Abstract
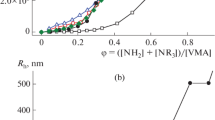
Polyelectrolyte complexes (PECs) were prepared from N,N,N-trimethylchitosan iodide (TMCh) of different molar mass and a weak polyacid-poly(acrylic acid) (PAA) or a strong polyacid-poly(2-acrylamido-2-methylpropanesulfonic acid) (PAMPS). The quaternization of the amino groups of chitosan enabled the formation of water-insoluble PECs in a broad pH range—from 3 to 12 and from 1 to 12 for TMCh/PAA and TMCh/PAMPS, respectively. Whereas the stoichiometry of the TMCh/PAA complex was pH dependant, the stoichiometry of the TMCh/PAMPS complex did not depend on pH. The stoichiometry and the yield of the complexes were influenced by the molar mass of TMCh. PEC nanoparticles were produced by mixing dilute solutions of the oppositely charged polyelectrolytes as revealed by dynamic light scattering analyses. The size of the particles was in the range of 135–924 nm and depended on the polyelectrolyte molar mass, the initial polyelectrolyte concentration, and the molar fraction of the TMCh units. Microbiological screening against Staphylococcus aureus and Escherichia coli revealed that PECs between TMCh and PAA or PAMPS have a good antibacterial effect, which is more slowly pronounced than that of the starting TMCh of different molar mass.







Similar content being viewed by others
References
Jin Z, Li W, Cao H, Zhang X, Chen G, Wu H, Guo C, Zhang Y, Kang H, Wang Y, Zhao K (2013) Antimicrobial activity and cytotoxicity of N-2-HACC and characterization of nanoparticles with N-2-HACC and CMC as a vaccine carrier. Chem Eng J 221:331–341
Briones AV, Sato T (2010) Encapsulation of glucose oxidase (GOD) in polyelectrolyte complexes of chitosan-carrageenan. React Funct Polym 70:19–27
Said AEHA (2005) Radiation synthesis of interpolymer polyelectrolyte complex and its application as a carrier for colon-specific drug delivery system. Biomaterials 26:2733–2739
Liu W, Sun S, Cao Z, Zhang X, Yao K, Lu WW, Luk KDK (2005) An investigation on the physicochemical properties of chitosan/DNA polyelectrolyte complexes. Biomaterials 26:2705–2711
Pirotton S, Muller C, Pantoustier N, Botteman F, Collinet S, Grandfils C, Dandrifosse G, Degée P, Dubois P, Raes M (2004) Enhancement of transfection efficiency through rapid and noncovalent post-PEGylation of poly(dimethylaminoethyl methacrylate)/DNA complexes. Pharm Res 21:1471–1479
Wolfert MA, Dash PR, Nazarova O, Oupicky D, Seymour LW, Smart S, Strohalm J, Ulbrich K (1999) Polyelectrolyte vectors for gene delivery: Influence of cationic polymer on biophysical properties of complexes formed with DNA. Bioconjug Chem 10:993–1004
Haisch A, Gröger A, Radke C, Ebmeyer J, Sudhoff H, Grasnick G, Jahnke V, Burmester GR, Sittinger M (2000) Macroencapsulation of human cartilage implants: pilot study with polyelectrolyte complex membrane encapsulation. Biomaterials 21:1561–1566
Domard A, Rinaudo M (1983) Preparation and characterization of fully deacetylated chitosan. Int J Biol Macromol 5:49–52
Domard A (1987) pH and c.d. measurements on a fully deacetylated chitosan: application to CuII-polymer interactions. Int J Biol Macromol 9:98–104
Gherghi IC, Girousi ST, Thanou M, Voulgaropoulos AN, Tzimou-Tsitouridou R (2005) Voltammetric study of interaction between polymers (PEI and TMO) and pDNA on a hanging mercury drop electrode. J Pharm Biomed Anal 39:177–180
Mao Z, Ma L, Yan J, Yan M, Gao C, Shen J (2007) The gene transfection efficiency of thermoresponsive N, N, N-trimethyl chitosan chloride-g-poly(N-isopropylacrylamide) copolymer. Biomaterials 28:4488–4500
Mao Z, Ma L, Jiang Y, Yan M, Gao C, Shen J (2007) N, N, N-Trimethylchitosan chloride as a gene vector: synthesis and application. Macromol Biosci 7:855–863
