Abstract
Purpose
We examined changes in plasma creatine kinase (CK) activity, hydroxyproline and cell-free DNA (cfDNA) concentrations in relation to changes in maximum voluntary isometric contraction (MVIC) torque and delayed-onset muscle soreness (DOMS) following a session of volume-matched higher- (HI) versus lower-intensity (LI) eccentric cycling exercise.
Methods
Healthy young men performed either 5 × 1-min HI at 20% of peak power output (n = 11) or 5 × 4-min LI eccentric cycling at 5% of peak power output (n = 9). Changes in knee extensor MVIC torque, DOMS, plasma CK activity, and hydroxyproline and cfDNA concentrations before, immediately after, and 24–72 h post-exercise were compared between groups.
Results
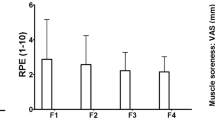
Plasma CK activity increased post-exercise (141 ± 73.5%) and MVIC torque decreased from immediately (13.3 ± 7.8%) to 48 h (6.7 ± 13.5%) post-exercise (P < 0.05), without significant differences between groups. DOMS was greater after HI (peak: 4.5 ± 3.0 on a 10-point scale) than LI (1.2 ± 1.0). Hydroxyproline concentration increased 40–53% at 24–72 h after both LI and HI (P < 0.05). cfDNA concentration increased immediately after HI only (2.3 ± 0.9-fold, P < 0.001), with a significant difference between groups (P = 0.002). Lack of detectable methylated HOXD4 indicated that the cfDNA was not derived from skeletal muscle. No significant correlations were evident between the magnitude of change in the measures, but the cfDNA increase immediately post-exercise was correlated with the maximal change in heart rate during exercise (r = 0.513, P = 0.025).
Conclusion
Changes in plasma hydroxyproline and cfDNA concentrations were not associated with muscle fiber damage, but the increased hydroxyproline in both groups suggests increased collagen turnover. cfDNA may be a useful metabolic-intensity exercise marker.





Similar content being viewed by others
Abbreviations
- cfDNA:
-
Cell-free DNA
- CK:
-
Creatine kinase
- ddPCR:
-
Droplet digital PCR
- DOMS:
-
Delayed-onset muscle soreness
- HI:
-
Higher intensity
- HR:
-
Heart rate
- Hyp:
-
Hydroxyproline
- LI:
-
Lower intensity
- MVIC:
-
Maximal voluntary isometric contraction
- PPO:
-
Peak power output
- RPE:
-
Rate of perceived effort
References
Andreatta MV, Curty VM, Coutinho JVS et al (2018) Cell-free DNA as an earlier predictor of exercise-induced performance decrement related to muscle damage. Int J Sports Physiol Perform 13:953–956. https://doi.org/10.1123/ijspp.2017-0421
Atamaniuk J, Vidotto C, Tschan H et al (2004) Increased concentrations of cell-free plasma DNA after exhaustive exercise. Clin Chem 50:1668–1670. https://doi.org/10.1373/clinchem.2004.034553
Atamaniuk J, Stuhlmeier KM, Vidotto C et al (2008) Effects of ultra-marathon on circulating DNA and mRNA expression of pro- and anti-apoptotic genes in mononuclear cells. Eur J Appl Physiol 104:711–717. https://doi.org/10.1007/s00421-008-0827-2
Atamaniuk J, Vidotto C, Kinzlbauer M et al (2010) Cell-free plasma DNA and purine nucleotide degradation markers following weightlifting exercise. Eur J Appl Physiol 110:695–701. https://doi.org/10.1007/s00421-010-1532-5
Beiter T, Fragasso A, Hudemann J et al (2011) Short-term treadmill running as a model for studying cell-free DNA kinetics in vivo. Clin Chem 57:633–636. https://doi.org/10.1373/clinchem.2010.158030
Beiter T, Fragasso A, Hudemann J et al (2014) Neutrophils release extracellular DNA traps in response to exercise. J Appl Physiol 117:325–333. https://doi.org/10.1152/japplphysiol.00173.2014
Beiter T, Fragasso A, Hartl D, Nieß AM (2015) Neutrophil extracellular traps: a walk on the wild side of exercise immunology. Sports Med 45:625–640. https://doi.org/10.1007/s40279-014-0296-1
Borg G (1998) Borg’s perceived exertion and pain scales. Human Kinetics, Champaign
Breitbach S, Tug S, Simon P (2012) Circulating cell-free DNA: an up-coming molecular marker in exercise physiology. Sports Med 42:565–586. https://doi.org/10.2165/11631380-000000000-00000
Breitbach S, Sterzing B, Magallanes C et al (2014) Direct measurement of cell-free DNA from serially collected capillary plasma during incremental exercise. J Appl Physiol 117:119–130. https://doi.org/10.1152/japplphysiol.00002.2014
