepressants correlated with the density and number of oligodendrocytes in CA2/3, indicating that antidepressants may affect our results. Treatment with neuroleptics expressed in chlorpromazine equivalents and benzodiazepines expressed in diazepam equivalents correlated negatively with the number of oligodendrocytes in CA2/3 and CA4, respectively, suggesting that treatment with these drugs do not influence cell number. We did not detect alterations in either volumes of substructures or numbers of astrocytes. Increased cell numbers argue for a denser packing of neurons and oligodendrocytes as a result of a decreased neuropils. This neuropathological process may be based on neurodevelopmental disturbances and may contribute to altered microconnectivity and cognitive deficits in affective disorders.







Similar content being viewed by others
References
Ali IM (1998) Long-term treatment with antidepressants in primary care: are sub-therapeutic doses still being used? Psychiatr Bull 22:15–19
Altshuler LL, Bartzokis G, Grieder T, Curran J, Jimenez T, Leight K, Wilkins J, Gerner R, Mintz J (2000) An MRI study of temporal lobe structures in men with bipolar disorder or schizophrenia. Biol Psychiatry 48(2):147–162
Arnone D, Cavanagh J, Gerber D, Lawrie SM, Ebmeier KP, McIntosh AM (2009) Magnetic resonance imaging studies in bipolar disorder and schizophrenia: meta-analysis. Br J Psychiatry 195(3):194–201. doi:10.1192/bjp.bp.108.059717
Arnone D, McIntosh AM, Ebmeier KP, Munafo MR, Anderson IM (2012) Magnetic resonance imaging studies in unipolar depression: systematic review and meta-regression analyses. Eur Neuropsychopharmacol 22(1):1–16. doi:10.1016/j.euroneuro.2011.05.003
Beyer JL, Kuchibhatla M, Payne ME, Moo-Young M, Cassidy F, Macfall J, Krishnan KR (2004) Hippocampal volume measurement in older adults with bipolar disorder. Am J Geriatr Psychiatry Off J Am Assoc Geriatr Psychiatry 12(6):613–620. doi:10.1176/appi.ajgp.12.6.613
Bielau H, Trubner K, Krell D, Agelink MW, Bernstein HG, Stauch R, Mawrin C, Danos P, Gerhard L, Bogerts B, Baumann B (2005) Volume deficits of subcortical nuclei in mood disorders A postmortem study. Eur Arch Psychiatry Clin Neurosci 255(6):401–412. doi:10.1007/s00406-005-0581-y
Bogerts B, Falkai P, Haupts M, Greve B, Ernst S, Tapernon-Franz U, Heinzmann U (1990) Post-mortem volume measurements of limbic system and basal ganglia structures in chronic schizophrenics. Initial results from a new brain collection. Schizophr Res 3(5–6):295–301
Bora E, Yucel M, Pantelis C (2010) Cognitive impairment in schizophrenia and affective psychoses: implications for DSM-V criteria and beyond. Schizophr Bull 36(1):36–42. doi:10.1093/schbul/sbp094
Canetta SE, Bao Y, Co MD, Ennis FA, Cruz J, Terajima M, Shen L, Kellendonk C, Schaefer CA, Brown AS (2014) Serological documentation of maternal influenza exposure and bipolar disorder in adult offspring. Am J Psychiatry 171(5):557–563. doi:10.1176/appi.ajp.2013.13070943
Cole J, Toga AW, Hojatkashani C, Thompson P, Costafreda SG, Cleare AJ, Williams SC, Bullmore ET, Scott JL, Mitterschiffthaler MT, Walsh ND, Donaldson C, Mirza M, Marquand A, Nosarti C, McGuffin P, Fu CH (2010) Subregional hippocampal deformations in major depressive disorder. J Affect Disord 126(1–2):272–277. doi:10.1016/j.jad.2010.03.004