Guo R, Xu S, Ma L, Huang A, Gao C (2010) Enhanced angiogenesis of gene-activated dermal equivalent for treatment of full thickness incisional wounds in a porcine model. Biomaterials 31:7308–7320
Wang W, Li B, Li Y, Jiang Y, Ouyang H, Gao C (2010) In vivo restoration of full-thickness cartilage defects by poly(lactide-co-glycolide) sponges filled with fibrin gel, bone marrow mesenchymal stem cells and DNA complexes. Biomaterials 31:5953–5965
Liu X, Ma L, Liang J, Zhang B, Teng J, Gao C (2013) RNAi functionalized collagen-chitosan/silicone membrane bilayer dermal equivalent for full-thickness skin regeneration with inhibited scarring. Biomaterials 34:2038–2048
Martins AF, Bueno PVA, Almeida EAMS, Rodrigues FHA, Rubira AF, Muniz EC (2013) Characterization of N-trimethyl chitosan/alginate complexes and curcumin release. Int J Biol Macromol 57:174–184
Juntaprama K, Praphairaksit N, Siraleartmukul K, Muangsin N (2012) Electrosprayed polyelectrolyte complexes between mucoadhesive N, N, N-trimethylchitosan-homocysteine thiolactone and alginate/carrageenan for camptothecin delivery. Carbohydr Polym 90:1469–1479
Verheul RJ, Slütter B, Bal SM, Bouwstra JA, Jiskoot W, Hennink WE (2011) Covalently stabilized trimethyl chitosan-hyaluronic acid nanoparticles for nasal and intradermal vaccination. J Control Release 156:46–52
Martins AF, Pereira AGB, Fajardo AR, Rubira AF, Muniz EC (2011) Characterization of polyelectrolytes complexes based on N, N, N-trimethyl chitosan/heparin prepared at different pH conditions. Carbohydr Polym 86:1266–1272
Martins AF, Piai JF, Schuquel ITA, Rubira AF, Muniz EC (2011) Polyelectrolyte complexes of chitosan/heparin and N, N, N-trimethyl chitosan/heparin obtained at different pH: I. Preparation, characterization, and controlled release of heparin. Colloid Polym Sci 289:1133–1144
Sayın B, Somavarapu S, Li XW, Sesardic D, Senel S, Alpar OH (2009) TMC–MCC (N-trimethyl chitosan-mono-N-carboxymethyl chitosan) nanocomplexes for mucosal delivery of vaccines. Eur J Pharm Sci 38:362–369
Shu S, Zhang X, Wu Z, Wang Z, Li C (2011) Delivery of protein drugs using nanoparticles self-assembled from dextran sulfate and quaternized chitosan. J Control Release 152:133–191
Wang TW, Xu Q, Wu Y, Zeng AJ, Li M, Gao H (2009) Quaternized chitosan (QCS)/poly(aspartic acid) nanoparticles as a protein drug-delivery system. Carbohydr Res 344:908–914
Dehousse V, Garbacki N, Colige A, Evrard B (2010) Development of pH-responsive nanocarriers using trimethylchitosans and methacrylic acid copolymer for siRNA delivery. Biomaterials 31:1839–1849
Stoilova O, Koseva N, Manolova N, Rashkov I (1999) Polyelectrolyte complex between chitosan and poly(2-acryloylamido-2-methylpropanesulfonic acid). Polym Bull 43:67–73
Chavasit V, Klenzle-Sterzer C, Torres JA (1988) Formation and characterisation of an insoluble polyelectrolyte complex: chitosan-polyacrylic acid. Polym Bull 19:223–230
Mincheva R, Manolova N, Paneva D, Rashkov I (2006) Novel polyelectrolyte complexes between N-carboxyethylchitosan and synthetic polyelectroly. Eur Polym J 42:858–868
Stoilova O, Koseva N, Manolova N, Rashkov I (2001) Template polymerisation of 2-acryloylamido-2-methylpropanesulfonic acid onto chitosan. Bulg Chem Commun (Engl) 33:197–205
Paneva D, Stoilova O, Manolova N, Rashkov I (2003) Novel polyelectrolyte complex between chitosan and poly(2-acryloylamido-2-methylpropanesulfonic acid-co-acrylic acid). e-Polymers no. 033
Paneva D, Manolova N, Rashkov I, Danchev D (2005) Gel beads composed of chitosan and polyacids and their blood compatibility. J Bioact Compat Polym 20:133–151
Paneva D, Mespouille L, Manolova N, Degée P, Rashkov I, Dubois P (2006) Comprehensive study on the formation of polyelectrolyte complexes from (quaternized) poly[2-(dimethylamino)ethyl methacrylate] and poly(2-acrylamido-2-methylpropane sodium sulfonate). J Polym Sci A Polym Chem 44:5468–5479
