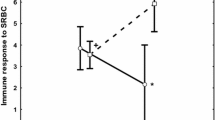
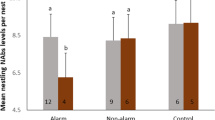
Abstract
Genetic color polymorphism is widespread in nature. There is an increasing interest in understanding the adaptive value of heritable color variation and trade-off resolution by differently colored individuals. Melanin-based pigmentation is often associated with variation in many different life history traits. These associations have recently been suggested to be the outcome of pleiotropic effects of the melanocortin system. Although pharmacological research supports that MC1R, a gene with a major role in vertebrate pigmentation, has important immunomodulatory effects, evidence regarding pleiotropy at MC1R in natural populations is still under debate. We experimentally assessed whether MC1R-based pigmentation covaries with both inflammatory and humoral immune responses in the color polymorphic Eleonora’s falcon. By means of a cross-fostering experiment, we disentangled potential genetic effects from environmental effects on the covariation between coloration and immunity. Variation in both immune responses was primarily due to genetic factors via the nestlings’ MC1R-related color genotype/phenotype, although environmental effects via the color morph of the foster father also had an influence. Overall, dark nestlings had lower immune responses than pale ones. The effect of the color morph of the foster father was also high, but in the opposite direction, and nestlings raised by dark eumelanic foster fathers had higher immune responses than those raised by pale foster fathers. Although we cannot completely discard alternative explanations, our results suggest that MC1R might influence immunity in this species. Morph-specific variation in immunity as well as pathogen pressure may therefore contribute to the long-term maintenance of genetic color polymorphism in natural populations.


Similar content being viewed by others
References
Adamo SA (2004) How should behavioural ecologists interpret measurements of immunity? Anim Behav 68(6):1443–1449
Ahmed TJ, Montero-Melendez T, Perretti M, Pitzalis C (2013) Curbing inflammation through endogenous pathways: focus on melanocortin peptides. Int J Inflamm 2013:1–10
Almasi B, Jenni L, Jenni-Eiermann S, Roulin A (2010) Regulation of stress response is heritable and functionally linked to melanin-based coloration. J Evol Biol 23:987–996. doi:10.1111/j.1420-9101.2010.01969.x
Alonso-Alvarez C, Tella JL (2001) Effects of experimental food restriction and body-mass changes on the avian T-cell-mediated immune response. Can J Zool 79:101–105. doi:10.1139/z00-190
Apanius V (1998) Ontogeny of the immune function. In: Starck JM, Ricklefs RE (eds) Avian growth and development. Oxford University Press, New York, pp 203–222
Ardia DR (2005) Tree swallows trade off immune function and reproductive effort differently across their range. Ecology 86(8):2040–2046
Arriero E, Majewska A, Martin TE (2013) Ontogeny of constitutive immunity: maternal vs. endogenous influences. Funct Ecol 27:472–478. doi:10.1111/1365-2435.12057
Baião PC, Schreiber EA, Parker PG (2007) The genetic basis of the plumage polymorphism in red-footed boobies (Sula sula): a melanocortin-1 receptor (MC1R) analysis. J Hered 98:287–292. doi:10.1093/jhered/esm030
Becher E, Mahnke K, Brzoska T, Kalden DH, Grabbe S, Luger TA (1999) Human peripheral blood-derived dendritic cells express functional melanocortin receptor MC-1R. Ann NY Acad Sci 885:188–195. doi:10.1111/j.1749-6632.1999.tb08676.x
