Abstract
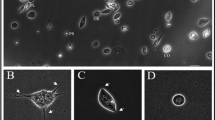
Anaplasma phagocytophilum (Ap) is a tick-borne pathogen, which can cause granulocytic anaplasmosis in humans and animals. In vivo this obligate intracellular pathogen is primarily located in circulating mature granulocytes, but it also infects endothelial cells. In order to study the interaction between Ap-infected endothelial cells and human granulocytes under conditions similar to those found naturally in the infected host, an in vitro model that mimics physiological flow conditions in the microvasculature was established. Cell-to-cell interactions were then visualized by microscopy, which showed that granulocytes adhered strongly to Ap-infected endothelial cells at a shear stress of 0.5 dyne/cm2. In addition, Ap-transmission assays under flow conditions showed that the bacteria transferred from infected endothelial cells to circulating granulocytes and were able to establish infection in constantly moving granulocytes. Cell surface analysis showed that Ap induced up-regulation of the cell adhesion molecules ICAM-1 and VCAM-1 on infected endothelial cells in a dose-dependent manner. Furthermore, IL-8 secretion by endothelial cells indicated that the presence of Ap induced a pro-inflammatory response. In summary, the results of this study suggest that endothelial cells of the microvasculature (1) provide an excellent site for Ap dissemination to peripheral blood granulocytes under flow conditions and therefore may play a crucial role in the development of persistent infection, and (2) are stimulated by Ap to express surface molecules and cytokines that may lead to inflammatory responses at the site of the infection.





Similar content being viewed by others
References
Ades EW, Candal FJ, Swerlick RA et al (1992) HMEC-1: establishment of an immortalized human microvascular endothelial cell line. J Invest Dermatol 99:683–690
Akkoyunlu M, Malawista SE, Anguita J et al (2001) Exploitation of interleukin-8-induced neutrophil chemotaxis by the agent of human granulocytic ehrlichiosis. Infect Immun 69:5577–5588
Arnez M, Petrovec M, Lotric-Furlan S et al (2001) First European pediatric case of human granulocytic ehrlichiosis. J Clin Microbiol 39:4591–4592
Bakken JS, Dumler S (2008) Human granulocytic anaplasmosis. Infect Dis Clin N Am 22:433–448
Bechah Y, Capo C, Raoult D et al (2008) Infection of endothelial cells with virulent Rickettsia prowazekii increases the transmigration of leukocytes. J Infect Dis 197:142–147
Bevilacqua MP, Pober JS, Wheeler ME et al (1985) Interleukin 1 acts on cultured human vascular endothelium to increase the adhesion of polymorphonuclear leukocytes, monocytes, and related leukocyte cell lines. J Clin Invest 76:2003–2011
Bjoersdorff A, Bagert B, Massung RF et al (2002) Isolation and characterization of two European strains of Ehrlichia phagocytophila of equine origin. Clin Diagn Lab Immunol 9:341–343
Bjoersdorff A, Svendenius L, Owens JH et al (1999) Feline granulocytic ehrlichiosis–a report of a new clinical entity and characterisation of the infectious agent. J Small Anim Pract 40:20–24
Borjesson DL (2008) Culture, isolation, and labeling of Anaplasma phagocytophilum for subsequent infection of human neutrophils. Methods Mol Biol 431:159–171
Brown CC, Skowronek AJ (1990) Histologic and immunochemical study of the pathogenesis of heartwater (Cowdria ruminantium infection) in goats and mice. Am J Vet Res 51:1476–1480
Carreno AD, Alleman AR, Barbet AF et al (2007) In vivo endothelial cell infection by Anaplasma marginale. Vet Pathol 44:116–118
Chiu JJ, Lee PL, Chang SF et al (2005) Shear stress regulates gene expression in vascular endothelial cells in response to tumor necrosis factor-alpha: a study of the transcription profile with complementary DNA microarray. J Biomed Sci 12:481–502
Coling D, Kachar B (2001) Principles and application of fluorescence microscopy. Curr Protoc Mol Biol Unit 14:10
Collins SJ, Ruscetti FW, Gallagher RE et al (1979) Normal functional characteristics of cultured human promyelocytic leukemia cells (HL-60) after induction of differentiation by dimethylsulfoxide. J Exp Med 149:969–974
Collins SJ, Ruscetti FW, Gallagher RE et al (1978) Terminal differentiation of human promyelocytic leukemia cells induced by dimethyl sulfoxide and other polar compounds. Proc Natl Acad Sci USA 75:2458–2462
English D, Andersen BR (1974) Single-step separation of red blood cells. Granulocytes and mononuclear leukocytes on discontinuous density gradients of Ficoll-Hypaque. J Immunol Methods 5:249–252
Epperson DE, Pober JS (1994) Antigen-presenting function of human endothelial cells. Direct activation of resting CD8 T cells. J Immunol 153:5402–5412
Felsheim RF, Herron MJ, Nelson CM et al (2006) Transformation of Anaplasma phagocytophilum. BMC Biotechnol 6:42
Goodman JL, Nelson CM, Klein MB et al (1999) Leukocyte infection by the granulocytic ehrlichiosis agent is linked to expression of a selectin ligand. J Clin Invest 103:407–412
Granquist EG, Aleksandersen M, Bergstrom K et al (2010) A morphological and molecular study of Anaplasma phagocytophilum transmission events at the time of Ixodes ricinus tick bite. Acta Vet Scand 52:43