Dragan E, Schwarz S (2004) Polyelectrolyte complexes. VI. Polycation structure, polyanion molar mass, and polyion concentration effects on complex nanoparticles based on poly(sodium 2-acrylamido-2-methylpropanesulfonate). J Polym Sci A: Polym Chem 42:2495–2505
Kalinov K, Ignatova M, Maximova V, Rashkov I, Manolova N (2014) Modification of electrospun poly(ε-caprolactone) mats by formation of a polyelectrolyte complex between poly(acrylic acid) and quaternized chitosan for tuning of their antibacterial properties. Eur Polym J 50:18–29
Kim CH, Choi JW, Chun HJ, Choi KS (1997) Synthesis of chitosan derivatives with quaternary ammonium salt and their antibacterial activity. Polym Bull 38:387–393
Polnok A, Borchard G, Verhoef JC, Sarisuta N, Junginger HE (2004) Influence of methylation process on the degree of quaternization of N-trimethyl chitosan chloride. Eur J Pharm Biopharm 57:77–83
Rúnarsson ÖV, Holappa J, Nevalainen T, Hjálmarsdóttir M, Järvinen T, Loftsson T, Einarsson JM, Jónsdóttir S, Valdimarsdóttir M, Másson M (2007) Antibacterial activity of methylated chitosan and chitooligomer derivatives: synthesis and structure activity relationships. Eur Polym J 43:2660–2671
Fisher L, Sochor A, Tan J (1977) Chain characteristics of poly(2-acrylamido-2-methylpropane sulfonate) polymers. 1. Light scattering and intrinsic viscosity studies. Macromolecules 10:949–954
Gicheva G, Paneva D, Manolova N, Naydenov M, Rashkov I (2012) New polyelectrolyte complex of chitosan: Preparation, characterization, and application as a biocontrol agent carrier. J Bioact Compat Polym 27:148–160
Dragan S, Dranca I, Ghimici L, Cristea M, Funduianu G, Lupasku T (1998) Thermal behaviour of some cationic polyelectrolytes and polyelectrolyte complexes. Eur Polym J 34:733–737
Hartig SM, Carlesso G, Davidson JM, Prokop A (2007) Development of improved nanoparticulate polyelectrolyte complex physicochemistry by nonstoichiometric mixing of polyions with similar molecular weights. Biomacromolecules 8:265–272
Kabanov KV, Bronich TK, Kabanov VA, Yu K, Eisenberg A (1996) Soluble stoichiometric complexes from poly(N-ethyl-4-vinylpyridinium) cations and poly(ethylene oxide)-block-polymethacrylate anions. Macromolecules 29:6797–6802
Popelka A, Novák I, Lehocký M, Junkar I, Mozetič M, Kleinová A, Janigová I, Šloufd M, Bilek F, Chodák I (2012) A new route for chitosan immobilization onto polyethylene surface. Carbohydr Polym 90:1501–1508
Gratzl G, Paulik C, Hild S, Guggenbichler JP, Lackner M (2014) Antimicrobial activity of poly(acrylic acid) block copolymers. Mater Sci Eng C 38:94–100
Yancheva E, Paneva D, Maximova V, Mespouille L, Dubois P, Manolova N, Rashkov I (2007) Polyelectrolyte complexes between (crosslinked) N-carboxyethylchitosan аnd (quaternized) poly[2-(dimethylamino)ethyl methacrylate]: preparation, characterization аnd antibacterial properties. Biomacromolecules 8:976–984
Sadeghi AMM, Dorkoosh FA, Avadi MR, Saadat P, Rafiee-Tehrani M, Junginger HE (2008) Preparation, characterization and antibacterial activities of chitosan, N-trimethyl chitosan (TMC) and N-diethylmethyl chitosan (DEMC) nanoparticles loaded with insulin using both the ionotropic gelation and polyelectrolyte complexation methods. Int J Pharm 355:299–306
Acknowledgments
Financial support from the National Science Fund (Grant DCVP 02/2–UNION) is kindly acknowledged. KK acknowledges the OP-HRD Grant BG051PO001-3.3.06-006 of the European Social Fund.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kalinov, K., Ignatova, M., Manolova, N. et al. N,N,N-trimethylchitosan iodide complexes with a weak or a strong polyacid and nanoparticles thereof. Colloid Polym Sci 292, 2899–2912 (2014). https://doi.org/10.1007/s00396-014-3325-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00396-014-3325-6