Abstract
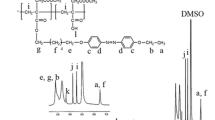
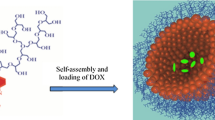
Amphiphilic random copolymer micelle of P(NVP-co-NHPM) that responds to both UV and NIR light was designed and investigated. N-hydroxyphthalimide methacrylate—bearing a photoacid generator functional group was synthesized and used to construct the hydrophobic segment of P(NVP-co-NHPM); the N–O bond can be photocleaved when exposing the polymer aqueous solution to 310 nm UV light irradiation, inducing the micellar dissociation and delivery of loaded substances. Dynamic light scattering (DLS), atomic force microscope (AFM), and transmission electron microscopy (TEM) were used to characterize the micellar dissociation and controlled release processes. Moreover, the upconverting nanoparticles (NaLuF4:Gd/Yb/Tm) that can effectively absorb NIR light and convert it into visible and UV light were coloaded into polymer micelles to activate the micellar dissociation and the delivery of loaded cargoes via 980 nm NIR light.







Similar content being viewed by others
References
Zhou YF, Huang W, Liu JY, Zhu XY, Yan DY (2010) Self-assembly of hyperbranched polymers and its biomedical applications. Adv Mater 22:4567–4590
Li MH, Patrick KP (2009) Stimuli-responsive polymer vesicles. Soft Matter 5:927–937
Zhang Q, Ko NR, Oh JK (2012) Recent advances in stimuli responsive degradable block copolymer micelles: synthesis and controlled drug delivery applications. Chem Commun 48:7542–7552
Jin Q, Cai TJ, Han HJ, Wang HB, Wang Y, Ji J (2014) Light and pH dual-degradable triblock copolymer micelles for controlled intracellular drug release. Macromol Rapid Commun 35:1372–1378
Palao-Suay R, Gómez-Mascaraque LG, Aguilar MR, Vázquez-Lasa B, San Román J (2016) Self-assembling polymer systems for advanced treatment of cancer and inflammation. Prog Polym Sci 53:207–248
Roy D, Cambre JN, Sumerlin BS (2010) Future perspectives and recent advances in stimuli-responsive materials. Prog Polym Sci 35:278–301
Heinrich M, Kulozik U (2011) Study of chymosin hydrolysis of casein micelles under ultrahigh pressure: effect on re-association upon pressure release. Int Dairy J 21:664–669
Guerry A, Cottaz S, Fleury E, Bernard J, Halila S (2014) Redox-stimuli responsive micelles from DOX-encapsulating polycaprolactone-g-chitosan oligosaccharide. Carbohydr Polym 112:746–752
Liu GQ, Li XL, Xiong SD, Li L, Chu PK, Yeung KWK, Wu SL, Xu ZS (2012) Fluorine-containing pH-responsive core/shell microgel particles: preparation, characterization, and their applications in controlled drug release. Colloid Polym Sci 290:349–357
Wang Y, Li QY, Liu XB, Zhang CY, Wu ZM, Guo XD (2015) Mesoscale simulations and experimental studies of pH-sensitive micelles for controlled drug delivery. ACS Appl Mater Interfaces 7:25592–25600
Salentinig S, Phan S, Darwish TA, Kirby N, Boyd BJ, Gilbert EP (2014) PH-responsive micelles based on caprylic acid. Langmuir 30:7296–7303
Laaser JE, Jiang YM, Sprouse D, Reineke TM, Lodge TP (2015) PH- and ionic-strength-induced contraction of polybasic micelles in buffered aqueous solutions. Macromolecules 48:2677–2685
Ma XP, Wang YG, Zhao T, Li Y, Su LC, Wang ZH, Huang G, Sumer BD, Gao JM (2014) Ultra-pH-sensitive nanoprobe library with broad pH tunability and fluorescence emissions. J Am Chem Soc 136:11085–11092
Liu L, Du PC, Zhao XB, Zeng J, Liu P (2015) Independent temperature and pH dual-stimuli responsive yolk/shell polymer microspheres for controlled release: structural effect. Eur Polym J 69:540–551
Jiang FJ, Chen S, Cao ZQ, Wang GJ (2016) A photo, temperature, and pH responsive spiropyran-functionalized polymer: synthesis, self-assembly and controlled release. Polymer 83:85–91
Zhao Y (2012) Light-responsive block copolymer micelles. Macromolecules 45:3647–3657
Jiang JQ, Tong X, Morris D, Zhao Y (2006) Toward photocontrolled release using light-dissociable block copolymer micelles. Macromolecules 39:4633–4640
Chang H, Liu Y, Shi M, Liu ZT, Liu ZW, Jiang JQ (2015) Photo-induced dynamic association of coumarin pendants within amphiphilic random copolymer micelles. Colloid Polym Sci 293:823–831
Wu YH, Hu HM, Hu JM, Liu T, Zhang GY, Liu SY (2013) Thermo- and light-regulated formation and disintegration of double hydrophilic block copolymer assemblies with tunable fluorescence emissions. Langmuir 29:3711–3720