Broggi J, García O, Miranda F, Pagés A, Soriguer RC, Figuerola J (2013) Immune response to newcastle disease virus vaccination in a wild passerine. J Wildl Dis 49:1004–1008. doi:10.7589/2012-10-266
Brommer JE (2004) Immunocompetence and its costs during development: an experimental study in blue tit nestlings. Proc R Soc Lond B Biol Sci 271:S110–S113. doi:10.1098/rsbl.2003.0103
Brzoska T, Luger TA, Maaser C, Abels C, Böhm M (2008) α-Melanocyte-Stimulating Hormone and related tripeptides: biochemistry, antiinflammatory and protective effects in vitro and in vivo, and future perspectives for the treatment of immune-mediated inflammatory diseases. Endocr Rev 29:581–602. doi:10.1210/er.2007-0027
Buggy JJ (1998) Binding of alpha-melanocyte-stimulating hormone to its G-protein-coupled receptor on B-lymphocytes activates the Jak/STAT pathway. Biochem J 331:211–216
Burnham KP, Anderson DR (2002) Model selection and multimodel inference: a practical information—theoretic approach, 2nd edn. Springer, New York
Catania A (2007) The melanocortin system in leukocyte biology. J Leukocyte Biol 81:383–392. doi:10.1189/jlb.0706426
Cooper A, Robinson SJ, Pickard C, Jackson CL, Friedmann PS, Healy E (2005) α-Melanocyte-stimulating hormone suppresses antigen-induced lymphocyte proliferation in humans independently of melanocortin 1 receptor gene status. J Immunol 175:4806–4813
Del Moral JC (2008) El halcón de Eleonora en España. Población en 2004–2007 y método de censo. SEO/BirdLife, Madrid
Dessinioti C, Antoniou C, Katsambas A, Stratigos AJ (2011) Melanocortin 1 receptor variants: functional role and pigmentary associations. Photochem Photobiol 87:978–987. doi:10.1111/j.1751-1097.2011.00970.x
Ducrest A-L, Keller L, Roulin A (2008) Pleiotropy in the melanocortin system, coloration and behavioural syndromes. Trends Ecol Evol 23:502–510. doi:10.1016/j.tree.2008.06.001
Eizirik E, Yuhki N, Johnson WE, Menotti-Raymond M, Hannah SS, O’Brien SJ (2003) Molecular genetics and evolution of melanism in the cat family. Curr Biol 13:448–453
Fargallo JA, Laaksonen T, Korpimäki E, Wakamatsu K (2007) A melanin-based trait reflects environmental growth conditions of nestling male Eurasian kestrels. Evol Ecol 21:157–171
Fellah JS, Jaffredo T, Dunon D (2008) Development of the avian immune system. In: Davison F, Kaspers B, Schat KA (eds) Avian immunology. Academic Press, London, pp 51–66
Fox J, Weisberg S (2011) An R companion to applied regression, 2nd edn. Sage, Thousand Oaks
Galván I, Gangoso L, Grande JM, Negro JJ, Rodríguez A, Figuerola J, Alonso-Alvarez C (2010) Antioxidant machinery differs between melanic and light nestlings of two polymorphic raptors. PLoS ONE 5:e13369. doi:10.1371/journal.pone.0013369
Gangoso L, Grande JM, Ducrest A-L, Figuerola J, Bortolotti GR, Andrés JA, Roulin A (2011) MC1R-dependent, melanin-based colour polymorphism is associated with cell-mediated response in the Eleonora’s falcon. J Evol Biol 24:2055–2063. doi:10.1111/j.1420-9101.2011.02336.x
Gangoso L, López-López P, Grande JM, Mellone U, Limiñana R, Urios V, Ferrer M (2013) Ecological specialization to fluctuating resources prevents long-distance migratory raptors from becoming sedentary on islands. PLoS ONE 8(4):e61615. doi:10.1371/journal.pone.0061615
Getting SJ, Allcock GH, Flower R, Perretti M (2001) Natural and synthetic agonists of the melanocortin receptor type 3 possess anti-inflammatory properties. J Leukocyte Biol 69:98–104