Herron MJ, Ericson ME, Kurtti TJ et al (2005) The interactions of Anaplasma phagocytophilum, endothelial cells, and human neutrophils. Ann N Y Acad Sci 1063:374–382
Herron MJ, Nelson CM, Larson J et al (2000) Intracellular parasitism by the human granulocytic ehrlichiosis bacterium through the P-selectin ligand, PSGL-1. Science 288:1653–1656
Herter J, Zarbock A (2013) Integrin regulation during leukocyte recruitment. J Immunol 190:4451–4457
Kaplanski G, Teysseire N, Farnarier C et al (1995) IL-6 and IL-8 production from cultured human endothelial cells stimulated by infection with Rickettsia conorii via a cell-associated IL-1 alpha-dependent pathway. J Clin Invest 96:2839–2844
Kobayashi Y (2008) The role of chemokines in neutrophil biology. Front Biosci 13:2400–2407
Kolaczkowska E, Kubes P (2013) Neutrophil recruitment and function in health and inflammation. Nat Rev Immunol 13:159–175
Kucik DF (2009) Measurement of adhesion under flow conditions. Curr Protoc Cell Biol Unit 9:6
Langer HF, Chavakis T (2009) Leukocyte-endothelial interactions in inflammation. J Cell Mol Med 13:1211–1220
Lee HC, Kioi M, Han J et al (2008) Anaplasma phagocytophilum-induced gene expression in both human neutrophils and HL-60 cells. Genomics 92:144–151
Lepidi H, Bunnell JE, Martin ME et al (2000) Comparative pathology, and immunohistology associated with clinical illness after Ehrlichia phagocytophila-group infections. Am J Trop Med Hyg 62:29–37
Munderloh UG, Lynch MJ, Herron MJ et al (2004) Infection of endothelial cells with Anaplasma marginale and A. phagocytophilum. Vet Microbiol 101:53–64
Nathan C (2006) Neutrophils and immunity: challenges and opportunities. Nat Rev Immunol 6:173–182
Nelson CM, Herron MJ, Felsheim RF et al (2008) Whole genome transcription profiling of Anaplasma phagocytophilum in human and tick host cells by tiling array analysis. BMC Genomics 9:364
Razakandrainibe R, Combes V, Grau GE et al (2013) Crossing the wall: the opening of endothelial cell junctions during infectious diseases. Int J Biochem Cell Biol 45:1165–1173
Reneman RS, Hoeks AP (2008) Wall shear stress as measured in vivo: consequences for the design of the arterial system. Med Biol Eng Comput 46:499–507
Rikihisa Y (2010) Anaplasma phagocytophilum and Ehrlichia chaffeensis: subversive manipulators of host cells. Nat Rev Microbiol 8:328–339
Rothermel AL, Wang Y, Schechner J et al (2004) Endothelial cells present antigens in vivo. BMC Immunol 5:5
Saverymuttu SH, Peters AM, Keshavarzian A et al (1985) The kinetics of 111indium distribution following injection of 111indium labelled autologous granulocytes in man. Br J Haematol 61:675–685
Schmidt S, Moser M, Sperandio M (2013) The molecular basis of leukocyte recruitment and its deficiencies. Mol Immunol 55:49–58
Sultana H, Neelakanta G, Kantor FS et al (2010) Anaplasma phagocytophilum induces actin phosphorylation to selectively regulate gene transcription in Ixodes scapularis ticks. J Exp Med 207:1727–1743
Thomas RJ, Birtles RJ, Radford AD et al (2012) Recurrent bacteraemia in sheep infected persistently with Anaplasma phagocytophilum. J Comp Pathol 147:360–367
Troese MJ, Carlyon JA (2009) Anaplasma phagocytophilum dense-cored organisms mediate cellular adherence through recognition of human P-selectin glycoprotein ligand 1. Infect Immun 77:4018–4027
Walker DH, Valbuena GA, Olano JP (2003) Pathogenic mechanisms of diseases caused by Rickettsia. Ann N Y Acad Sci 990:1–11
Walpola PL, Gotlieb AI, Langille BL (1993) Monocyte adhesion and changes in endothelial cell number, morphology, and F-actin distribution elicited by low shear stress in vivo. Am J Pathol 142:1392–1400
Witko-Sarsat V, Rieu P, Descamps-Latscha B et al (2000) Neutrophils: molecules, functions and pathophysiological aspects. Lab Invest 80:617–653
Woldehiwet Z (2010) The natural history of Anaplasma phagocytophilum. Vet Parasitol 167:108–122
Zen K, Liu Y, Mccall IC et al (2005) Neutrophil migration across tight junctions is mediated by adhesive interactions between epithelial coxsackie and adenovirus receptor and a junctional adhesion molecule-like protein on neutrophils. Mol Biol Cell 16:2694–2703
Zweygarth E, Josemans AI, Steyn HC (2008) In vitro isolation of Ehrlichia ruminantium from ovine blood into Ixodes scapularis (IDE8) cell cultures. Onderstepoort J Vet Res 75:121–126
Acknowledgments
The authors thank Anke Schiller and Stephanie Hiereth for their assistance in cell and bacterial culture; Dr. Silke Kadlez-Gebhardt, Dr. Winfried Kapfhammer and Dr. Vollert Lüthje for collecting blood samples from our donors; and Dr. Sonja Härtle for her useful discussion about flow cytometric data analysis. The authors thank Dr. Evelyn Overzier and Dr. Anna Rettinger for their careful reading of our manuscript. J.W. is a research fellow of the China Scholarship Council (CSC).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, J., Dyachenko, V., Munderloh, U.G. et al. Transmission of Anaplasma phagocytophilum from endothelial cells to peripheral granulocytes in vitro under shear flow conditions. Med Microbiol Immunol 204, 593–603 (2015). https://doi.org/10.1007/s00430-015-0387-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00430-015-0387-0