Abstract
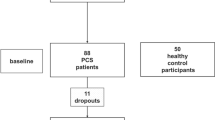
The autonomic nervous system (ANS) is regularly affected in Parkinson’s disease (PD). Information on autonomic dysfunction can be derived from e.g. altered heart rate variability (HRV) and sympathetic skin response (SSR). Such parameters can be quantified easily and measured repeatedly which might be helpful for evaluating disease progression and therapeutic outcome. In this 2-center study, HRV and SSR of 45 PD patients and 26 controls were recorded. HRV was measured during supine metronomic breathing and analyzed in time- and frequency-domains. SSR was evoked by repetitive auditory stimulation. Various ANS parameters were compared (1) between patients and healthy controls, (2) to clinical scales (Unified Parkinson’s disease rating scale, Mini-Mental State Examination, Becks Depression Inventory), and (3) to disease duration. Root mean square of successive differences (RMSSD) and low frequency/high frequency (LF/HF) ratio differed significantly between PD and controls. Both, HRV and SSR parameters showed low or no association with clinical scores. Time-domain parameters tended to be affected already at early PD stages but did not consistently change with longer disease duration. In contrast, frequency-domain parameters were not altered in early PD phases but tended to be lower (LF, LF/HF ratio), respectively higher (HF) with increasing disease duration. This report confirms previous results of altered ANS parameters in PD. In addition, it suggests that (1) these ANS parameters are not relevantly associated with motor, behavioral, and cognitive changes in PD, (2) time-domain parameters are useful for the assessment of early PD, and (3) frequency-domain parameters are more closely associated with disease duration.

Similar content being viewed by others
References
Beck AT, Steer RA, Ball R, Ranieri W (1996) Comparison of Beck depression inventories -IA and -II in psychiatric outpatients. J Pers Assess 67:588–597
Braune HJ, Korchounov AM, Schipper HI (1997) Autonomic dysfunction in Parkinson’s disease assessed by sympathetic skin response: a prospective clinical and neurophysiological trial on 50 patients. Acta Neurol Scand 95:293–297
Brisinda D, Sorbo AR, Di Giacopo R, Venuti A, Bentivoglio AR et al (2014) Cardiovascular autonomic nervous system evaluation in Parkinson disease and multiple system atrophy. J Neurol Sci 336:197–202. doi:10.1016/j.jns.2013.10.039
Choi BO, Bang OY, Sohn YH, Sunwoo IN (1998) Sympathetic skin response and cardiovascular autonomic function tests in Parkinson’s disease. Yonsei Med J 39:439–445
Folstein MF, Folstein SE, McHugh PR (1975) “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 12:189–198
Friedrich C, Rudiger H, Schmidt C, Herting B, Prieur S et al (2008) Baroreflex sensitivity and power spectral analysis in different extrapyramidal syndromes. J Neural Transm 115:1527–1536
Friedrich C, Rudiger H, Schmidt C, Herting B, Prieur S et al (2010) Baroreflex sensitivity and power spectral analysis during autonomic testing in different extrapyramidal syndromes. Mov Disord 25:315–324
Gibb WR, Lees AJ (1988) A comparison of clinical and pathological features of young- and old-onset Parkinson’s disease. Neurology 38:1402–1406
Goetz CG, Tilley BC, Shaftman SR, Stebbins GT, Fahn S et al (2008) Movement Disorder Society-sponsored revision of the Unified Parkinson’s Disease Rating Scale (MDS-UPDRS): scale presentation and clinimetric testing results. Mov Disord 23:2129–2170
Haapaniemi TH, Korpelainen JT, Tolonen U, Suominen K, Sotaniemi KA et al (2000) Suppressed sympathetic skin response in Parkinson disease. Clin Auton Res 10:337–342
Haapaniemi TH, Pursiainen V, Korpelainen JT, Huikuri HV, Sotaniemi KA et al (2001) Ambulatory ECG and analysis of heart rate variability in Parkinson’s disease. J Neurol Neurosurg Psychiatry 70:305–310
Hoehn MM, Yahr MD (1967) Parkinsonism: onset, progression and mortality. Neurology 17:427–442
Jain S, Ton TG, Perera S, Zheng Y, Stein PK et al (2012) Cardiovascular physiology in premotor Parkinson’s disease: a neuroepidemiologic study. Mov Disord 27:988–995
