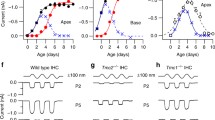
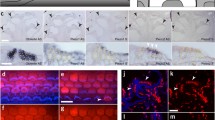
Abstract
Mutations of the transmembrane channel-like 1 (TMC1) gene can cause dominant and recessive forms of deafness in humans and mice. TMC1 is one of eight mammalian TMC genes of unknown function. The multi-pass transmembrane topologic structure of the proteins they encode suggests roles as a receptor, transporter, channel, or pump. Tmc1 and the closely related Tmc2 gene are expressed in neurosensory hair cells of the auditory and vestibular end organs of the mouse inner ear. Recent studies have demonstrated that Tmc1 and Tmc2 are specifically required for mechanoelectrical transduction in hair cells. The exact role of these proteins in mechanoelectrical transduction is unknown. TMC1 and TMC2 are viable candidates for the mechanoelectrical transduction channel of hair cells, whose component molecules have eluded identification for over 30 years. We expect that studies of TMC proteins will yield insights into molecular components and mechanisms of mechanosensation in auditory and vestibular hair cells, as well as in other tissues and organs.








Similar content being viewed by others
References
Ahmed ZM, Goodyear R, Riazuddin S, Lagziel A, Legan PK, Behra M, Burgess SM, Lilley KS, Wilcox ER, Griffith AJ, Frolenkov GI, Belyantseva IA, Richardson GP, Friedman TB (2006) The tip-link antigen, a protein associated with the transduction complex of sensory hair cells, is protocadherin-15. J Neurosci 26(26):7022–7034. doi:10.1523/JNEUROSCI.1163-06.2006
Beurg M, Fettiplace R, Nam JH, Ricci AJ (2009) Localization of inner hair cell mechanotransducer channels using high-speed calcium imaging. Nat Neurosci 12(5):553–558. doi:10.1038/nn.2295
Beurg M, Kim KX, Fettiplace R (2014) Conductance and block of hair-cell mechanotransducer channels in transmembrane channel-like protein mutants. J Gen Physiol 144(1):55–69. doi:10.1085/jgp.201411173
Chatzigeorgiou M, Bang S, Hwang SW, Schafer WR (2013) tmc-1 encodes a sodium-sensitive channel required for salt chemosensation in C. elegans. Nature 494(7435):95–99. doi:10.1038/nature11845
de Heer A-MR, Collin RWJ, Huygen PLM, Schraders M, Oostrik J, Rouwette M, Kunst HPM, Kremer H, Cremers CWRJ (2011) Progressive sensorineural hearing loss and normal vestibular function in a Dutch DFNB7/11 family with a novel mutation in TMC1. Audiol Neurotol 16:93–105. doi:10.1159/000313282
Fortnum HM, Summerfield AQ, Marshall DH, Davis AC, Bamford JM (2001) Prevalence of permanent childhood hearing impairment in the United Kingdom and implications for universal neonatal hearing screening: questionnaire based ascertainment study. BMJ 323(7312):536–540. doi:10.1136/bmj.323.7312.536
Geleoc GS, Holt JR (2003) Developmental acquisition of sensory transduction in hair cells of the mouse inner ear. Nat Neurosci 6(10):1019–1020. doi:10.1038/nn1120
Gillespie PG, Muller U (2009) Mechanotransduction by hair cells: models, molecules, and mechanisms. Cell 139(1):33–44. doi:10.1016/j.cell.2009.09.010
Griffith AJ, Friedman TB (2002) Autosomal and X-linked auditory disorders. Springer, New York
Holt JR, Pan B, Koussa MA, Asai Y (2014) TMC function in hair cell transduction. Hear Res. doi:10.1016/j.heares.2014.01.001
Hudspeth AJ (1982) Extracellular current flow and the site of transduction by vertebrate hair cells. J Neurosci 2(1):1–10
