Summary
Background
Papillary carcinoma of the thyroid (PTC) is generally a slow growing tumor with favorable prognosis, while anaplastic thyroid carcinoma (ATC) is highly aggressive malignancy. Genetic defects in apoptotic pathways may contribute to differences in their biological behavior.
Methods
In this study, we analyzed immunohistochemically the expression of apoptosis-related molecules: galectin-3, Bcl-2, survivin (antiapoptotic), and Bax (pro-apoptotic), in archival tissue sections of PTC (n = 69) and ATC (n = 30) and correlated the results with clinicopathological parameters of these tumors.
Results
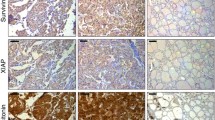
Galectin-3 and Bcl-2 showed a similar trend of down-regulation from high levels of both in PTC to low levels in ATC (p < 0.05). Bax was expressed at high levels in both type of thyroid carcinoma. Expression of survivin increased from PTC to ATC (p < 0.05), which may, at least in part, further facilitate the ability of malignant thyroid cell of ATC to escape programmed cell death despite high Bax expression. Only survivin, but not galectin-3, Bcl-2, or Bax, correlated significantly with lymph node metastasis presence and advanced stages of malignancy.
Conclusions
In conclusion, this study documented down-regulation of galectin-3 and Bcl-2 (antiapoptotic molecules) and stepwise increase of survivin (inhibitor of apoptosis), during thyroid tumor progression from PTC to ATC. Correlation of high survivin expression with aggressive behavior implies its role in progression of thyroid tumor malignancy and suggests that survivin could be a useful tool in the prediction of aggressiveness of a subset of papillary carcinomas and a possible target for molecular therapy for ATC patients.
Zusammenfassung
Grundlagen
Das papilläre Schilddrüsenkarzinom (PTC) ist im Allgemeinen ein langsam wachsender Tumor mit einer guten Prognose. Das anaplastische Schilddrüsenkarzinom (ATC) dahingegen ist ein hoch aggressiver bösartiger Tumor. Genetische Defekte der Apoptose könnten zu diesen Unterschieden im biologischen Verhalten beitragen.
Methodik
In dieser Studie analysierten wir in Gewebsschnitten aus dem Archivmaterial von 69 PTC und 30 ATC histochemisch die Expression von Molekülen, die mit der Apoptose einen Zusammenhang haben: Galectin-3, Bcl-2, Survivin (antiapoptotisch) und Bax (proapoptotisch). Die Ergebnisse wurden mit klinisch-pathologischen Parametern dieser Tumore korreliert.
Ergebnisse
Galectin-3 und Bcl-2 zeigten einen ähnlichen Trend der Down-Regulation von hohen Werten beim PTC zu niedrigen Werten beim ATC (p < 0,05). Bax wurde in beiden Karzinomtypen hoch exprimiert. Die Expression von Survivin stieg von PTC zum ATC an (p < 0,05). Dies könnte zumindest teilweise die Fähigkeit der malignen Schilddrüsenzelle des ATC zusätzlich erleichtern, dem programmierten Zelltod trotz hoher Bax Expression zu entkommen. Nur Survivin, nicht jedoch Galectin-3, Bcl-2 oder Bax, korrelierte signifikant mit dem Vorhandensein von Lymphknotenmetastasen und fortgeschrittenen Stadien der Bösartigkeit.
Schlussfolgerungen
Zusammenfassend dokumentierte unsere Studie eine Down-Regulation von Galektin3 und Bcl-2 (antiapoptotische Moleküle) und einen stufenweise Anstieg von Survivin (Hemmer der Apoptose) während der Progression der Malignität von Schilddrüsentumoren vom PTC zum ATC. Die Korrelation von hoher Survivin Expression mit dem aggressiven Verhalten des Tumors impliziert dessen Rolle bei der Progression der Bösartigkeit von Schilddrüsentumoren. Survivin könnte daher ein nützlicher Parameter zur Vorhersage der Aggressivität einer Untergruppe von PTC sein. Außerdem wäre es ein mögliches Ziel einer molekularen Therapie von Patienten mit ATC.