Gohy JF, Zhao Y (2013) Photo-responsive block copolymer micelles: design and behavior. Chem Soc Rev 42:7117–7129
Jiang JQ, Shu QZ, Chen X, Yang YQ, Yi CL, Song XQ, Liu XY, Chen MQ (2010) Photoinduced morphology switching of polymer nanoaggregates in aqueous solution. Langmuir 26:14247–14254
Biswas S, Kumari P, Lakhani PM, Ghosh B (2016) Recent advances in polymeric micelles for anti-cancer drug delivery. Eur J Pharm Sci 83:184–202
Zhao Y (2009) Photocontrollable block copolymer micelles: what can we control? J Mater Chem 19:4887–4895
Pearson S, Vitucci D, Khine YY, Dag A, Lu HX, Save M, Billon L, Stenzel MH (2015) Light-responsive azobenzene-based glycopolymer micelles for targeted drug delivery to melanoma cells. Eur Polym J 69:616–627
Jiang JQ, Tong X, Zhao Y (2005) A new design for light-breakable polymer micelles. J Am Chem Soc 127:8290–8829
Ye Z, Guo JJ, Wu DW, Tan MY, Xiong X, Yin YH, He GH (2015) Photo-responsive shell cross-linked micelles based on carboxymethyl chitosan and their application in controlled release of pesticide. Carbohydr Polym 132:520–528
Chen CJ, Jin Q, Liu GY, Li DD, Wang JL, Ji J (2012) Reversibly light-responsive micelles constructed via a simple modification of hyperbranched polymers with chromophores. Polymer 53:3695–3703
Epstein WW, Garrossian M (1987) P-Methoxyphenacyl esters as photodeblockable protecting groups for phosphates. Chem Commun 8:532–533
Cheng RD, Tian M, Sun SJ, Liu CH, Wang YP, Liu ZT, Liu ZW, Jiang JQ (2015) Light-triggered disruption of PAG-based amphiphilic random copolymer micelles. Langmuir 31:7758–7763
Okada K, Okamato K, Oda MA (1988) A new and practical method of decarboxylation: photosensitized decarboxylation of N-acyloxyphthalimides via electron-transfer mechanism. J Am Chem Soc 110:8736–8738
Bélékian D, Cassagnau P, Flat JJ, Quinebeche S, Autissier L, Bertin D, Siri D, Gigmes D, Guillaneuf Y, Chaumont P, Beyou E (2013) N-Acetoxy-phthalimide (NAPI) as a new H-abstracting agent at high temperature: application to the melt functionalization of polyethylene. Polym Chem 4:2676–2679
Ikbal M, Jana A, Pradeep Singh ND, Banerjee R, Dhara D (2011) Photoacid generators (PAGs) based on N-acyl-N-phenylhydroxylaminesfor carboxylic and sulfonic acids. Tetrahedron 67:3733–3742
Tian M, Cheng RD, Zhang J, Liu ZT, Liu ZW, Jiang JQ (2016) Amphiphilic polymer micellar disruption based on main-chain photodegradation. Langmuir 32:12–18
Yan B, Boyer JC, Branda NR, Zhao Y (2011) Near-infrared light-triggered dissociation of block copolymer micelles using upconverting nanoparticles. J Am Chem Soc 133:19714–11971
Zeng SJ, Wang HB, Lu W, Yi ZG, Rao LH, Liu G, Hao JH (2014) Dual-modal upconversion fluorescent/X-ray imaging using ligand-free hexagonal phase NaLuF4:Gd/Yb/Ernanorods for blood vessel visualization. Biomaterials 35:2934–2941
Wang F, Han Y, Lim CS, Lu YH, Wang J, Xu J, Chen HY, Zhang C, Hong MH, Liu XG (2010) Simultaneous phase and size control of upconversion nanocrystals through lanthanide doping. Nature 463:1061–1065
Chae KH, Cho HI, Kim YH, Yang UC (2012) Photo-crosslinking and negative-type micropattern formation of a polymeric photobase generator containing phthalimido carbamate groups. Eur Polym J 48:1186–1194
Liu B, Li CX, Cheng ZY, Hou ZY, Huang SS, Lin J (2016) Functional nanomaterials for near-infraredtriggered cancer therapy. Biomater Sci 4:890–909
Acknowledgements
This work is supported by the Nature Science Foundation of China (NSFC 21374056), the Program for Changjiang Scholars and Innovative Research Team in University (IRT 14R33), the Fundamental Research Funds for the Central Universities (GK201603038, GK201601005), Shaanxi Innovative Research Team for Key Science and Technology (2013KCT-17), and the One Hundred Plan of Shaanxi Province.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
We would like to declare that all of the authors in this work have no conflict of interest.
Electronic supplementary material
ESM 1
(DOCX 618 kb).
Rights and permissions
About this article
Cite this article
Wang, Y., Li, G., Cheng, R. et al. NIR- and UV-dual responsive amphiphilic copolymer micelles with light-dissociable PAG-side groups. Colloid Polym Sci 295, 371–378 (2017). https://doi.org/10.1007/s00396-017-4013-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00396-017-4013-0