Abstract
Background
The mutations responsible for cystinosis in South African patients are currently unknown. A pertinent question is whether they are similar to those described elsewhere in the world.
Methods
Children who were being managed for cystinosis in the Western Cape Province of South Africa between 2002 and 2013 were studied. All underwent molecular analysis to detect sequence variations in the cystinosis gene.
Results
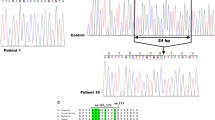
This cohort study included 20 patients, 13 of whom were Xhosa-speaking black South Africans and seven were Cape Coloureds (mixed race); none were Caucasian. All had nephropathic infantile-type cystinosis with evidence of proximal tubulopathy, with glycosuria and renal phosphate wasting. Diagnosis was confirmed in 19 cases by demonstrating an elevated cystine concentration in leukocytes. Molecular analysis of the cystinosin gene revealed that 19 patients had a G > A mutation in intron 11 (CTNS-c.971-12G > A p.D324AfsX44) which caused an out-of-frame 10-bp insertion. Of these 19 patients, 16 were homozygous for this mutation, which was the most frequent mutation identified in the alleles of the black South African and Cape Coloured patients (96 and 71 %, respectively).
Conclusion
We recommend that black South African and Cape Coloured patients presenting with cystinosis be tested for CTNS-c.971-12G > A in the first instance, with the possibility of prenatal testing being offered to at-risk families.
Similar content being viewed by others
References
Gahl WA, Bashan N, Tietze F, Bernardini I, Schulman JD (1982) Cystine transport is defective in isolated leukocyte lysosomes from patients with cystinosis. Science 217:1263–1265
Gahl WA, Thoene JG, Schneider JA (2002) Cystinosis. N Engl J Med 347:111–121
Gahl WA, Balog JZ, Kleta R (2007) Nephropathic cystinosis in adults: natural history and effects of oral cysteamine therapy. Ann Intern Med 147:242–250
Gahl WA, Reed GF, Thoene JG, Schulman JD, Rizzo WB, Jonas AJ, Denman DW, Schlesselman JJ, Corden BJ, Schneider JA (1987) Cysteamine therapy for children with nephropathic cystinosis. N Engl J Med 316:971–977
Stenson PD, Ball EV, Mort M, Phillips AD, Shiel JA, Thomas NS, Abeysinghe S, Krawczak M, Cooper DN (2003) The human gene mutation database (HGMD®): 2003 update. Hum Mutat 21:577–581
Topaloglu R, Vilboux T, Coskun T, Ozaltin F, Tinloy B, Gunay-Aygun M, Bakkaloglu A, Besbas N, van den Heuvel L, Kleta R, Gahl WA (2012) Genetic basis of cystinosis in Turkish patients:a single-center experience. Pediatr Nephrol 27:115–121
Yeetong P, Tongkobpetch S, Kingwatanakul P, Deekajorndech T, Bernardini IM, Suphapeetiporn K, Gahl WA, Shotelersuk V (2012) Two novel CTNS mutations in cystinosis patients in Thailand. Gene 499(2):323–325
Shahkarami S, Galehdari H, Ahmadzadeh A, Babaahmadi M, Pedram M (2013) The first molecular genetics analysis of individuals suffering from nephropathic cystinosis in the southwestern Iran. Nefrologia 33(3):308–315
Shotelersuk V, Larson D, Anikster Y, McDowell G, Lemons R, Bernardini I, Guo J, Thoene J, Gahl WA (1998) CTNS mutations in an American-based population of cystinosis patients. Am J Hum Genet 63:1352–1362
McGowan-Jordan J, Stoddard KLP, Orrbine E, McLaine P, Town M, Goodyer P, MacKenzie A, Heick H (1999) Molecular analysis of cystinosis: probable Irish origin of the most common french Canadian mutation. Eur J Hum Genet 7:671–678
Kalatzis V, Cherqui S, Jean G, Cordier B, Cochat P, Broyer M, Antignac C (2001) Characterization of a putative founder mutation that accounts for the high incidence of cystinosis in Brittany. J Am Soc Nephrol 12:2170–2174
Kleta R, Anikster Y, Lucero C, Shotelersuk V, Huizing M, Bernardini I, Park M, Thoene J, Schneider J, Gahl WA (2001) CTNS mutations in African American patients with cystinosis. Mol Genet Metab 74:332–337
Soliman NA, Elmonem MA, van den Heuvel L, Hamid RH, Gamal M, Bongaers I, Marie S, Levtchenko E (2014) Mutational spectrum of the CTNS gene in Egyptian patients with nephropathic cystinosis. JIMD Rep. doi:10.1007/8904_2013_288