Similar content being viewed by others
References
Van der Laan B, Freeman JL, Tsang RW, Asa SL. The association of well-differentiated thyroid carcinoma with insular or anaplastic thyroid carcinoma: Evidence for dedifferentiation in tumor progression. Endocr Pathol. 1993;4:215–21.
Li Volsi VA. Surgical pathology of the thyroid. Philadelphia: WB Saunders; 1990. S. 136–72.
Mazzaferri EL, Jhiang S. Long-term impact of initial surgical and medical therapy on papillary and follicular thyroid cancer. Am J Med. 1994;97:418–28.
Giuffrida D, Gharib H. Anaplastic thyroid carcinoma: current diagnosis and treatment. Ann Oncol. 2000;11:1083–9.
Wiseman SM, Masoudi H, Niblock P, Turbin D, Rajput A, Hay J, et al. Anaplastic thyroid carcinoma: expression profile of targets for therapy offers new insights for disease treatment. Ann Surg Oncol. 2007;14:719–29.
Smallridge RC, Marlow LA, Copland JA. Anaplastic thyroid cancer: molecular pathogenesis and emerging therapies. Endocr Relat Cancer. 2009;16:17–44.
Burlacu A. Regulation of apoptosis by Bcl-2 family proteins. J Cell Mol Med. 2003;7:249–57.
Perillo NL, Marcus ME, Baum LG. Galectins: versatile modulators of cell adhesion, cell proliferation and cell death. J Mol Med. 1998;76:402–12.
Califice S, Castronovo V, Van Den Brule F. Galectin-3 and cancer (Review). Int J Oncol. 2004;25:983–92.
Xu XC, El-Naggar AK, Lotan R. Differential expression of galectin-1 and galectin-3 in thyroid tumors. Potential diagnostic implications. Am J Pathol. 1995;147:815–22.
Cvejic D, Savin S, Paunovic I, Tatic S, Havelka M, Sinadinovic J. Immunohistochemical localization of galectin-3 in malignant and benign human thyroid tissue. Anticancer Res. 1998;18:2637–42.
Akahani S, Nangia-Makker P, Inohara H, Chol Kim H-R, Raz A. Galectin-3: a novel antiapoptotic molecule with a functional BH1 (NWGR) domain of Bcl-2 family. Cancer Res. 1997;57:5272–6.
Nakahara S, Oka N, Raz A. On the role of galectin-3 in cancer apoptosis. Apoptosis. 2005;10:267–75.
Ambrosini G, Adida C, Altieri DC. A novel anti-apoptosis gene, survivin, expressed in cancer and lymphoma. Nat Med. 1997;3:917–21.
Deveraux QL, Reed JC. IAP family proteins—suppressors of apoptosis. Genes Dev. 1999;13:239–52.
Mita AC, Mita MM, Nawrocki ST, Giles FJ. Survivin: key regulator of mitosis and apoptosis and novel target for cancer therapeutics. Clin Cancer Res. 2008;14:5000–5.
Waligórska-Stachura J, Jankowska A, Was´ko R, Liebert W, Biczysko M, Czarnywojtek A, et al. Survivin-prognostic tumor biomarker in human neoplasms-review. Ginekol Pol. 2012;83:537–40.
Ito Y, Yoshida H, Uruno T, Nakano K, Miya A, Kobayashi K, et al. Survivin expression is significantly linked to the dedifferentiation of thyroid carcinoma. Oncol Rep. 2003;10:1337–40.
Antonaci A, Consorti F, Mardente S, Natalizi S, Giovannone G, Della Rocca C. Survivin and cyclin D1 are jointly expressed in thyroid papillary carcinoma and microcarcinoma. Oncol Rep. 2008;20:63–7.