Doring TM, Kubo TT, Cruz LC Jr, Juruena MF, Fainberg J, Domingues RC, Gasparetto EL (2011) Evaluation of hippocampal volume based on MR imaging in patients with bipolar affective disorder applying manual and automatic segmentation techniques. J Magn Reson Imaging JMRI 33(3):565–572. doi:10.1002/jmri.22473
Ellison-Wright I, Bullmore E (2010) Anatomy of bipolar disorder and schizophrenia: a meta-analysis. Schizophr Res 117(1):1–12. doi:10.1016/j.schres.2009.12.022
Elsayed M, Banasr M, Duric V, Fournier NM, Licznerski P, Duman RS (2012) Antidepressant effects of fibroblast growth factor-2 in behavioral and cellular models of depression. Biol Psychiatry 72(4):258–265. doi:10.1016/j.biopsych.2012.03.003
Fanselow MS, Dong HW (2010) Are the dorsal and ventral hippocampus functionally distinct structures? Neuron 65(1):7–19. doi:10.1016/j.neuron.2009.11.031
Gundersen HJ, Bagger P, Bendtsen TF, Evans SM, Korbo L, Marcussen N, Moller A, Nielsen K, Nyengaard JR, Pakkenberg B et al (1988) The new stereological tools: disector, fractionator, nucleator and point sampled intercepts and their use in pathological research and diagnosis. APMIS : acta pathologica, microbiologica, et immunologica Scandinavica 96(10):857–881
Hallahan B, Newell J, Soares JC, Brambilla P, Strakowski SM, Fleck DE, Kieseppa T, Altshuler LL, Fornito A, Malhi GS, McIntosh AM, Yurgelun-Todd DA, Labar KS, Sharma V, MacQueen GM, Murray RM, McDonald C (2011) Structural magnetic resonance imaging in bipolar disorder: an international collaborative mega-analysis of individual adult patient data. Biol Psychiatry 69(4):326–335. doi:10.1016/j.biopsych.2010.08.029
Hamilton JP, Gotlib IH (2008) Neural substrates of increased memory sensitivity for negative stimuli in major depression. Biol Psychiatry 63(12):1155–1162. doi:10.1016/j.biopsych.2007.12.015
Haukvik UK, McNeil T, Lange EH, Melle I, Dale AM, Andreassen OA, Agartz I (2014) Pre- and perinatal hypoxia associated with hippocampus/amygdala volume in bipolar disorder. Psychol Med 44(5):975–985. doi:10.1017/S0033291713001529
Hof PR, Haroutunian V, Friedrich VL Jr, Byne W, Buitron C, Perl DP, Davis KL (2003) Loss and altered spatial distribution of oligodendrocytes in the superior frontal gyrus in schizophrenia. Biol Psychiatry 53(12):1075–1085
Hsiao YL, Wu YS, Wu JY, Hsu MH, Chen HC, Lee SY, Lee IH, Yeh TL, Yang YK, Ko HC, Lu RB (2009) Neuropsychological functions in patients with bipolar I and bipolar II disorder. Bipolar Disord 11(5):547–554. doi:10.1111/j.1399-5618.2009.00723.x
Kempton MJ, Geddes JR, Ettinger U, Williams SC, Grasby PM (2008) Meta-analysis, database, and meta-regression of 98 structural imaging studies in bipolar disorder. Arch Gen Psychiatry 65(9):1017–1032. doi:10.1001/archpsyc.65.9.1017
Kempton MJ, Salvador Z, Munafo MR, Geddes JR, Simmons A, Frangou S, Williams SC (2011) Structural neuroimaging studies in major depressive disorder. Meta-analysis and comparison with bipolar disorder. Arch Gen Psychiatry 68(7):675–690. doi:10.1001/archgenpsychiatry.2011.60
Knable MB, Barci BM, Webster MJ, Meador-Woodruff J, Torrey EF (2004) Molecular abnormalities of the hippocampus in severe psychiatric illness: postmortem findings from the Stanley Neuropathology Consortium. Mol Psychiatry 9(6):609–620, 544. doi:10.1038/sj.mp.4001471
Koolschijn PC, van Haren NE, Lensvelt-Mulders GJ, Hulshoff Pol HE, Kahn RS (2009) Brain volume abnormalities in major depressive disorder: a meta-analysis of magnetic resonance imaging studies. Hum Brain Mapp 30(11):3719–3735. doi:10.1002/hbm.20801
