Abstract
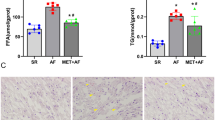
Atrial fibrillation (AF) is characterized by irregular contractions of atrial cardiomyocytes and increased energy demand. The aim of this study was to characterize the influence of arrhythmia on glucose and fatty acid (FA) metabolism in cardiomyocytes, mice and human left atrial myocardium. Compared to regular pacing, irregular (pseudo-random variation at the same number of contractions/min) pacing of neonatal rat cardiomyocytes induced shorter action potential durations and effective refractory periods and increased diastolic [Ca2+]c. This was associated with the activation of Ca2+/calmodulin-dependent protein kinase II (CaMKII) and AMP-activated protein kinase (AMPK). Membrane expression of fatty acid translocase (FAT/CD36) and 14C-palmitic acid uptake were augmented while membrane expression of glucose transporter subtype 4 (GLUT-4) as well as 3H-glucose uptake were reduced. Inhibition of AMPK and CaMKII prevented these arrhythmia-induced metabolic changes. Similar alterations of FA metabolism were observed in a transgenic mouse model (RacET) for spontaneous AF. Consistent with these findings samples of left atrial myocardium of patients with AF compared to matched samples of patients with sinus rhythm showed up-regulation of CaMKII and AMPK and increased membrane expression of FAT/CD36, resulting in lipid accumulation. These changes of FA metabolism were accompanied by decreased membrane expression of GLUT-4, increased glycogen content and increased expression of the pro-apoptotic protein bax. Irregular pacing of cardiomyocytes increases diastolic [Ca2+]c and activation of CaMKII and AMPK resulting in lipid accumulation, reduced glucose uptake and increased glycogen synthesis. These metabolic changes are accompanied by an activation of pro-apoptotic signalling pathways.












Similar content being viewed by others
References
Adam O, Frost G, Custodis F, Sussman MA, Schafers HJ, Böhm M, Laufs U (2007) Role of Rac1 GTPase activation in atrial fibrillation. J Am Coll Cardiol 50:359–367. doi:10.1016/j.jacc.2007.03.041
Adam O, Lavall D, Theobald K, Hohl M, Grube M, Ameling S, Sussman MA, Rosenkranz S, Kroemer HK, Schafers HJ, Böhm M, Laufs U (2010) Rac1-induced connective tissue growth factor regulates connexin 43 and N-cadherin expression in atrial fibrillation. J Am Coll Cardiol 55:469–480. doi:10.1016/j.jacc.2009.08.064
Adam O, Lohfelm B, Thum T, Gupta SK, Puhl SL, Schafers HJ, Böhm M, Laufs U (2012) Role of miR-21 in the pathogenesis of atrial fibrosis. Basic Res Cardiol 107:278. doi:10.1007/s00395-012-0278-0
Allard MF, Henning SL, Wambolt RB, Granleese SR, English DR, Lopaschuk GD (1997) Glycogen metabolism in the aerobic hypertrophied rat heart. Circulation 96:676–682
Allard MF, Schonekess BO, Henning SL, English DR, Lopaschuk GD (1994) Contribution of oxidative metabolism and glycolysis to ATP production in hypertrophied hearts. Am J Physiol 267:H742–H750
Allessie M, Ausma J, Schotten U (2002) Electrical, contractile and structural remodeling during atrial fibrillation. Cardiovasc Res 54:230–246
Arad M, Seidman CE, Seidman JG (2007) AMP-activated protein kinase in the heart: role during health and disease. Circ Res 100:474–488. doi:10.1161/01.RES.0000258446.23525.37
Ausma J, Wijffels M, Thone F, Wouters L, Allessie M, Borgers M (1997) Structural changes of atrial myocardium due to sustained atrial fibrillation in the goat. Circulation 96:3157–3163