Chen Z, Liu N, Zhu G, Dralle H, Hoang-Vu C. Targeting of the anti-apoptotic gene survivin in human thyroid carcinoma. Int J Mol Med. 2012;30:465–72.
Pannone G, Santoro A, Pasquali D, Zamparese R, Mattoni M, Russo G, et al. The role of survivin in thyroid tumors: differences of expression in well-differentiated, non-well-differentiated, and anaplastic thyroid cancers. Thyroid. 2014;24:511–9.
Sobin LH, Wittekind Ch, editors. International Union Against Cancer (UICC). TNM classification of malignant tumors. 6th ed. New York: Wiley-Liss; 2002.
Lin CI, Wang EE, Abramson MA, Donner DB, Bertagnolli MM, Moore FD Jr., Ruan DT. Galectin-3 regulates apoptosis and doxorubicin chemoresistance in papillary thyroid cancer cells. Biochem Biophys Res Commun. 2009;379:626–31.
Yang R-Y, Hsu DK, Liu F-T. Expression of galectin-3 modulates T-cell growth and apoptosis. Proc Natl Acad Sci USA. 1996;93:6737–42.
Pollina L, Pacini F, Fontanini G, Vignati S, Bevilacqua G, Basolo F. Bcl-2, p53 and proliferating cell nuclear antigen expression are related to the degree of differentiation in thyroid carcinomas. Br J Cancer. 1996;73:139–43.
Tirrò E, Consoli ML, Massimino ML, Manzella L, Frasca F, Sciacca L, et al. Altered expression of c-IAP1, survivin, and Smac contributes to chemotherapy resistance in thyroid cancer cells. Cancer Res. 2006;66:4263–72.
Orlandi F, Saggiorato E, Pivano G, Puligheddu B, Termine A, Cappia S, et al. Galectin-3 is a presurgical marker of human thyroid carcinoma. Cancer Res. 1998;58:3013–20.
Cvejic DS, Savin SB, Petrovic IM, Paunovic IR, Tatic SB, Havelka MJ. Galectin-3 expression in papillary thyroid carcinoma: relation to histomorphologic growth pattern, lymph node metastasis, extrathyroid invasion, and tumor size. Head Neck. 2005;12:1049–55.
Türköz HK, Oksüz H, Yurdakul Z, Ozcan D. Galectin-3 expression in tumor progression and metastasis of papillary thyroid carcinoma. Endocr Pathol. 2008;19:92–6.
Kawachi K, Yoshifumi M, Yonezawa S, Nakano S, Shirao K, Natsugoe S, et al. Galectin-3 expression in various thyroid neoplasms and its possible role in metastasis formation. Hum Pathol. 2000;31:428–33.
Aksoy M, Giles Y, Kapran Y, Terzioglu T, Tezelman S. Expression of Bcl-2 in papillary thyroid cancers an its prognostic value. Acta Chir Belg. 2005;105:644–8.
Martinez-Brocca MA, Castilla C, Navarro E, Amaya MJ, Travado P, Japon MA, Saez C. Clinicopathological correlation of Bcl-xL and Bax expression in differentiated thyroid carcinoma. Clin Endocrinol (Oxf). 2008;68:190–7.
Mehrotra S, Languino LR, Raskett CM, Mercurio AM, Dohi T, Altieri DC. IAP regulation of metastasis. Cancer Cell. 2010;17:53–77.
Acknowledgment
The authors are grateful to Dr J. Anna Nikolic, for language correction of the manuscript. This work is supported by the Ministry of Education, Science and Technological Development of the Republic of Serbia, project 173050: “Molecular characterization of thyroid gland tumors: biological and clinical aspects”.
Conflict of interest
No actual or potential conflicts of interest exist.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Selemetjev, S., Savin, S., Paunovic, I. et al. Changes in the expression pattern of apoptotic molecules (galectin-3, Bcl-2, Bax, survivin) during progression of thyroid malignancy and their clinical significance. Wien Klin Wochenschr 127, 337–344 (2015). https://doi.org/10.1007/s00508-014-0674-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00508-014-0674-6