Lavenex P (2012) Functional Anatomy, Development, and Pathology of the Hippocampus. In: Bartsch T (ed) The Clinical Neurobiology of the Hippocampus—an integrative view. Oxford University Press, Cambridge
Liao Y, Huang X, Wu Q, Yang C, Kuang W, Du M, Lui S, Yue Q, Chan RC, Kemp GJ, Gong Q (2013) Is depression a disconnection syndrome? Meta-analysis of diffusion tensor imaging studies in patients with MDD. J Psychiatry Neurosci 38(1):49–56. doi:10.1503/jpn.110180
McDonald C, Zanelli J, Rabe-Hesketh S, Ellison-Wright I, Sham P, Kalidindi S, Murray RM, Kennedy N (2004) Meta-analysis of magnetic resonance imaging brain morphometry studies in bipolar disorder. Biol Psychiatry 56(6):411–417. doi:10.1016/j.biopsych.2004.06.021
McDonald C, Marshall N, Sham PC, Bullmore ET, Schulze K, Chapple B, Bramon E, Filbey F, Quraishi S, Walshe M, Murray RM (2006) Regional brain morphometry in patients with schizophrenia or bipolar disorder and their unaffected relatives. Am J Psychiatry 163(3):478–487. doi:10.1176/appi.ajp.163.3.478
McEwen BS, Magarinos AM (2001) Stress and hippocampal plasticity: implications for the pathophysiology of affective disorders. Hum Psychopharmacol 16(S1):S7–S19. doi:10.1002/hup.266
Mosebach J, Keilhoff G, Gos T, Schiltz K, Schoeneck L, Dobrowolny H, Mawrin C, Muller S, Schroeter ML, Bernstein HG, Bogerts B, Steiner J (2013) Increased nuclear Olig1-expression in the pregenual anterior cingulate white matter of patients with major depression: a regenerative attempt to compensate oligodendrocyte loss? J Psychiatr Res 47(8):1069–1079. doi:10.1016/j.jpsychires.2013.03.018
Price JL, Ko AI, Wade MJ, Tsou SK, McKeel DW, Morris JC (2001) Neuron number in the entorhinal cortex and CA1 in preclinical Alzheimer disease. Arch Neurol 58(9):1395–1402
Rosoklija G, Toomayan G, Ellis SP, Keilp J, Mann JJ, Latov N, Hays AP, Dwork AJ (2000) Structural abnormalities of subicular dendrites in subjects with schizophrenia and mood disorders: preliminary findings. Arch Gen Psychiatry 57(4):349–356
Santos JL, Aparicio A, Bagney A, Sanchez-Morla EM, Rodriguez-Jimenez R, Mateo J, Jimenez-Arriero MA (2014) A five-year follow-up study of neurocognitive functioning in bipolar disorder. Bipolar Disord. doi:10.1111/bdi.12215
Schmitt A, Steyskal C, Bernstein HG, Schneider-Axmann T, Parlapani E, Schaeffer EL, Gattaz WF, Bogerts B, Schmitz C, Falkai P (2009) Stereologic investigation of the posterior part of the hippocampus in schizophrenia. Acta Neuropathol 117(4):395–407. doi:10.1007/s00401-008-0430-y
Schmitz C (1998) Variation of fractionator estimates and its prediction. Anat Embryol (Berl) 198(5):371–397
Schmitz C, Hof PR (2005) Design-based stereology in neuroscience. Neuroscience 130(4):813–831. doi:10.1016/j.neuroscience.2004.08.050
Schmitz C, Eastwood BS, Tappan SJ, Glaser JR, Peterson DA, Hof PR (2014) Current automated 3D cell detection methods are not a suitable replacement for manual stereologic cell counting. Front Neuroanat 8:27. doi:10.3389/fnana.2014.00027
Selemon LD, Rajkowska G (2003) Cellular pathology in the dorsolateral prefrontal cortex distinguishes schizophrenia from bipolar disorder. Curr Mol Med 3(5):427–436
Stark AK, Petersen AO, Gardi J, Gundersen HJ, Pakkenberg B (2007) Spatial distribution of human neocortical neurons and glial cells according to sex and age measured by the saucer method. J Neurosci Methods 164(1):19–26. doi:10.1016/j.jneumeth.2007.03.019
