Abstract
Introduction
Donor PaO2 levels are used for assessing organs for lung transplantation (LTx), but survival implications of PaO2 levels in adult cystic fibrosis (CF) patients receiving LTx are unclear.
Methods
UNOS registry data spanning 2005–2013 were used to test for associations of donor PaO2 with patient survival and bronchiolitis obliterans syndrome (BOS) in adult (age ≥ 18 years) first-time LTx recipients diagnosed with CF.
Results
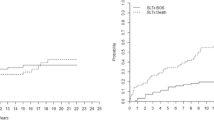
The analysis included 1587 patients, of whom 1420 had complete data for multivariable Cox models. No statistically significant differences among donor PaO2 categories of ≤200, 201–300, 301–400, or >400 mmHg were found in univariate survival analysis (log-rank test p = 0.290). BOS onset did not significantly differ across donor PaO2 categories (Chi-square p = 0.480). Multivariable Cox models of patient survival supported the lack of difference across donor PaO2 categories. Interaction analysis found a modest difference in survival between the two top categories of donor PaO2 when examining patients with body mass index (BMI) in the lowest decile (≤16.5 kg/m2).
Conclusions
Donor PaO2 was not associated with survival or BOS onset in adult CF patients undergoing LTx. Notwithstanding statistically significant interactions between donor PaO2 and BMI, there was no evidence of post-LTx survival risk associated with donor PaO2 below conventional thresholds in any subgroup of adults with CF.

Similar content being viewed by others
References
Yusen RD, Edwards LB, Kucheryavaya AY et al (2014) The registry of the International Society for Heart and Lung Transplantation: thirty-first adult lung and heart-lung transplant report–2014; focus theme: retransplantation. J Heart Lung Transpl 33(10):1009–1024
Benden C, Goldfarb SB, Edwards LB et al (2014) The registry of the International Society for Heart and Lung Transplantation: seventeenth official pediatric lung and heart-lung transplantation report–2014; focus theme: retransplantation. J Heart Lung Transpl 33(10):1025–1033
Thabut G, Christie JD, Mal H et al (2013) Survival benefit of lung transplant for cystic fibrosis since lung allocation score implementation. Am J Respir Crit Care Med 187(12):1335–1340
Kawut SM, Reyentovich A, Wilt JS et al (2005) Outcomes of extended donor lung recipients after lung transplantation. Transplantation 79(3):310–316
Yeung JC, Cypel M, Waddell TK et al (2009) Update on donor assessment, resuscitation, and acceptance criteria, including novel techniques–non-heart-beating donor lung retrieval and ex vivo donor lung perfusion. Thorac Surg Clin 19(2):261–274
Ingemansson R, Eyjolfsson A, Mared L et al (2009) Clinical transplantation of initially rejected donor lungs after reconditioning ex vivo. Ann Thorac Surg 87(1):255–260
Klein AS, Messersmith EE, Ratner LE et al (2010) Organ donation and utilization in the United States, 1999–2008. Am J Transpl 10(4 Pt 2):973–986
Mason DP, Thuita L, Nowicki ER et al (2010) Should lung transplantation be performed for patients on mechanical respiratory support? The US experience. J Thorac Cardiovasc Surg 139(3):765.el–773.el
Reyes KG, Mason DP, Thuita L et al. (2010) Guidelines for donor lung selection: time for revision? Ann Thorac Surg 89(6):1756–1764; discussion 1764-5
Cypel M, Yeung JC, Liu M et al (2011) Normothermic ex vivo lung perfusion in clinical lung transplantation. N Engl J Med 364(15):1431–1440
Levvey BJ, Harkess M, Hopkins P et al (2012) Excellent clinical outcomes from a national donation-after-determination-of-cardiac-death lung transplant collaborative. Am J Transpl 12(9):2406–2413
Bernhardt AM, Rahmel A, Reichenspurner H (2013) The unsolved problem of organ allocation in times of organ shortage: the German solution? J Heart Lung Transpl 32(11):1049–1051
Morrissey PE, Monaco AP (2014) Donation after circulatory death: current practices, ongoing challenges, and potential improvements. Transplantation 97(3):258–264