Ausma J, Wijffels M, van Eys G, Koide M, Ramaekers F, Allessie M, Borgers M (1997) Dedifferentiation of atrial cardiomyocytes as a result of chronic atrial fibrillation. Am J Pathol 151:985–997
Bostrom P, Andersson L, Li L, Perkins R, Hojlund K, Boren J, Olofsson SO (2009) The assembly of lipid droplets and its relation to cellular insulin sensitivity. Biochem Soc Trans 37:981–985. doi:10.1042/BST0370981
de las Fuentes L, Herrero P, Peterson LR, Kelly DP, Gropler RJ, Davila-Roman VG (2003) Myocardial fatty acid metabolism: independent predictor of left ventricular mass in hypertensive heart disease. Hypertension 41:83–87
De Souza AI, Cardin S, Wait R, Chung YL, Vijayakumar M, Maguy A, Camm AJ, Nattel S (2010) Proteomic and metabolomic analysis of atrial profibrillatory remodelling in congestive heart failure. J Mol Cell Cardiol 49:851–863. doi:10.1016/j.yjmcc.2010.07.008
Dobrev D, Wehrens XH (2010) Calmodulin kinase II, sarcoplasmic reticulum Ca2+ leak, and atrial fibrillation. Trends Cardiovasc Med 20:30–34. doi:10.1016/j.tcm.2010.03.004
Dyck JR, Lopaschuk GD (2006) AMPK alterations in cardiac physiology and pathology: enemy or ally? J Physiol 574:95–112. doi:10.1113/jphysiol.2006.109389
Fischer Y, Thomas J, Sevilla L, Munoz P, Becker C, Holman G, Kozka IJ, Palacin M, Testar X, Kammermeier H, Zorzano A (1997) Insulin-induced recruitment of glucose transporter 4 (GLUT4) and GLUT1 in isolated rat cardiac myocytes. Evidence of the existence of different intracellular GLUT4 vesicle populations. J Biol Chem 272:7085–7092
Frederich M, Balschi JA (2002) The relationship between AMP-activated protein kinase activity and AMP concentration in the isolated perfused rat heart. J Biol Chem 277:1928–1932. doi:10.1074/jbc.M107128200
Goodwin GW, Taylor CS, Taegtmeyer H (1998) Regulation of energy metabolism of the heart during acute increase in heart work. J Biol Chem 273:29530–29539
Harada M, Nattel SN, Nattel S (2012) AMP-activated protein kinase: potential role in cardiac electrophysiology and arrhythmias. Circ Arrhythm Electrophysiol 5:860–867. doi:10.1161/CIRCEP.112.972265
Hardie DG, Hawley SA, Scott JW (2006) AMP-activated protein kinase–development of the energy sensor concept. J Physiol 574:7–15. doi:10.1113/jphysiol.2006.108944
Heid HW, Moll R, Schwetlick I, Rackwitz HR, Keenan TW (1998) Adipophilin is a specific marker of lipid accumulation in diverse cell types and diseases. Cell Tissue Res 294:309–321
Heijman J, Voigt N, Nattel S, Dobrev D (2014) Cellular and molecular electrophysiology of atrial fibrillation initiation, maintenance, and progression. Circ Res 114:1483–1499. doi:10.1161/CIRCRESAHA.114.302226
Heijman J, Voigt N, Wehrens XH, Dobrev D (2014) Calcium dysregulation in atrial fibrillation: the role of CaMKII. Front Pharmacol 5:30. doi:10.3389/fphar.2014.00030
Kaestner L, Scholz A, Hammer K, Vecerdea A, Ruppenthal S, Lipp P (2009) Isolation and genetic manipulation of adult cardiac myocytes for confocal imaging. J Vis Exp. doi:10.3791/1433
Kohlhaas M, Maack C (2010) Adverse bioenergetic consequences of Na + -Ca2 + exchanger-mediated Ca2 + influx in cardiac myocytes. Circulation 122:2273–2280. doi:10.1161/CIRCULATIONAHA.110.968057
Kuang M, Febbraio M, Wagg C, Lopaschuk GD, Dyck JR (2004) Fatty acid translocase/CD36 deficiency does not energetically or functionally compromise hearts before or after ischemia. Circulation 109:1550–1557. doi:10.1161/01.CIR.0000121730.41801.12
Laufs U, Kilter H, Konkol C, Wassmann S, Böhm M, Nickenig G (2002) Impact of HMG CoA reductase inhibition on small GTPases in the heart. Cardiovasc Res 53:911–920