Grace JB (2006) Structural equation modeling and natural systems. Cambridge University Press, Cambridge
Gross JB, Borowsky R, Tabin CJ (2009) A novel role for Mc1r in the parallel evolution of depigmentation in independent populations of the cavefish Astyanax mexicanus. PLoS Gen 5:e1000326. doi:10.1371/journal.pgen.1000326
Hasselquist D, Nilsson J-Å (2009) Maternal transfer of antibodies in vertebrates: trans-generational effects on offspring immunity. Philos Trans R Soc B 364:51–60. doi:10.1098/rstb.2008.0137
Hawley DM, Jennelle CS, Sydenstricker KV, Dhondt AA (2007) Pathogen resistance and immunocompetence covary with social status in house finches (Carpodacus mexicanus). Funct Ecol 21(3):520–527
Hõrak P, Zilmer M, Saks L, Ots I, Karu U, Zilmer K (2006) Antioxidant protection, carotenoids and the costs of immune challenge in greenfinches. J Exp Biol 209:4329–4338. doi:10.1242/jeb.02502
Jacquin L, Blottiere L, Haussy C, Perret S, Gasparini J (2012) Prenatal and postnatal parental effects on immunity and growth in ‘lactating’ pigeons. Funct Ecol 26(4):866–875
Kim SY, Fargallo JA, Vergara P, Martínez-Padilla J (2013) Multivariate heredity of melanin-based coloration, body mass and immunity. Heredity 111(2):139–146. doi:10.1038/hdy.2013.29
King MO, Owen JP, Schwabl HG (2010) Are maternal antibodies really that important? Patterns in the immunologic development of altricial passerine house sparrows (Passer domesticus). PLoS ONE 5:e9639. doi:10.1371/journal.pone.0009639
Klasing KC, Leshchinsky TV (1999) Functions, costs, and benefits of the immune system during development. In: Adams NJ, Slotow RH (eds) Proceedings of the 22nd International Ornithological Congress. BirdLife South Africa, Johannesburg, pp 2817–2835
Lazzaro BP, Little TJ (2009) Immunity in a variable world. Philos Trans R Soc B 364(1513):15–26. doi:10.1098/rstb.2008.0141
Lin JY, Fisher DE (2007) Melanocyte biology and skin pigmentation. Nature 445:843–850. doi:10.1038/nature05660
Loser K, Brzoska T, Oji V, Auriemma M, Voskort M, Kupas V, Klenner L, Mensing C, Hauschild A, Beissert S, Luger TA (2010) The neuropeptide alpha-melanocyte-stimulating hormone is critically involved in the development of cytotoxic CD8+ T cells in mice and humans. PLoS ONE 5:e8958. doi:10.1371/journal.pone.0008958
Maaser C, Kannengiesser K, Kucharzik T (2006) Role of the melanocortin system in inflammation. Ann NY Acad Sci 1072:123–134. doi:10.1196/annals.1326.016
Martin LB II, Han P, Lewittes J, Kuhlman JR, Klasing KC, Wikelski M (2006) Phytohemagglutinin-induced skin swelling in birds: histological support for a classic immunoecological technique. Funct Ecol 20:290–299. doi:10.1111/j.1365-2435.2006.01094.x
Matson KD, Cohen AA, Klasing KC, Ricklefs RE, Scheuerlein A (2006) No simple answers for ecological immunology: relationships among immune indices at the individual level break down at the species level in waterfowl. Proc R Soc Lond B Biol Sci 273:815–822. doi:10.1098/rspb.2005.3376
Mundy NI (2005) A window on the genetics of evolution: MC1R and plumage colouration in birds. Proc R Soc Lond B Biol Sci 272:1633–1640. doi:10.1098/rspb.2005.3107
Mundy NI, Badcock NS, Hart T, Scribner K, Nadeau NJ (2004) Conserved genetic basis of a quantitative plumage trait involved in mate choice. Science 303:1870–1873. doi:10.1126/science.1093834
