Abstract
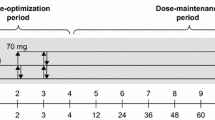
Multiple sclerosis (MS) causes cognitive impairment including slowed processing speed and problems with learning and memory. Stimulants are attractive candidates for improving mental speed but carry risk of addiction and other adverse behavioral effects. Lisdexamfetamine dimesylate (LDX) is a d-amphetamine prodrug currently approved for attention deficit (hyperactivity) disorder with the potential to be better tolerated due to its prolonged clinical effect. This phase II placebo-controlled, double-blind study aimed to assess the safety and efficacy of LDX in cognitively impaired MS patients. Subjects were patients with clinically definite MS, aged 18–56 years, and impaired on either of two primary outcomes: the Symbol Digit Modalities Test (SDMT) or the Paced Auditory Serial Addition Test (PASAT). Both SDMT and PASAT are measures of cognitive processing speed. Of 174 MS patients screened, 63 were randomized to 30 mg of LDX or placebo in a 2:1 fashion; the dose was increased as tolerated to 70 mg over 4 weeks and then maintained for another 4 weeks. Secondary outcomes were the Brief Visuospatial Memory Test Revised (BVMTR), the California Verbal Learning Test 2nd edition (CVLT2), both measures of episodic memory, and the Behavioral Rating Inventory of Executive Function for adults (BRIEF-A), a self-report measure of executive function. Fatigue and depression were also evaluated. There was significant improvement in the SDMT score (+4.6 vs. +1.3) and CVLT2 score (+4.7 vs. −0.9) in the LDX group compared with the placebo group among the 49 completers. There was no change on the other outcomes. A high proportion of both LDX-treated and placebo-treated subjects reported adverse events (73.5 % vs. 68.4 %). However, there were no serious adverse events noted in the study. These preliminary data indicate that LDX has the potential to be an efficacious treatment for MS patients with cognitive impairment.



Similar content being viewed by others
References
Hauser SL, Oksenberg JR, Hauser SL, Oksenberg JR (2006) The neurobiology of multiple sclerosis: genes, inflammation, and neurodegeneration. Neuron 52:61–76
Benedict RH, Cookfair D, Gavett R, Gunther M, Munschauer F, Garg N, Weinstock-Guttman B (2006) Validity of the minimal assessment of cognitive function in multiple sclerosis (MACFIMS). J Int Neuropsychol Soc 12:549–558
Rao SM, Leo GJ, Bernardin L, Unverzagt F (1991) Cognitive dysfunction in multiple sclerosis. I. Frequency, patterns, and prediction. Neurology 41:685–691
Bobholz JA, Rao SM (2003) Cognitive dysfunction in multiple sclerosis: a review of recent developments. Curr Opin Neurol 16:283–288
Beatty WW, Blanco CR, Wilbanks SL, Paul RH (1995) Demographic, clinical, and cognitive characteristics of multiple sclerosis patients who continue to work. J Neurol Rehabil 9:167–173
Parmenter BA, Zivadinov R, Kerenyi L, Gavett R, Weinstock-Guttman B, Dwyer M, Garg N, Munschauer F, Benedict RH (2007) Validity of the Wisconsin Card Sorting and Delis-Kaplan Executive Function System (DKEFS) Sorting Tests in multiple sclerosis. J Clin Exp Neuropsychol 29:215–223
Benedict RH, Wahlig E, Bakshi R, Fishman I, Munschauer F, Zivadinov R, Weinstock-Guttman B (2005) Predicting quality of life in multiple sclerosis: accounting for physical disability, fatigue, cognition, mood disorder, personality, and behavior change. J Neurol Sci 231(1–2):29–34
Carone D, Benedict RH, Munschauer FE III, Fishman I, Weinstock-Guttman B (2005) Interpreting patient/informant discrepancies of reported cognitive symptoms in MS. J Int Neuropsychol Soc 11:574–583
Benedict RH, Priore RL, Miller C, Munschauer F, Jacobs L (2001) Personality disorder in multiple sclerosis correlates with cognitive impairment. J Neuropsychiatry Clin Neurosci 13:70–76
Amato MP, Ponziani G, Siracusa G, Sorbi S (2001) Cognitive dysfunction in early-onset multiple sclerosis: a reappraisal after 10 years. Arch Neurol 58:1602–1606
Benedict RH, Zivadinov R (2006) Predicting neuropsychological abnormalities in multiple sclerosis. J Neurol Sci 245:67–72
Jacobs LD, Cookfair DL, Rudick RA, Herndon RM, Richert JR, Salazar AM, Fischer JS, Goodkin DE, Granger CV, Simon JH et al (1995) A phase III trial of intramuscular recombinant interferon beta as treatment for exacerbating-remitting multiple sclerosis: design and conduct of study and baseline characteristics of patients. Multiple Sclerosis Collaborative Research Group (MSCRG). Mult Scler 1:118–135
The IFNB Multiple Sclerosis Study Group; The University of British Columbia MS/MRI Analysis Group (1995) Interferon beta-1b in the treatment of multiple sclerosis: final outcome of the randomized controlled trial. Neurology 45:1277–1285
