Summary
Aim
The aim of this study was to establish erythropoietin as a protective factor against brain ischemia during open heart surgery.
Methods
A total of 36 consecutive patients scheduled for revascularization heart surgery were included in the study. Of the patients 18 received 3 intravenous doses of recombinant human erythropoietin (rHuEpo, 24,000 IU) and 18 patients received a placebo. Magnetic resonance imaging (MRI) to detect new brain ischemic lesions was performed. Additionally, S100A, S100B, neuron-specific enolase A and B (NSE-A and B) and the concentration of antibodies against N‑methyl-D-aspartate receptors (NMDAR) to identify new neurological complications were determined.
Results
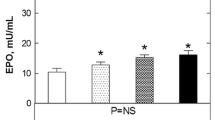
Patients who received rHuEpo showed no postoperative ischemic changes in the brain on MRI images. In the control group 5 (27.8 %) new ischemic lesions were found. The NMDAR antibody concentration, S100A, S100B and NSE showed no significant differences between the groups for new cerebral ischemia. High levels of lactate before and after external aortic compression (p = 0.022 and p = 0.048, respectively) and duration of operation could predict new ischemic lesions (p = 0.009).
Conclusions
The addition of rHuEpo reduced the formation of lesions detectable by MRI in the brain and could be used clinically as neuroprotection in cardiac surgery.
Similar content being viewed by others
References
Hogue CW, Murphy SF, Schechtman KB, et al. Risk factors for early or delayed stroke after cardiac surgery. Circulation. 1999;100:642–7.
Hogue CW, Barzilai B, Pieper KS, et al. Sex differences in neurological outcomes and mortality after cardiac surgery: a society of thoracic surgery national database report. Circulation. 2001;103:2133–7.
Newman MF, Kirchner JL, Phillips-Bute B, et al. Longitudinal assessment of neurocognitive function after coronary-artery bypass surgery. Neurological Outcome Research Group and the Cardiothoracic Anesthesiology Research Endeavors Investigators. N Engl J Med. 2001;344:395–402.
Arrowsmith JE, Grocott HP, Reves JG, et al. Central nervous system complications of cardiac surgery. Br J Anaesth. 2000;84:378–93.
Van Dijk D, Keizer AM, Diephuis JC, et al. Neurocognitive dysfunction after coronary artery bypass surgery: a systematic review. J Thorac Cardiovasc Surg. 2000;120:632–9.
Shroyer AL, Coombs LP, Peterson ED, et al. 30-day operative mortality and morbidity risk models. Ann Thorac Surg. 2003;75:1856–64.
Ehrenreich H, Weissenborn K, Prange H, et al. Recombinant human erythropoietin in the treatment ofacute ischemic stroke. Stroke. 2009;40:647–56.
Ehrenreich H, Timner W, Sirén AL. A novel role for an established player: anemia drug erythropoietin for the treatment of cerebral hypoxia/ischemia. Transfus Apher Sci. 2004;31:39–44.
Mahanna EP, Blumenthal JA, White WD, et al. Defining neuropsychological dysfunction after coronary artery bypass grafting. Ann Thorac Surg. 1996;61:1342–7.
Jönsson H, Johnsson P, Alling C, et al. S100beta after coronary artery surgery: release pattern, source of contamination, and relation to neuropsychological outcome. Ann Thorac Surg. 1999;68:2202–8.
Herrmann M, Ebert AD, Galazky I, et al. Neurobehavioral outcome prediction after cardiac surgery: role of neurobiochemical markers of damage to neuronal and glial brain tissue. Stroke. 2000;31:645–50.
Donato R. Functional roles of S100 proteins, calcium-binding proteinsof the EF-hand type. Biochim Biophys Acta. 1999;1450:191–231.
Hogue CW Jr, Palin CA, Arrowsmith JE. Cardiopulmonary bypass management and neurologicoutcomes: an evidence-based appraisal of current practices. Anesth Analg. 2006;103:21–37.
Ehrenreich H, Hasselblatt M, Dembowski C, et al. Erythropoietin therapy for acute stroke is both safe and beneficial. Mol Med. 2002;8(8):495–505.