Brown SJ, Child RB, Day SH, Donnelly AE (1997) Indices of skeletal muscle damage and connective tissue breakdown following eccentric muscle contractions. Eur J Appl Physiol Occup Physiol 75:369–374. https://doi.org/10.1007/s004210050174
Brown S, Day S, Donnelly A (1999) Indirect evidence of human skeletal muscle damage and collagen breakdown after eccentric muscle actions. J Sports Sci 17:397–402. https://doi.org/10.1080/026404199365911
Clarkson PM, Nosaka K, Braun B (1992) Muscle function after exercise-induced muscle damage and rapid adaptation. Med Sci Sports Exerc 24:512–520. https://doi.org/10.1249/00005768-199205000-00004
Crameri RM, Aagaard P, Qvortrup K et al (2007) Myofibre damage in human skeletal muscle: effects of electrical stimulation versus voluntary contraction. J Physiol 583:365–380. https://doi.org/10.1113/jphysiol.2007.128827
Ehrlich M, Lacey M (2013) DNA methylation and differentiation: silencing, upregulation and modulation of gene expression. Epigenomics 5:553–568. https://doi.org/10.2217/epi.13.43
Fatouros IG, Jamurtas AZ, Nikolaidis MG et al (2010) Time of sampling is crucial for measurement of cell-free plasma DNA following acute aseptic inflammation induced by exercise. Clin Biochem 43:1368–1370. https://doi.org/10.1016/j.clinbiochem.2010.08.020
Ferrandi PJ, Fico BG, Whitehurst M et al (2018) Acute high-intensity interval exercise induces comparable levels of circulating cell-free DNA and Interleukin-6 in obese and normal-weight individuals. Life Sci 202:161–166. https://doi.org/10.1016/j.lfs.2018.04.007
Frühbeis C, Helmig S, Tug S et al (2015) Physical exercise induces rapid release of small extracellular vesicles into the circulation. J Extracell Vesicles 4:28239. https://doi.org/10.3402/jev.v4.28239
Grounds MD, Sorokin L, White J (2005) Strength at the extracellular matrix-muscle interface. Scand J Med Sci Sports 15:381–391. https://doi.org/10.1111/j.1600-0838.2005.00467.x
Haller N, Tug S, Breitbach S et al (2017) Increases in circulating cell-free DNA during aerobic running depend on intensity and duration. Int J Sports Physiol Perform 12:455–462. https://doi.org/10.1123/ijspp.2015-0540
Haller N, Helmig S, Taenny P et al (2018) Circulating, cell-free DNA as a marker for exercise load in intermittent sports. PLoS ONE 13:e0191915. https://doi.org/10.1371/journal.pone.0191915
Helmig S, Frühbeis C, Krämer-Albers E-M et al (2015) Release of bulk cell free DNA during physical exercise occurs independent of extracellular vesicles. Eur J Appl Physiol 115:2271–2280. https://doi.org/10.1007/s00421-015-3207-8
Julian V, Thivel D, Costes F et al (2018) Eccentric training improves body composition by inducing mechanical and metabolic adaptations: a promising approach for overweight and obese individuals. Front Physiol. https://doi.org/10.3389/fphys.2018.01013
Kish K, Mezil Y, Ward WE et al (2015) Effects of plyometric exercise session on markers of bone turnover in boys and young men. Eur J Appl Physiol 115:2115–2124. https://doi.org/10.1007/s00421-015-3191-z
Koskinen SOA, Ahtikoski AM, Komulainen J et al (2002) Short-term effects of forced eccentric contractions on collagen synthesis and degradation in rat skeletal muscle. Pflügers Arch 444:59–72. https://doi.org/10.1007/s00424-002-0792-2
Kotch FW, Guzei IA, Raines RT (2008) Stabilization of the collagen triple helix by o-methylation of hydroxyproline residues. J Am Chem Soc 130:2952–2953. https://doi.org/10.1021/ja800225k
LaStayo PC, Pierotti DJ, Pifer J et al (2000) Eccentric ergometry: increases in locomotor muscle size and strength at low training intensities. Am J Physiol Regul Integr Comp Physiol 278:R1282-1288. https://doi.org/10.1152/ajpregu.2000.278.5.R1282
Maïmoun L, Manetta J, Couret I et al (2006) The intensity level of physical exercise and the bone metabolism response. Int J Sports Med 27:105–111. https://doi.org/10.1055/s-2005-837621
Mavropalias G, Koeda T, Barley OR et al (2020) Comparison between high- and low-intensity eccentric cycling of equal mechanical work for muscle damage and the repeated bout effect. Eur J Appl Physiol 120:1015–1025. https://doi.org/10.1007/s00421-020-04341-5
Murguia MJ, Vailas A, Mandelbaum B et al (1988) Elevated plasma hydroxyproline. A possible risk factor associated with connective tissue injuries during overuse. Am J Sports Med 16:660–664. https://doi.org/10.1177/036354658801600619