Kallio M, Haapaniemi T, Turkka J, Suominen K, Tolonen U et al (2000) Heart rate variability in patients with untreated Parkinson’s disease. Eur J Neurol 7:667–672
Lanfranchi PA, Fradette L, Gagnon J-F, Colombo R, Montplaisir J (2007) Cardiac autonomic regulation during sleep in idiopathic REM sleep behavior disorder. Sleep 30:1019–1025
Linden D, Diehl RR, Berlit P (1997) Sympathetic cardiovascular dysfunction in long-standing idiopathic Parkinson’s disease. Clin Aut Res 7:311–314
Maetzler W, Liepelt I, Berg D (2009) Progression of Parkinson’s disease in the clinical phase: potential markers. Lancet Neurol 8:1158–1171
Mesec A, Sega S, Trost M, Pogacnik T (1999) The deterioration of cardiovascular reflexes in Parkinson’s disease. Acta Neurol Scand 100:296–299
Niwa F, Kuriyama N, Nakagawa M, Imanishi J (2011) Circadian rhythm of rest activity and autonomic nervous system activity at different stages in Parkinson’s disease. Aut Neurosci 165:195–200
Orimo S, Ozawa E, Nakade S, Sugimoto T, Mizusawa H (1999) (123)I-metaiodobenzylguanidine myocardial scintigraphy in Parkinson’s disease. J Neurol Neurosurg Psychiatry 67:189–194
Pursiainen V, Haapaniemi TH, Korpelainen JT, Huikuri HV, Sotaniemi KA et al (2002) Circadian heart rate variability in Parkinson’s disease. J Neurol 249:1535–1540
Reimann M, Friedrich C, Gasch J, Reichmann H, Rudiger H et al (2010a) Trigonometric regressive spectral analysis reliably maps dynamic changes in baroreflex sensitivity and autonomic tone: the effect of gender and age. PLoS One 5:e12187
Reimann M, Schmidt C, Herting B, Prieur S, Junghanns S et al (2010b) Comprehensive autonomic assessment does not differentiate between Parkinson’s disease, multiple system atrophy and progressive supranuclear palsy. J Neural Transm 117:69–76
Rüdiger H, Klinghammer L, Scheuch K (1999) The trigonometric regressive spectral analysis–a method for mapping of beat-to-beat recorded cardiovascular parameters on to frequency domain in comparison with Fourier transformation. Comput Methods Programs Biomed 58:1–15
Sauvageot N, Vaillant M, Diederich NJ (2011) Reduced sympathetically driven heart rate variability during sleep in Parkinson’s disease: a case-control polysomnography-based study. Mov Disord 26:234–240. doi:10.1002/mds.23479
Schestatsky P, Ehlers JA, Rieder CR, Gomes I (2006) Evaluation of sympathetic skin response in Parkinson’s disease. Park Relat Disord 12:486–491
Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology (1996) Heart rate variability: standards of measurement, physiological interpretation and clinical use. Circulation 93:1043–1065
Turkka JT, Tolonen U, Myllylä VV (1987) Cardiovascular reflexes in Parkinson’s disease. Eur Neurol 26:104–112
Van Dijk JG, Haan J, Zwinderman K, Kremer B, van Hilten BJ et al (1993) Autonomic nervous system dysfunction in Parkinson’s disease: relationships with age, medication, duration, and severity. J Neurol Neurosurg Psychiatry 56:1090–1095
Visser M, van Rooden SM, Verbaan D, Marinus J, Stiggelbout AM et al (2008) A comprehensive model of health-related quality of life in Parkinson’s disease. J Neurol 255:1580–1587. doi:10.1007/s00415-008-0994-4
Wang N, Gibbons CH, Lafo J, Freeman R (2013) α-Synuclein in cutaneous autonomic nerves. Neurology 81:1604–1610. doi:10.1212/WNL.0b013e3182a9f449
Ziemssen T, Reichmann H (2010) Cardiovascular autonomic dysfunction in Parkinson’s disease. J Neurol Sci 289:74–80
Ziemssen T, Reichmann H (2011) Cardiovascular autonomic testing in extrapyramidal disorders. J Neurol Sci 310:129–132. doi:10.1016/j.jns.2011.07.032
Acknowledgments
We thank all individuals who took part in the study. WM was partly supported by the European Union project SENSE-PARK, funded under the Seventh Framework Programme, Cooperation-ICT (grant agreement no. 288557).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Maetzler, W., Karam, M., Berger, M.F. et al. Time- and frequency-domain parameters of heart rate variability and sympathetic skin response in Parkinson’s disease. J Neural Transm 122, 419–425 (2015). https://doi.org/10.1007/s00702-014-1276-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00702-014-1276-1