Jaramillo F, Hudspeth AJ (1991) Localization of the hair cell's transduction channels at the hair bundle’s top by iontophoretic application of a channel blocker. Neuron 7(3):409–420. doi:10.1016/0896-6273(91)90293-9
Kawashima Y, Geleoc GS, Kurima K, Labay V, Lelli A, Asai Y, Makishima T, Wu DK, Della Santina CC, Holt JR, Griffith AJ (2011) Mechanotransduction in mouse inner ear hair cells requires transmembrane channel-like genes. J Clin Invest 121(12):4796–4809. doi:10.1172/JCI60405
Kazmierczak P, Sakaguchi H, Tokita J, Wilson-Kubalek EM, Milligan RA, Muller U, Kachar B (2007) Cadherin 23 and protocadherin 15 interact to form tip-link filaments in sensory hair cells. Nature 449(7158):87–91. doi:10.1038/nature06091
Keresztes G, Mutai H, Heller S (2003) TMC and EVER genes belong to a larger novel family, the TMC gene family encoding transmembrane proteins. BMC Genomics 4(1):24. doi:10.1186/1471-2164-4-24
Kim KX, Beurg M, Hackney CM, Furness DN, Mahendrasingam S, Fettiplace R (2013) The role of transmembrane channel-like proteins in the operation of hair cell mechanotransducer channels. J Gen Physiol. doi:10.1085/jgp.201311068
Kim KX, Fettiplace R (2013) Developmental changes in the cochlear hair cell mechanotransducer channel and their regulation by transmembrane channel-like proteins. J Gen Physiol 141(1):141–148. doi:10.1085/jgp.201210913
Kitajiri S, Makishima T, Friedman TB, Griffith AJ (2007) A novel mutation at the DFNA36 hearing loss locus reveals a critical function and potential genotype-phenotype correlation for amino acid-572 of TMC1. Clin Genet 71(2):148–152. doi:10.1111/j.1399-0004.2007.00739.x
Kurima K, Peters LM, Yang Y, Riazuddin S, Ahmed ZM, Naz S, Arnaud D, Drury S, Mo J, Makishima T, Ghosh M, Menon PS, Deshmukh D, Oddoux C, Ostrer H, Khan S, Deininger PL, Hampton LL, Sullivan SL, Battey JF Jr, Keats BJ, Wilcox ER, Friedman TB, Griffith AJ (2002) Dominant and recessive deafness caused by mutations of a novel gene, TMC1, required for cochlear hair-cell function. Nat Genet 30(3):277–284. doi:10.1038/ng842
Kurima K, Yang Y, Sorber K, Griffith AJ (2003) Characterization of the transmembrane channel-like (TMC) gene family: functional clues from hearing loss and epidermodysplasia verruciformis. Genomics 82(3):300–308. doi:10.1016/S0888-7543(03)00154-X
Labay V, Weichert RM, Makishima T, Griffith AJ (2010) Topology of Transmembrane Channel-like Gene 1 Protein. Biochemistry 49:8592–8598. doi:10.1021/bi1004377
Lagziel A, Ahmed ZM, Schultz JM, Morell RJ, Belyantseva IA, Friedman TB (2005) Spatiotemporal pattern and isoforms of cadherin 23 in wild type and waltzer mice during inner ear hair cell development. Dev Biol 280(2):295–306. doi:10.1016/j.ydbio.2005.01.015
Lelli A, Asai Y, Forge A, Holt JR, Geleoc GS (2009) Tonotopic gradient in the developmental acquisition of sensory transduction in outer hair cells of the mouse cochlea. J Neurophysiol 101(6):2961–2973. doi:10.1152/jn.00136
Makishima T, Kurima K, Brewer CC, Griffith AJ (2004) Early onset and rapid progression of dominant nonsyndromic DFNA36 hearing loss. Otol Neurotol 25(5):714–719
Marazita ML, Ploughman LM, Rawlings B, Remington E, Arnos KS, Nance WE (1993) Genetic epidemiological studies of early-onset deafness in the U.S. school-age population. Am J Med Genet 46(5):486–491. doi:10.1002/ajmg.1320460504
Marcotti W, Erven A, Johnson SL, Steel KP, Kros CJ (2006) Tmc1 is necessary for normal functional maturation and survival of inner and outer hair cells in the mouse cochlea. J Physiol 574(Pt 3):677–698. doi:10.1113/jphysiol.2005.095661