Neumann Andersen G, Nagaeva O, Mandrika I, Petrovska R, Muceniece R, Mincheva-Nilsson L, Wikberg JES (2001) MC1 receptors are constitutively expressed on leucocyte subpopulations with antigen presenting and cytotoxic functions. Clin Exp Immunol 126:441–446. doi:10.1046/j.1365-2249.2001.01604.x
Norris K, Evans MR (2000) Ecological immunology: life history trade-offs and immune defense in birds. Behav Ecol 11:19–26. doi:10.1093/beheco/11.1.19
Oberski D (2014) Lavaan survey: an R package for complex survey analysis of structural equation models. J Stat Softw 57:1–27
Office International de Epizooties (OIE) (2000) Manuals of standards for diagnostic tests and vaccines, 4th edn. OIE, Paris
Palacios MG, Cunnick JE, Vleck D, Vleck CM (2009) Ontogeny of innate and adaptive immune defense components in free-living tree swallows, Tachycineta bicolor. Dev Comp Immunol 33:456–463. doi:10.1016/j.dci.2008.09.006
Piault R, Gasparini J, Bize P, Jenni-Eiermann S, Roulin A (2009) Pheomelanin-based coloration and the ability to cope with variation in food supply and parasitism. Am Nat 174:548–556. doi:10.1086/605374
Pigliucci M, Preston K (2004) Phenotypic integration. Studying the ecology and evolution of complex phenotypes. University Press, Oxford
Pitala N, Siitari H, Gustafsson L, Brommer JE (2010) Costs and benefits of experimentally induced changes in the allocation of growth versus immune function under differential exposure to ectoparasites. PLoS ONE 5:e10814. doi:10.1371/journal.pone.0010814
Py I, Ducrest A, Duvoisin N, Fumagalli L, Roulin A (2006) Ultraviolet reflectance in a melanin-based plumage trait is heritable. Evol Ecol Res 8:483–491
R Core Team (2013) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Råberg L, Stjernman M (2003) Natural selection on immune responsiveness in blue tits Parus caeruleus. Evolution 57:1670–1678. doi:10.1111/j.0014-3820.2003.tb00372.x
Reissmann M, Ludwig A (2013) Pleiotropic effects of coat colour-associated mutations in humans, mice and other mammals. Semin Cell Dev Biol 24(6):576–586. doi:10.1016/j.semcdb.2013.03.014
Rennalls LVP, Seidl T, Larkin JMG, Wellbrock C, Gore ME, Eisen T, Bruno L (2010) The melanocortin receptor agonist NDP-MSH impairs the allostimulatory function of dendritic cells. Immunology 129:610–619. doi:10.1111/j.1365-2567.2009.03210.x
Ristow D, Wink M (2004) Seasonal variation in sex ratio of nestling Eleonora’s falcons. J Raptor Res 38:320–325
Ristow D, Wink C, Wink M, Scharlau W (1998) Colour polymorphism in Eleonora’s falcon Falco eleonorae. Sandgrouse 20:56–64
Rodriguez S, Gaunt TR, Day INM (2009) Hardy–Weinberg equilibrium testing of biological ascertainment for Mendelian randomization studies. Am J Epidemiol. doi:10.1093/aje/kwn359
Roitt IM, Brostoff J, Male D (1998) Immunology. Mosby, London
Rosenblum EB, Römpler H, Schöneberg T, Hoekstra HE (2010) Molecular and functional basis of phenotypic convergence in white lizards at White Sands. Proc Natl Acad Sci USA 107:2113–2117. doi:10.1073/pnas.0911042107
Rosseel Y (2012) lavaan: an R package for structural equation modeling. J Stat Softw 48:1–36
Roulin A, Ducrest A-L (2011) Association between melanism, physiology and behaviour: a role for the melanocortin system. Eur J Pharmacol 660:226–233. doi:10.1016/j.ejphar.2011.01.036
Roulin A, Ducrest A-L (2013) Genetics of colouration in birds. Semin Cell Dev Biol 24:594–608
Roulin A, Jungi TW, Pfister H, Dijkstra C (2000) Female barn owls (Tyto alba) advertise good genes. Proc R Soc Lond B Biol Sci 267(1446):937–941