Paschalis V, Koutedakis Y, Jamurtas AZ et al (2005) Equal volumes of high and low intensity of eccentric exercise in relation to muscle damage and performance. J Strength Cond Res 19:184–188. https://doi.org/10.1519/R-14763.1
Paulsen G, Crameri R, Benestad HB et al (2010) Time course of leukocyte accumulation in human muscle after eccentric exercise. Med Sci Sports Exerc 42:75–85. https://doi.org/10.1249/MSS.0b013e3181ac7adb
Peñailillo L, Blazevich A, Numazawa H, Nosaka K (2013) Metabolic and muscle damage profiles of concentric versus repeated eccentric cycling. Med Sci Sports Exerc 45:1773–1781. https://doi.org/10.1249/MSS.0b013e31828f8a73
Peñailillo L, Mackay K, Abbiss CR (2018) Rating of perceived exertion during concentric and eccentric cycling: are we measuring effort or exertion? Int J Sports Physiol Perform 13:517–523. https://doi.org/10.1123/ijspp.2017-0171
Stawski R, Walczak K, Kosielski P et al (2017) Repeated bouts of exhaustive exercise increase circulating cell free nuclear and mitochondrial DNA without development of tolerance in healthy men. PLoS ONE 12:e0178216. https://doi.org/10.1371/journal.pone.0178216
Tofas T, Jamurtas AZ, Fatouros I et al (2008) Plyometric exercise increases serum indices of muscle damage and collagen breakdown. J Strength Cond Res 22:490–496. https://doi.org/10.1519/JSC.0b013e31816605a0
Tug S, Helmig S, Deichmann ER et al (2015) Exercise-induced increases in cell free DNA in human plasma originate predominantly from cells of the haematopoietic lineage. Exerc Immunol Rev 21:164–173
Tug S, Mehdorn M, Helmig S et al (2017a) Exploring the potential of cell-free-DNA measurements after an exhaustive cycle-ergometer test as a marker for performance-related parameters. Int J Sports Physiol Perform 12:597–604. https://doi.org/10.1123/ijspp.2016-0157
Tug S, Tross A-K, Hegen P et al (2017b) Acute effects of strength exercises and effects of regular strength training on cell free DNA concentrations in blood plasma. PLoS ONE 12:e0184668. https://doi.org/10.1371/journal.pone.0184668
Velders M, Treff G, Machus K et al (2014) Exercise is a potent stimulus for enhancing circulating DNase activity. Clin Biochem 47:471–474. https://doi.org/10.1016/j.clinbiochem.2013.12.017
Virtanen P, Viitasalo JT, Vuori J et al (1993) Effect of concentric exercise on serum muscle and collagen markers. J Appl Physiol 75:1272–1277. https://doi.org/10.1152/jappl.1993.75.3.1272
Wobbrock JO, Findlater L, Gergle D, Higgins JJ (2011) The aligned rank transform for nonparametric factorial analyses using only ANOVA procedures. In: CHI 2011—29th Annual CHI Conference on Human Factors in Computing Systems, Conference Proceedings and Extended Abstracts. Vancouver, BC, Canada, pp 143–146
Woodward MI, Cunningham JL (1993) Skeletal accelerations measured during different exercises. Proc Inst Mech Eng H 207:79–85. https://doi.org/10.1243/PIME_PROC_1993_207_274_02
Acknowledgements
The authors thank all the study participants for their assistance with the study. We are thankful to Dr Gina Ravenscroft and Dr Hayley Goullee for providing the primary human myoblast line used as positive control.
Funding
This study was funded by an Intra-School Collaboration Seed Fund from the School of Medical and Health Sciences at Edith Cowan University. GM has received financial support through a scholarship from the Australian Government Research Training Program. EG is supported by fellowships from Cancer Council Western Australia (CCWA).
Author information
Authors and Affiliations
Contributions
GM, AB and KN conceived and designed the study, and GM and LC conducted the experiments. GM collected, and GM, OB, WP, LC, MM, TK, and EG analyzed the data. GM drafted the manuscript with AB and KN. All authors read, edited and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare there are no competing interests.
Additional information
Communicated by Michalis G Nikolaidis.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mavropalias, G., Calapre, L., Morici, M. et al. Changes in plasma hydroxyproline and plasma cell-free DNA concentrations after higher- versus lower-intensity eccentric cycling. Eur J Appl Physiol 121, 1087–1097 (2021). https://doi.org/10.1007/s00421-020-04593-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-020-04593-1