Sterio DC (1984) The unbiased estimation of number and sizes of arbitrary particles using the disector. J Microsc 134(Pt 2):127–136
Stockmeier CA, Mahajan GJ, Konick LC, Overholser JC, Jurjus GJ, Meltzer HY, Uylings HB, Friedman L, Rajkowska G (2004) Cellular changes in the postmortem hippocampus in major depression. Biol Psychiatry 56(9):640–650. doi:10.1016/j.biopsych.2004.08.022
Strasser HC, Lilyestrom J, Ashby ER, Honeycutt NA, Schretlen DJ, Pulver AE, Hopkins RO, Depaulo JR, Potash JB, Schweizer B, Yates KO, Kurian E, Barta PE, Pearlson GD (2005) Hippocampal and ventricular volumes in psychotic and nonpsychotic bipolar patients compared with schizophrenia patients and community control subjects: a pilot study. Biol Psychiatry 57(6):633–639. doi:10.1016/j.biopsych.2004.12.009
Uranova NA, Vostrikov VM, Orlovskaya DD, Rachmanova VI (2004) Oligodendroglial density in the prefrontal cortex in schizophrenia and mood disorders: a study from the Stanley Neuropathology Consortium. Schizophr Res 67(2–3):269–275. doi:10.1016/S0920-9964(03)00181-6
West MJ, Slomianka L, Gundersen HJ (1991) Unbiased stereological estimation of the total number of neurons in thesubdivisions of the rat hippocampus using the optical fractionator. Anat Rec 231(4):482–497. doi:10.1002/ar.1092310411
West MJ, Coleman PD, Flood DG, Troncoso JC (1994) Differences in the pattern of hippocampal neuronal loss in normal ageing and Alzheimer’s disease. Lancet 344(8925):769–772
Williams RW, Rakic P (1988) Three-dimensional counting: an accurate and direct method to estimate numbers of cells in sectioned material. J Comp Neurol 278(3):344–352. doi:10.1002/cne.902780305
Woods SW (2003) Chlorpromazine equivalent doses for the newer atypical antipsychotics. J Clin Psychiatry 64(6):663–667
Zakzanis KK, Leach L, Kaplan E (1998) On the nature and pattern of neurocognitive function in major depressive disorder. Neuropsychiatry Neuropsychol Behav Neurol 11(3):111–119
Acknowledgments
The study was supported by the European Community under the Sixth Framework Program (BrainNet Europe II, LSHM-CT-2004-503039). This paper reflects only the authors’ views, and the Community is not liable for any use that may be made of the information contained therein. The authors thank Jacquie Klesing, Board-certified Editor in the Life Sciences (ELS), for editing assistance with the manuscript.
Conflicts of interest
B. Malchow, S. Strocka, F. Frank, T. Schneider-Axmann, J. Steiner H.-G. Bernstein, B. Bogerts, C. Schmitz and D. Reich-Erkelenz declare no conflicts of interest. A. Hasan has been invited to scientific meetings by Lundbeck, Janssen-Cilag and Pfizer and received a paid speakership from Desitin. He is a member of the advisory board of Roche. A. Schmitt was honorary speaker for TAD Pharma and Roche and has been a member of advisory boards for Roche. P. Falkai has been a member of the advisory boards for Janssen-Cilag, BMS, Lundbeck, Pfizer, Lilly and AstraZeneca and received an educational grant from AstraZeneca and honoraria as a lecturer from Janssen-Cilag, BMS, Lundbeck, Pfizer, Lilly and AstraZeneca.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Malchow, B., Strocka, S., Frank, F. et al. Stereological investigation of the posterior hippocampus in affective disorders. J Neural Transm 122, 1019–1033 (2015). https://doi.org/10.1007/s00702-014-1316-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00702-014-1316-x