Lee EH, Ma YL (1995) Amphetamine enhances memory retention and facilitates norepinephrine release from the hippocampus in rats. Brain Res Bull 37:411–416
Wright FK, White KG (2003) Effects of methylphenidate on working memory in pigeons. Cogn Affect Behav Neurosci 3:300–308
Mattay VS, Callicott JH, Bertolino A, Heaton I, Frank JA, Coppola R, Berman KF, Goldberg TE, Weinberger DR (2000) Effects of dextroamphetamine on cognitive performance and cortical activation. Neuroimage 12:268–275
Makris AP, Rush CR, Frederich RC, Taylor AC, Kelly TH (2007) Behavioral and subjective effects of d-amphetamine and modafinil in healthy adults. Exp Clin Psychopharmacol 15:123–133
Barch DM, Carter CS (2005) Amphetamine improves cognitive function in medicated individuals with schizophrenia and in healthy volunteers. Schizophr Res 77:43–58
Pietrzak RH, Snyder PJ, Maruff P (2010) Use of an acute challenge with d-amphetamine to model cognitive improvement in chronic schizophrenia. Hum Psychopharmacol 25:353–358
Magill RA, Waters WF, Bray GA, Volaufova J, Smith SR, Lieberman HR, McNevin N, Ryan DH (2003) Effects of tyrosine, phentermine, caffeine D-amphetamine, and placebo on cognitive and motor performance deficits during sleep deprivation. Nutr Neurosci 6:237–246
Newhouse PA, Belenky G, Thomas M, Thorne D, Sing HC, Fertig J (1989) The effects of d-amphetamine on arousal, cognition, and mood after prolonged total sleep deprivation. Neuropsychopharmacology 2:153–164
Mason ST (1983) The neurochemistry and pharmacology of extinction behavior. Neurosci Biobehav Rev 7:325–347
Wise RA, Rompre PP (1989) Brain dopamine and reward. Annu Rev Psychol 40:191–225
Polman CH, Reingold SC, Edan G, Filippi M, Hartung HP, Kappos L, Lublin FD, Metz LM, McFarland HF, O’Connor PW, Sandberg-Wollheim M, Thompson AJ, Weinshenker BG, Wolinsky JS (2005) Diagnostic criteria for multiple sclerosis: 2005 revisions to the “McDonald Criteria”. Ann Neurol 58:840–846
Benedict RH, Zivadinov R (2007) Reliability and validity of neuropsychological screening and assessment strategies in MS. J Neurol 254(Suppl 2):II22–II25
Rao SM (1991) A manual for the brief repeatable battery of neuropsychological tests in multiple sclerosis. National Multiple Sclerosis Society, New York
Gronwall DMA (1977) Paced auditory serial addition task: a measure of recovery from concussion. Percept Mot Skills 44:367–373
Smith A (1982) Symbol digit modalities test: Manual. Western Psychological Services, Los Angeles
Benedict RH (1997) Brief Visuospatial Memory Test – Revised: Professional Manual. Psychological Assessment Resources, Odessa, Florida
Delis DC, Kramer JH, Kaplan E, Ober BA (2000) California Verbal Learning Test: Adult version. Psychological Corporation, San Antonio, TX
Langdon D, Amato M, Boringa J, Brochet B, Foley F, Fredrikson S, Hamalainen P, Hartung HP, Krupp L, Penner I, Reder A, Benedict R (2012) Recommendations for a Brief International Cognitive Assessment for Multiple Sclerosis (BICAMS). Mult Scler 18:891–898
Roth RM, Isquith PK, Gioia GA (2005) Behavioral Rating Inventory of Executive Function – Adult version. Psychological Assessment Resources, Lutz
Krupp LB, LaRocca NG, Muir-Nash J, Steinberg AD (1989) The fatigue severity scale. application to patients with multiple sclerosis and systemic lupus erythematosus. Arch Neurol 46(10):1121–1123
Benedict RH, Fishman I, McClellan MM, Bakshi R, Weinstock-Guttman B (2003) Validity of the Beck Depression Inventory-Fast Screen in multiple sclerosis. Mult Scler 9:393–396
Beck AT, Steer RA, Brown GK (2000) BDI-Fast Screen for Medical Patients: Manual. Psychological Corporation, San Antonio, TX
Heal DJ, Smith SL, Kulkarni RS, Rowley HL (2008) New perspectives from microdialysis studies in freely-moving, spontaneously hypertensive rats on the pharmacology of drugs for the treatment of ADHD. Pharmacol Biochem Behav 90:184–197
Sagvolden T, Xu T (2008) l-Amphetamine improves poor sustained attention while d-amphetamine reduces overactivity and impulsiveness as well as improves sustained attention in an animal model of Attention-Deficit/Hyperactivity Disorder (ADHD). Behav Brain Funct 4:3
Izquierdo I, Medina JH (1997) Memory formation: the sequence of biochemical events in the hippocampus and its connection to activity in other brain structures. Neurobiol Learn Mem 68:285–316
Drake AS, Weinstock-Guttman B, Morrow SA, Hojnacki D, Munschauer FE, Benedict RH (2010) Psychometrics and normative data for the Multiple Sclerosis Functional Composite: replacing the PASAT with the Symbol Digit Modalities Test. Mult Scler 16:228–237
