Abstract
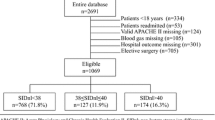
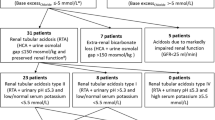
Acid–base disturbances have been usually evaluated with the traditional Henderson–Hasselbach method and Stewart’s physiochemical approach by quantifying anions of tissue acids (TA). It is hypothesized that an increase in tissue acids during metabolic acidosis would cause a compensatory decrease in the plasma chloride (Cl) relative to sodium (Cl–Na ratio) in order to preserve electroneutral balance. Therefore, we aimed to investigate the use of Cl–Na ratio as a bedside tool to evaluate the identifying raised TA in neonates as an alternative to complex calculations of Stewart’s physiochemical approach. This retrospective study was conducted between January 2008 and December 2009. Infants were included in the study when blood gas analysis reveals a metabolic acidosis; pH <7.25 and sHCO3 concentration was <22 mEq/L. The Cl–Na ratio, sodium–chloride difference (DiffNaCl), anion gap (AG), albumin-corrected AG (AGcorr), strong ion difference (SID), unmeasured anions (UMA), and TA were calculated at each episode of metabolic acidosis. A total of 105 metabolic acidosis episodes occurred in 59 infants during follow-up. Hypochloremic metabolic acidosis occurred in 17 (16%) of samples, and all had increased TA. The dominant component of TA was UMA rather than lactate. There was a negative correlation between the Cl–Na ratio and SID, AGcorr, UMA, and TA. Also, there was a positive correlation between DiffNaCl and SID, AGcorr, UMA, and TA. Base deficit and actual bicarbonate performed poorly in identifying the TA. In conclusion, our study suggested that DiffNaCl and Cl–Na ratio are simple and fast, and may be an alternative method to complex Stewart’s physiochemical approach in identifying raised UMA and TA in critically ill neonates.


Similar content being viewed by others
References
Aschner JL, Poland RL (2008) Sodium bicarbonate: basically useless therapy. Pediatrics 122:831–835
Balasubramanyan N, Havens PL, Hoffman GM (1999) Unmeasured anions identified by the Fencl Stewart method predict mortality better than base excess, anion gap, and lactate in patients in the pediatric intensive care unit. Crit Care Med 27:1577–1581
Boyle M, Lawrence J (2003) An easy method of mentally estimating the metabolic component of acid/base balance using the Fencl–Stewart approach. Anaesth Intensive Care 31:538–547
Durward A, Skellet S, Mayer A, Taylor D, Tibby SM, Murdoch IA (2001) The value of the chloride–sodium ratio in differentiating the aetiology of metabolic acidosis. Intensive Care Med 27:828–835
Durward A, Murdoch I (2003) Understanding acid–base balance. Current Paediatrics 13:513–519
Durward A, Tibby SM, Skellet S, Austin C, Anderson D, Murdoch IA (2005) The strong ion gap predicts mortality in children following cardiopulmonary bypass surgery. Pediatr Crit Care Med 6:281–285
Fencl V, Leith DE (1993) Stewart’s quantitative acid–base chemistry: applications in biology and medicine. Respir Physiol 91:1–16
Fidkowski C, Helstrom J (2009) Diagnosing metabolic acidosis in the critically ill: bridging the anion gap, Stewart, and base excess methods. Can J Anesth 56:247–256
Figge J, Mydosh T, Fencle V (1992) Serum proteins and acid–base equilibria: a follow up. J Lab Clin Med 120:713–719
Figge J, Jabor A, Kazda A, Fencl V (1998) Anion gap and hypoalbuminemia. Crit Care Med 26:1807–1810
Kellum JA (1998) Metabolic acidosis in the critically ill: lessons from physical chemistry. Kidney Int Suppl 66:81–86
Lawn CJ, Weir FJ, McGuire W (2005) Base administration or fluid bolus for preventing morbidity and mortality in preterm infants with metabolic acidosis. Cochrane Database Syst Rev 2:CD003215
Morris CG, Low J (2008) Metabolic acidosis in the critically ill: part 1. Classification and pathophysiology. Anaesthesia 63:294–301
Moviat M, van Haren F, van der Hoeven H (2003) Conventional or physiochemical approach in intensive care unit patients with metabolic acidosis. Crit Care 7:R41–R45
Moviat M, Terpstra AM, Ruitenbeek W, Kluijtmans LA, Pickkers P, van der Hoeven JG (2008) Contribution of various metabolites to the “unmeasured” anions in critically ill patients with metabolic acidosis. Crit Care Med 36:752–758
Murray DM, Olhsson V, Fraser JI (2004) Defining acidosis in postoperative cardiac patients using Stewart’s method of strong ion difference. Pediatr Crit Care Med 5:240–245
Nagaoka D, Nassar AP, Maciel AT, Taniguchi LU, Noritomi DT, Azevedo LCP, da Cruz Neto LM, Park M (2010) The use of sodium–chloride difference and chloride–sodium ratio as strong ion difference surrogates in the evaluation of metabolic acidosis in critically ill patients. J Crit Care 55:525–531
Quintard H, Hubert S, Ichai C (2007) What is the contribution of Stewart’s concept in acid–base disorders analysis? Ann Fr Anesth Reanim 26:423–433
Severinghaus JW (1993) Siggard–Andersen and the great trans-Atlantic acid–base debate. Scand J Clin Lab Invest 53:99–104
Stewart PA (1983) Modern quantitative acid–base chemistry. Can J Physiol Pharmacol 61:1444–1461
Story DA, Morimatsu H, Bellomo R (2004) Strong ions, weak acids and base excess: a simplified Fencl–Stewart approach to clinical acid–base disorders. Br J Anaesth 92:54–60
Acknowledgment
This study was not supported by drug companies and there is no potential source.
Conflict of interest
We have no a financial relationship with the organization that sponsored the research.
Author information
Authors and Affiliations
Corresponding author
Additional information
For the study, written permit were taken from each of the families of the newborns, as well as an approval of the regional Ethic Committee.
Rights and permissions
About this article
Cite this article
Kurt, A., Ecevit, A., Ozkiraz, S. et al. The use of chloride–sodium ratio in the evaluation of metabolic acidosis in critically ill neonates. Eur J Pediatr 171, 963–969 (2012). https://doi.org/10.1007/s00431-011-1666-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00431-011-1666-4