Ehrenreich H, Degner D, Meller J, et al. Erythropoietin: a candidate compound for neuroprotection in schizophrenia. Mol Psychiatry. 2004;9:42–54.
Lacombe C, Mayeux P. The molecular biology of erythropoietin. Nephrol Dial Transplant. 1999;14:22–8.
Lipsic E, Schoemaker RG, van der Meer P, et al. Protective effects of erythropoietin in cardiac ischemia: from bench to bedside. J Am Coll Cardiol. 2006;48:2161–7.
Parsa CJ, Matsumoto A, Kim J, et al. A novel protective effect of erythropoietin in the infarcted heart. J Clin Invest. 2003;112:999–1007.
Miskowiak K, O’Sullivan U, Harmer CJ. Erythropoietin reduces neural and cognitive processing of fear in human models of antidepressant drug action. Biol Psychiatry. 2007;62:1244–50.
Nagpal AD, Bhatnagar G, Cutrara CA, et al. Early outcomes of coronary artery bypass with and without cardiopulmonary bypass in octogenarians. Can J Cardiol. 2006;22:849–53.
Zanatta P, Forti A, Minniti G, et al. Brain emboli distribution and differentiation during cardiopulmonary bypass. J Cardiothorac Vasc Anesth. 2013;27(5):865–75. doi:10.1053/j.jvca.2012.12.022.
Douglas JM, Spaniol SE. A multimodal approach to the prevention of postoperative stroke in patients undergoing coronary artery bypass surgery. Am J Surg. 2009;197:587–90.
Masabni K, Raza S, Blackstone EH, et al. Does preoperative carotid stenosis screening reduce perioperative stroke in patients undergoing coronary artery bypass grafting? J Thorac Cardiovasc Surg. 2015;149(5):1253–60.
Lee EJ, Choi KH, Ryu JS, et al. Stroke risk after coronary artery bypass graft surgery and extent of cerebral artery atherosclerosis. J Am Coll Cardiol. 2011;57:1811–8.
Gottesman RF, Sherman PM, Grega MA. Watershed strokes after cardiac surgery: diagnosis, etiology, and outcome. Stroke. 2006;37:2306–11.
Lakic N, Surlan K, Jerin A, et al. Importance of erythropoietin in brain protection aftercardiac surgery: a pilot study. Heart Surg Forum. 2010;13:185–9.
Lipsic E, Westenbrink BD, van der Meer P, et al. Erythropoietin induces neovascularisation and improves cardiac function in rats with heart failure after myocardial infarction. J Am Coll Cardiol. 2005;46(1):125–33.
Sakanaka M, Wen TC, Matsuda S, et al. Invivo evidence that erythropoietin protects neurons fromischemic damage. Proc Natl Acad Sci USA. 1998;95:4635–40.
Junk AK, Mammis A, Savitz SI, et al. Erythropoietin administration protects retinal neurons fromacute ischemia-reperfusion injury. Proc Natl Acad Sci USA. 2002;99:10659–64.
Bokesch PM, Izykenova GA, Justice JB, et al. NMDA receptor antibodies predict adverse neurological outcome after cardiac surgery in high-risk patients. Stroke. 2006;37:1432–6.
Stolz E, Gerriets T, Kluge A, et al. Diffusion-weighted magnetic resonance imaging and neurobiochemical markers after aortic valve replacement: implication for future neuroprotective trials? Stroke. 2004;35:888–92.
Dambinova SA, Khounteev GA, Izykenova GA, et al. Blood test detecting autoantibodies to N‑methyl-D-aspartate neuroreceptors for evaluation of patients with transient ischemic attack and stroke. Clin Chem. 2003;49:1752–62.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
N. Lakič, M. Mrak, M. Šušteršič, P. Rakovec, and M. Bunc declare that they have no competing interests.
Ethical standards
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975 (in its current revised form). Informed consent was obtained from all patients included in the study.
Rights and permissions
About this article
Cite this article
Lakič, N., Mrak, M., Šušteršič, M. et al. Perioperative erythropoietin protects the CNS against ischemic lesions in patients after open heart surgery. Wien Klin Wochenschr 128, 875–881 (2016). https://doi.org/10.1007/s00508-016-1063-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00508-016-1063-0