Brochet B, Deloire MS, Bonnet M, Salort-Campana E, Ouallet JC, Petry KG, Dousset V (2008) Should SDMT substitute for PASAT in MSFC? A 5-year longitudinal study. Mult Scler 14:1242–1249
Morrow S, O’Connor P, Polman C, Goodman A, Kappos L, Lublin F, Rudick R, Jurgensen S, Paes D, Forrestal F, Benedict R (2010) Evaluation of the Symbol Digit Modalities Test (SDMT) and MS Neuropsychological Screening Questionnaire (MSNQ) in natalizumab-treated MS patients over 48 weeks. Mult Scler 16:1385–1392
Benedict RH, Duquin JA, Jurgensen S, Rudick RA, Feitcher J, Munschauer FE, Panzara MA, Weinstock-Guttman B (2008) Repeated assessment of neuropsychological deficits in multiple sclerosis using the Symbol Digit Modalities Test and the MS Neuropsychological Screening Questionnaire. Mult Scler 14:940–946
DeLuca J, Gaudino EA, Diamond BJ, Christodoulou C, Engel RA (1998) Acquisition and storage deficits in multiple sclerosis. J Clin Exp Neuropsychol 20:376–390
Gioia G, Isquith P, Guy S, Kenworthy L (2000) The Behavior Rating Inventory of Executive Function. Psychological Assessment Resources, Lutz, FL
Benedict RH, Munschauer F, Linn R, Miller C, Murphy E, Foley F, Jacobs L (2003) Screening for multiple sclerosis cognitive impairment using a self-administered 15-item questionnaire. Mult Scler 9:95–101
Deloire MS, Bonnet MC, Salort E, Arimone Y, Boudineau M, Petry KG, Brochet B (2006) How to detect cognitive dysfunction at early stages of multiple sclerosis? Mult Scler 12:445–452
Benedict RH, Walton MK (2012) Evaluating cognitive outcome measures for MS clinical trials: what is a clinically meaningful change? Mult Scler. doi:10.1177/1352458512454774
FDA (2010) Vyvanse (lisdexamfetamine dimesylate). Prescribing information. US Department of Health and Human Services
Lower EE, Harman S, Baughman RP (2008) Double-blind, randomized trial of dexmethylphenidate hydrochloride for the treatment of sarcoidosis-associated fatigue. Chest 133(5):1189–1195
Roth AJ, Nelson C, Rosenfeld B, Scher H, Slovin S, Morris M, O’Shea N, Arauz G, Breitbart W (2010) Methylphenidate for fatigue in ambulatory men with prostate cancer. Cancer 116:5102–5110
Gore RK, Webb TS, Hermes ED (2010) Fatigue and stimulant use in military fighter aircrew during combat operations. Aviat Space Environ Med 81:719–727
Kirkpatrick MG, Haney M, Vosburg SK, Comer SD, Foltin RW, Hart CL (2009) Methamphetamine self-administration by humans subjected to abrupt shift and sleep schedule changes. Psychopharmacology 203:771–780
Peuckmann-Post V, Elsner F, Krumm N, Trottenberg P, Radbruch L (2010) Pharmacological treatments for fatigue associated with palliative care. Cochrane Database of Systematic Reviews, Issue 11. Art. No.: CD006788. doi:10.1002/14651858.CD006788.pub2
Morrow SA, Weinstock-Guttman B, Munschauer FE, Hojnacki D, Benedict RH (2009) Subjective fatigue is not associated with cognitive impairment in multiple sclerosis: cross-sectional and longitudinal analysis. Mult Scler 15:998–1005
Acknowledgments
This study was funded by Shire Development LLC as an investigator-initiated trial.
Conflicts of interest
Dr. Morrow has participated in speaker’s bureaus and/or served as a consultant for Bayer, Biogen Idec, EMD Serono, Teva Neurosciences and Novartis. She has also received grant/research support from Biogen Idec and Novartis. She has no other industry financial relationships. Dr. Smerbeck, Dr. Cookfair and Ms. Patrick have nothing to disclose. Dr. Weinstock-Guttman has participated in speaker’s bureaus and served as a consultant for Biogen Idec, Teva Neurosciences, EMD Serono, Pfizer, Novartis, Genzyme and Accorda. Excluding Genzyme, she has also received grant/research support from the agencies listed above as well as ITN, Questcor and Shire. She has no other industry financial relationships. Dr. Ralph Benedict has served as a consultant for Actelion, Biogen Idec, Bayer and Novartis. He has also received grant/research support from Accorda, Biogen Idec and Shire. He also receives royalties from Psychological Assessment Resources. He has no other industry financial relationships.
Ethical standard
This study has been approved by the appropriate ethics committee and has therefore been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Morrow, S.A., Smerbeck, A., Patrick, K. et al. Lisdexamfetamine dimesylate improves processing speed and memory in cognitively impaired MS patients: a phase II study. J Neurol 260, 489–497 (2013). https://doi.org/10.1007/s00415-012-6663-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-012-6663-7