Marcotti W, Corns LF, Desmonds T, Kirkwood NK, Richardson GP, Kros CJ (2014) Transduction without tip links in cochlear hair cells is mediated by ion channels with permeation properties distinct from those of the mechano-electrical transducer channel. J Neurosci 34(16):5505–5514. doi:10.1523/JNEUROSCI.4086-13.2014
Morton CC, Nance WE (2006) Newborn hearing screening—a silent revolution. N Engl J Med 354(20):2151–2164. doi:10.1056/NEJMra050700
Mutai H, Mann S, Heller S (2005) Identification of chicken transmembrane channel-like (TMC) genes: expression analysis in the cochlea. Neuroscience 132(4):1115–1122. doi:10.1016/j.neuroscience.2005.01.046
Noguchi Y, Kurima K, Makishima T, de Angelis MH, Fuchs H, Frolenkov G, Kitamura K, Griffith AJ (2006) Multiple quantitative trait loci modify cochlear hair cell degeneration in the Beethoven (Tmc1 Bth) mouse model of progressive hearing loss DFNA36. Genetics 173(4):2111–2119. doi:10.1534/genetics.106.057372
Pan B, Geleoc GS, Asai Y, Horwitz GC, Kurima K, Ishikawa K, Kawashima Y, Griffith AJ, Holt JR (2013) TMC1 and TMC2 are components of the mechanotransduction channel in hair cells of the mammalian inner ear. Neuron 79(3):504–515. doi:10.1016/j.neuron.2013.06.019
Steel KP, Bock GR (1980) The nature of inherited deafness in deafness mice. Nature 288(5787):159–161
Vreugde S, Erven A, Kros CJ, Marcotti W, Fuchs H, Kurima K, Wilcox ER, Friedman TB, Griffith AJ, Balling R, Hrabe De Angelis M, Avraham KB, Steel KP (2002) Beethoven, a mouse model for dominant, progressive hearing loss DFNA36. Nat Genet 30(3):257–258. doi:10.1038/ng848
Wang Q, Xue Y, Zhang Y, Long Q, Asan YF, Turner DJ, Fitzgerald T, Ng BL, Zhao Y, Chen Y, Liu Q, Yang W, Han D, Quail MA, Swerdlow H, Burton J, Fahey C, Ning Z, Hurles ME, Carter NP, Yang H, Tyler-Smith C (2013) Genetic basis of Y-linked hearing impairment. Am J Hum Genet 92(2):301–306. doi:10.1016/j.ajhg.2012.12.015
Yang T, Kahrizi K, Bazazzadeghan N, Meyer N, Najmabadi H, Smith RJ (2010) A novel mutation adjacent to the Bth mouse mutation in the TMC1 gene makes this mouse an excellent model of human deafness at the DFNA36 locus. Clin Genet 77(4):395–398. doi:10.1111/j.1399-0004.2009.01338.x
Zhao Y, Wang D, Zong L, Zhao F, Guan L, Zhang P, Shi W, Lan L, Wang H, Li Q, Han B, Yang L, Jin X, Wang J, Wang J, Wang Q (2014) A novel DFNA36 mutation in TMC1 orthologous to the Beethoven (Bth) mouse associated with autosomal dominant hearing loss in a Chinese family. PLoS ONE 9(5):e97064. doi:10.1371/journal.pone.0097064
Acknowledgments
Kiyoto Kurima and Andrew Griffith are supported by NIH intramural research fund Z01-DC000060-13. Bifeng Pan and Jeffrey R. Holt are supported by NIH/NIDCD grant no. RO1DC013521. Yoshiyuki Kawashima is supported by JSPS KAKENHI grant no. 26462551.
Conflict of interest
Kiyoto Kurima and Andrew J. Griffith hold the following U.S. patents: 7,166,433 (Transductin-2 and Applications to Hereditary Deafness), 7,192,705 (Transductin-1 and Applications to Hereditary Deafness), and 7,659,115 (Nucleic Acid Encoding Human Transductin-1 Polypeptide).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Kawashima, Y., Kurima, K., Pan, B. et al. Transmembrane channel-like (TMC) genes are required for auditory and vestibular mechanosensation. Pflugers Arch - Eur J Physiol 467, 85–94 (2015). https://doi.org/10.1007/s00424-014-1582-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-014-1582-3