Thabut G, Mal H, Cerrina J et al (2005) Influence of donor characteristics on outcome after lung transplantation: a multicenter study. J Heart Lung Transpl 24(9):1347–1353
Frost AE (1997) Donor criteria and evaluation. Clin Chest Med 18(2):231–237
Orens JB, Boehler A, de Perrot M et al (2003) A review of lung transplant donor acceptability criteria. J Heart Lung Transpl 22(11):1183–1200
Zafar F, Khan MS, Heinle JS et al (2012) Does donor arterial partial pressure of oxygen affect outcomes after lung transplantation? A review of more than 12,000 lung transplants. J Thorac Cardiovasc Surg 143(4):919–925
Harjula A, Baldwin JC, Starnes VA et al (1987) Proper donor selection for heart-lung transplantation. The Stanford experience. J Thorac Cardiovasc Surg 94(6):874–880
Hennessy SA, Hranjec T, Swenson BR et al (2010) Donor factors are associated with bronchiolitis obliterans syndrome after lung transplantation. Ann Thorac Surg 89(5):1555–1562
United Network for Organ Sharing/Organ Procurement and Transplantation Network Standard Transplant Analysis and Research Database. http://optn.transplant.hrsa.gov/data/about/OPTNDatabase.asp. Accessed 6 Sept 2013
Iribarne A, Russo MJ, Davies RR et al (2009) Despite decreased wait-list times for lung transplantation, lung allocation scores continue to increase. Chest 135(4):923–928
Valapour M, Skeans MA, Heubner BM et al (2014) OPTN/SRTR 2012 Annual Data Report: lung. Am J Transpl 14(Suppl 1):139–165
McShane PJ, Garrity ER Jr (2013) Impact of the lung allocation score. Semin Respir Crit Care Med 34(3):275–280
Hayes D Jr, Kirkby S, Whitson BA et al (2015) Mortality risk and pulmonary function in adults with cystic fibrosis at time of wait listing for lung transplantation. Ann Thorac Surg 100(2):474–479
Hayes D Jr, Daniels CJ, McCoy KS et al (2016) Pulmonary artery pressure and benefit of lung transplantation in adult cystic fibrosis patients. Ann Thorac Surg 101(3):1104–1109
Gabbay E, Williams TJ, Griffiths AP et al (1999) Maximizing the utilization of donor organs offered for lung transplantation. Am J Respir Crit Care Med 160(1):265–271
Lardinois D, Banysch M, Korom S et al (2005) Extended donor lungs: eleven years experience in a consecutive series. Eur J Cardiothorac Surg 27(5):762–767
Du ZY, Hicks M, Winlaw D, Spratt P, Macdonald P (1996) Ischemic preconditioning enhances donor lung preservation in the rat. J Heart Lung Transpl 15(12):1258–1267
Friedrich I, Spillner J, Lu EX et al (2001) Ischemic pre-conditioning of 5 minutes but not of 10 minutes improves lung function after warm ischemia in a canine model. J Heart Lung Transpl 20(9):985–995
Markart P, Schmidt R, Ruppert C et al (2005) Ischemic and endotoxin pre-conditioning reduce lung reperfusion injury-induced surfactant alterations. J Heart Lung Transpl 24(10):1680–1689
Merry HE, Wolf PS, Fitzsullivan E, Keech JC, Mulligan MS (2010) Lipopolysaccharide pre-conditioning is protective in lung ischemia-reperfusion injury. J Heart Lung Transpl 29(4):471–478
Tierney DF (1974) Lung metabolism and biochemistry. Annu Rev Physiol 36:209–231
Touya JJ, Rahimian J, Corbus HF et al (1986) The lung as a metabolic organ. Semin Nucl Med 16(4):296–305
Author Contributions
Don Hayes, Jr.: Conception and design, acquisition of data, interpretation of data, drafted the manuscript; Benjamin T. Kopp, Stephen E. Kirkby, Susan D. Reynolds, Heidi M. Mansour, Joseph D. Tobias: Interpretation of data, revision of the manuscript; Dmitry Tumin: Conception and design, interpretation of data, statistical analysis, revision of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests
The authors have no relevant disclosures or conflicts of interest to report.
Rights and permissions
About this article
Cite this article
Hayes, D., Kopp, B.T., Kirkby, S.E. et al. Impact of Donor Arterial Partial Pressure of Oxygen on Outcomes After Lung Transplantation in Adult Cystic Fibrosis Recipients. Lung 194, 547–553 (2016). https://doi.org/10.1007/s00408-016-9902-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00408-016-9902-3