Patterson N, Petersen DC, van der Ross RE, Sudoyo H, Glashoff RH, Marzuki S, Reich D, Hayes VM (2009) Genetic structure of a unique admixed population: implications for medical research. Hum Mol Genet 19:411–419
Anikster Y, Lucero C, Touchmann JW, Huizing M, McDowell G, Shotelersuk V, Green ED, Gahl WA (1999) Identification and detection of the common 65-kb deletion breakpoint in the nephropathic cystinosis gene (CTNS). Mol Genet Metab 66:111–116
Town M, Jean G, Cherqui S, Attard M, Forestier L, Whitmore SA, Callen DF, Gribouval O, Broyer M, Bates GP, van’t Hoff W, Antignac C (1998) A novel gene encoding an integral membrane protein is mutated in nephropathic cystinosis. Nat Genet 18(4):319–324
Macías-Vidal J, Rodés M, Hernández-Pérez JM, Vilaseca MA, Coll MJ (2009) Analysis of the CTNS gene in 32 cystinosis patients from Spain. Clin Genet 76:486–489
Kaletzis V, Nevo N, Cherqui S, Gasnier B, Antignac C (2004) Molecular pathogenesis of cystinosis: effect of CTNS mutations on the transport activity and subcellular localization of cystinosin. Hum Mol Genet 13:1361–1371
The 1000 Genomes Project Consortium (2012). An integrated map of genetic variation from 1,092 human genomes. Nature 491:56–65
Geer LY, Marchler-Bauer A, Geer RC, Han L, He J, He S, Liu C, Shi W, Bryant SH (2010) The NCBI BioSystems database. Nucleic Acids Res 38:492–496
Adzhubei IA, Schmidt S, Peshkin L, Ramensky VE, Gerasimova A, Bork P, Kondrashov AS, Sunyaev SR (2010) A method and server for predicting damaging missense mutations. Nat Methods 7(4):248–249
Attard M, Jean G, Forestier L, Cherqui S, van’t Hoff W, Broyer M, Antignac C, Town M (1999) Severity of phenotype in cystinosis varies with mutations in the CTNS gene: predicted effect on the model of cystinosin. Hum Mol Genet 8:2507–2514
Kalatzis V, Cohen-Solal L, Cordier B, Frishberg Y, Kemper M, Nuutinen EM, Legrand E, Cochat P, Antignac C (2002) Identification of 14 novel CTNS mutations and characterization of seven splice site mutations associated with cystinosis. Hum Mutat 20:439–446
Kersey PJ, Staines DM, Lawson D, Kulesha E, Derwent P, Humphrey JC, Hughes DS, Keenan S, Kerhornou A, Koscielny G, Langridge N, McDowall MD, Megy K, Maheswari U, Nuhn M, Paulini M, Pedro H, Toneva I, Wilson D, Yates A, Birney E (2011) Ensembl genomes: an integrative resource for genome-scale data from non-vertebrate species. Nucleic Acids Res 40(D1):D91–D97
Statistics South Africa (2012) South African 2011 population census. Available at: http://www.statssa.gov.za/Publications/P03014/P030142011.pdf
Henderson H, Leisegang F, Brown R, Eley B (2002) The clinical and molecular spectrum of galactosemia in patients from the Cape Town region of South Africa. BMC Pediatr 2:7
Arndt S, Heitner R, Lane A, Ramsay M (2009) Gluco cerebrosidase gene mutations in black South Africans with Gaucher disease. Blood Cells Mol Dis 43(1):129–33
Van der Watt G, Owen EP, Berman P, Meldau S, Watermeyer N, Olpin SE, Manning NJ, Baumgarten I, Leisegang F, Henderson H (2010) Glutaric aciduria type 1 in South Africa—high incidence of glutaryl-CoA dehydrogenase deficiency in black South Africans. Mol Genet Metab 101:178–182
Owen EP, Leisegang F, Whitelaw A, Simpson J, Baker S, Würzner R, Potter P, Orren A (2010) Complement component C5 and C6 mutation screening indicated in meningococcal disease in South Africa. S Afr Med J 102(6):525–527
Children’s Institute (2011) Statistics on children in South Africa. Available at: www.childrencount.ci.org.za
Acknowledgments
This study was supported by the MRC, University of Cape Town and the NHLS of South Africa. We thank all the patients and parents for their participation in the study.
Conflict of interest
None
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Owen, E.P., Nandhlal, J., Leisegang, F. et al. Common mutation causes cystinosis in the majority of black South African patients. Pediatr Nephrol 30, 595–601 (2015). https://doi.org/10.1007/s00467-014-2980-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-014-2980-7