Roulin A, Ducret B, Ravussin P-A, Altwegg R (2003) Female colour polymorphism covaries with reproductive strategies in the tawny owl Strix aluco. J Avian Biol 34:393–401. doi:10.1111/j.0908-8857.2003.03139.x
Roulin A, Almasi B, Meichtry-Stier KS, Jenni L (2011) Eumelanin- and pheomelanin-based colour advertise resistance to oxidative stress in opposite ways. J Evol Biol 24:2241–2247. doi:10.1111/j.1420-9101.2011.02353.x
Schmid-Hempel P (2003) Variation in immune defence as a question of evolutionary ecology. Proc R Soc Lond B Biol Sci 270(1513):357–366. doi:10.1098/rspb.2002.2265
Smits JE, Bortolotti GR, Tella JL (1999) Simplifying the phytohaemagglutinin skin-testing technique in studies of avian immunocompetence. Funct Ecol 13:567–572. doi:10.1046/j.1365-2435.1999.00338.x
Taylor AW, Namba K (2001) In vitro induction of CD25+ CD4+ regulatory T cells by the neuropeptide alpha-melanocyte stimulating hormone (α-MSH). Immunol Cell Biol 79:358–367. doi:10.1046/j.1440-1711.2001.01022.x
Teofoli P, Frezzolini A, Puddu P, De Pità O, Mauviel A, Lotti T (1999) The role of proopiomelanocortin-derived peptides in skin fibroblast and mast cell functions. Ann NY Acad Sci 885:268–276
Theron E, Hawkins K, Bermingham E, Ricklefs RE, Mundy NI (2001) The molecular basis of an avian plumage polymorphism in the wild: a melanocortin-1-receptor point mutation is perfectly associated with the melanic plumage morph of the bananaquit, Coereba flaveola. Curr Biol 11:550–557
Viney ME, Riley EM, Buchanan KL (2005) Optimal immune responses: immunocompetence revisited. Trends Ecol Evol 20:665–669. doi:10.1016/j.tree.2005.10.003
Vinkler M, Bainová H, Albrecht T (2010) Functional analysis of the skin-swelling response to phytohaemagglutinin. Funct Ecol 24:1081–1086. doi:10.1111/j.1365-2435.2010.01711.x
Walter H (1979) Eleonora’s falcon. Adaptations to prey and habitat in a social raptor. University of Chicago Press, Chicago
Wikberg JES, Mutulis F (2008) Targeting melanocortin receptors: an approach to treat weight disorders and sexual dysfunction. Nat Rev Drug Discov 7:307–323. doi:10.1038/nrd2331
Wink C, Ristow D (2000) Biology and molecular genetics of Eleonora’s falcon Falco eleonorae, a colonial raptor of Mediterranean islands. In: Chancellor RD, Meyburg BU (eds) Raptors at risk. WWGBP, Hancock House, Blaine, WA, pp 653–668
Wink M, Wink C, Ristow D (1978) Biology of Eleonora’s falcon Falco eleonorae. Part 2. Heredity of the dark and light phases. J Ornithol 119:421–428
Wittkopp PJ, Beldade P (2009) Development and evolution of insect pigmentation: genetic mechanisms and the potential consequences of pleiotropy. Semin Cell Dev Biol 20:65–71
Acknowledgments
We are especially grateful to J. J. Moreno and J. L. Barroso, as well as to I. Henry and C. Dufresnes, for their invaluable help during the fieldwork. During the writing of this manuscript, L. Gangoso was supported by the FP7-REGPOT 2010-1 EcoGenes Project (grant no. 264125) and A. Roulin was supported by the Swiss National Science Foundation (grant no. 3100AO_120517).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Markku Orell.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gangoso, L., Roulin, A., Ducrest, AL. et al. Morph-specific genetic and environmental variation in innate and acquired immune response in a color polymorphic raptor. Oecologia 178, 1113–1123 (2015). https://doi.org/10.1007/s00442-015-3306-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-015-3306-6