Laufs U, Werner N, Link A, Endres M, Wassmann S, Jurgens K, Miche E, Böhm M, Nickenig G (2004) Physical training increases endothelial progenitor cells, inhibits neointima formation, and enhances angiogenesis. Circulation 109:220–226. doi:10.1161/01.CIR.0000109141.48980.37
Lavall D, Selzer C, Schuster P, Lenski M, Adam O, Schaefers HJ, Boehm M, Laufs U (2014) The mineralocorticoid receptor promotes fibrotic remodeling in atrial fibrillation. J Biol Chem. doi:10.1074/jbc.M113.519256
Lenski M, Kazakov A, Marx N, Böhm M, Laufs U (2011) Effects of DPP-4 inhibition on cardiac metabolism and function in mice. J Mol Cell Cardiol 51:906–918. doi:10.1016/j.yjmcc.2011.08.001
Li J, Zeng Z, Viollet B, Ronnett GV, McCullough LD (2007) Neuroprotective effects of adenosine monophosphate-activated protein kinase inhibition and gene deletion in stroke. Stroke 38:2992–2999. doi:10.1161/STROKEAHA.107.490904
Luiken JJ, Coort SL, Koonen DP, van der Horst DJ, Bonen A, Zorzano A, Glatz JF (2004) Regulation of cardiac long-chain fatty acid and glucose uptake by translocation of substrate transporters. Pflugers Arch 448:1–15. doi:10.1007/s00424-003-1199-4
Luiken JJ, Coort SL, Willems J, Coumans WA, Bonen A, van der Vusse GJ, Glatz JF (2003) Contraction-induced fatty acid translocase/CD36 translocation in rat cardiac myocytes is mediated through AMP-activated protein kinase signaling. Diabetes 52:1627–1634
Mak KM, Ren C, Ponomarenko A, Cao Q, Lieber CS (2008) Adipose differentiation-related protein is a reliable lipid droplet marker in alcoholic fatty liver of rats. Alcohol Clin Exp Res 32:683–689. doi:10.1111/j.1530-0277.2008.00624.x
McAuliffe JJ, Perry SB, Brooks EE, Ingwall JS (1991) Kinetics of the creatine kinase reaction in neonatal rabbit heart: an empirical analysis of the rate equation. Biochemistry 30:2585–2593
Morillo CA, Klein GJ, Jones DL, Guiraudon CM (1995) Chronic rapid atrial pacing. Structural, functional, and electrophysiological characteristics of a new model of sustained atrial fibrillation. Circulation 91:1588–1595
Muthusamy K, Halbert G, Roberts F (2006) Immunohistochemical staining for adipophilin, perilipin and TIP47. J Clin Pathol 59:1166–1170. doi:10.1136/jcp.2005.033381
Neef S, Dybkova N, Sossalla S, Ort KR, Fluschnik N, Neumann K, Seipelt R, Schondube FA, Hasenfuss G, Maier LS (2010) CaMKII-dependent diastolic SR Ca2+ leak and elevated diastolic Ca2+ levels in right atrial myocardium of patients with atrial fibrillation. Circ Res 106:1134–1144. doi:10.1161/CIRCRESAHA.109.203836
Peters CG, Miller DF, Giovannucci DR (2006) Identification, localization and interaction of SNARE proteins in atrial cardiac myocytes. J Mol Cell Cardiol 40:361–374. doi:10.1016/j.yjmcc.2005.12.007
Raney MA, Turcotte LP (2008) Evidence for the involvement of CaMKII and AMPK in Ca2+ -dependent signaling pathways regulating FA uptake and oxidation in contracting rodent muscle. J Appl Physiol 104:1366–1373. doi:10.1152/japplphysiol.01282.2007
Reil JC, Hohl M, Oberhofer M, Kazakov A, Kaestner L, Mueller P, Adam O, Maack C, Lipp P, Mewis C, Allessie M, Laufs U, Böhm M, Neuberger HR (2010) Cardiac Rac1 overexpression in mice creates a substrate for atrial arrhythmias characterized by structural remodelling. Cardiovasc Res 87:485–493. doi:10.1093/cvr/cvq079
Reilly SN, Jayaram R, Nahar K, Antoniades C, Verheule S, Channon KM, Alp NJ, Schotten U, Casadei B (2011) Atrial sources of reactive oxygen species vary with the duration and substrate of atrial fibrillation: implications for the antiarrhythmic effect of statins. Circulation 124:1107–1117. doi:10.1161/CIRCULATIONAHA.111.029223
Remondino A, Rosenblatt-Velin N, Montessuit C, Tardy I, Papageorgiou I, Dorsaz PA, Jorge-Costa M, Lerch R (2000) Altered expression of proteins of metabolic regulation during remodeling of the left ventricle after myocardial infarction. J Mol Cell Cardiol 32:2025–2034. doi:10.1006/jmcc.2000.1234
Schotten U, Verheule S, Kirchhof P, Goette A (2011) Pathophysiological mechanisms of atrial fibrillation: a translational appraisal. Physiol Rev 91:265–325. doi:10.1152/physrev.00031.2009
Stuck BJ, Lenski M, Böhm M, Laufs U (2008) Metabolic switch and hypertrophy of cardiomyocytes following treatment with angiotensin II are prevented by AMP-activated protein kinase. J Biol Chem 283:32562–32569. doi:10.1074/jbc.M801904200
Taegtmeyer H, Sen S, Vela D (2010) Return to the fetal gene program: a suggested metabolic link to gene expression in the heart. Ann N Y Acad Sci 1188:191–198. doi:10.1111/j.1749-6632.2009.05100.x
Tovar O, Tung L (1992) Electroporation and recovery of cardiac cell membrane with rectangular voltage pulses. Am J Physiol 263:H1128–H1136
Towler MC, Hardie DG (2007) AMP-activated protein kinase in metabolic control and insulin signaling. Circ Res 100:328–341. doi:10.1161/01.RES.0000256090.42690.05
Viollet B, Horman S, Leclerc J, Lantier L, Foretz M, Billaud M, Giri S, Andreelli F (2010) AMPK inhibition in health and disease. Crit Rev Biochem Mol Biol 45:276–295. doi:10.3109/10409238.2010.488215
Voigt N, Li N, Wang Q, Wang W, Trafford AW, Abu-Taha I, Sun Q, Wieland T, Ravens U, Nattel S, Wehrens XH, Dobrev D (2012) Enhanced sarcoplasmic reticulum Ca2+ leak and increased Na+ -Ca2+ exchanger function underlie delayed afterdepolarizations in patients with chronic atrial fibrillation. Circulation 125:2059–2070. doi:10.1161/CIRCULATIONAHA.111.067306
White CW, Holida MD, Marcus ML (1986) Effects of acute atrial fibrillation on the vasodilator reserve of the canine atrium. Cardiovasc Res 20:683–689
White CW, Kerber RE, Weiss HR, Marcus ML (1982) The effects of atrial fibrillation on atrial pressure-volume and flow relationships. Circ Res 51:205–215
Yue L, Feng J, Gaspo R, Li GR, Wang Z, Nattel S (1997) Ionic remodeling underlying action potential changes in a canine model of atrial fibrillation. Circ Res 81:512–525
Zhou G, Myers R, Li Y, Chen Y, Shen X, Fenyk-Melody J, Wu M, Ventre J, Doebber T, Fujii N, Musi N, Hirshman MF, Goodyear LJ, Moller DE (2001) Role of AMP-activated protein kinase in mechanism of metformin action. J Clin Invest 108:1167–1174. doi:10.1172/JCI13505
Zhou YT, Grayburn P, Karim A, Shimabukuro M, Higa M, Baetens D, Orci L, Unger RH (2000) Lipotoxic heart disease in obese rats: implications for human obesity. Proc Natl Acad Sci USA 97:1784–1789
Acknowledgments
We thank Ellen Becker, Simone Jäger and Catrin Pittke for their excellent technical assistance. This study was funded by grants of the Deutsche Forschungsgemeinschaft (DFG) to UL, OA, CM, MB), the Deutsche Gesellschaft für Kardiologie (DGK) to ML, the HOMFOR Programm to ML, OA, and the Universität des Saarlandes to ML, OA.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
To this original contribution an invited editorial is available at doi:10.1007/s00395-015-0498-1.
Rights and permissions
About this article
Cite this article
Lenski, M., Schleider, G., Kohlhaas, M. et al. Arrhythmia causes lipid accumulation and reduced glucose uptake. Basic Res Cardiol 110, 40 (2015). https://doi.org/10.1007/s00395-015-0497-2
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00395-015-0497-2