Abstract
Purpose
Muffins made with wheat flour are a popular snack consumed in western and emerging countries. This study aimed to examine the content of amylose, glycemic response (GR) and glycemic index (GI) of muffins baked with refined wheat and rice flours, as well as wholegrain corn, oat and barley flours.
Methods
This study adopted a randomized, controlled, crossover, non-blind design. Twelve healthy participants consumed wheat, rice, corn, oat and barley muffins once and the reference glucose solution three times in a random order on non-consecutive day. Capillary blood samples were taken every 15 min in the first 60 min and every 30 min for the remaining 60 min for blood glucose analysis. The Megazyme amylose/amylopectin assay procedure was employed to measure amylose content.
Results
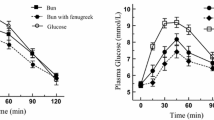
The GR elicited from the consumption of wheat, rice and corn muffins was comparable between these samples but significantly greater when compared with oat and barley muffins. Consumption of wholegrain muffins, apart from corn muffin, blunted postprandial GR when compared with muffins baked with refined cereal flours. Muffins baked with wheat, rice, corn, oat and barley flours gave rise to GI values of 74, 79, 74, 53 and 55, respectively. The content of amylose was significantly higher in corn, oat and barley muffins than wheat and rice muffins.
Conclusions
The greater content of amylose and fibre may play a part in the reduced glycemic potency of oat and barley muffins. Wheat flour can be substituted with oat and barley flours for healthier muffins and other bakery products.


Similar content being viewed by others
References
Singh J, Dartois A, Kaur L (2010) Starch digestibility in food matrix: a review. Trends Food Sci Tech 21:168–180
Salmerón J, Manson JE, Stampfer MJ, Colditz GA, Wing AL, Willett WC (1997) Dietary fiber, glycemic load, and risk of non-insulin-dependent diabetes mellitus in women. JAMA 277:472–477
Soong YY, Tan SP, Leong LP, Henry JK (2014) Total antioxidant capacity and starch digestibility of muffins baked with rice, wheat, oat, corn and barley flour. Food Chem 164:462–469
Englyst KN, Englyst HN, Hudson GJ, Cole TJ, Cummings JH (1999) Rapidly available glucose in foods: an in vitro measurement that reflects the glycemic response. Am J Clin Nutr 69:448–454
Henry CJ, Ranawana V (2012) Sugar: a problem of developed countries. Nature 482:471
Ludwig DS (2002) The glycemic index: physiological mechanisms relating to obesity, diabetes, and cardiovascular disease. JAMA 287:2414–2423
Baecke JA, Burema J, Frijters JE (1982) A short questionnaire for the measurement of habitual physical activity in epidemiological studies. Am J Clin Nutr 36:936–942
Brouns F, Bjorck I, Frayn KN, Gibbs AL, Lang V, Slama G et al (2005) Glycaemic index methodology. Nutr Res Rev 18:145–171
FAO WHO (1998) Carbohydrates in human nutrition—report of a joint FAO/WHO expert consultation. Food and Agriculture Organisation, Rome
Stork AD, Kemperman H, Erkelens DW, Veneman TF (2005) Comparison of the accuracy of the HemoCue glucose analyzer with the yellow springs instrument glucose oxidase analyzer, particularly in hypoglycemia. Eur J Endocrinol 153:275–281
Wolever TMS (2006) The glycaemic index: a physiological classification of dietary carbohydrate. CABI, Oxfordshire
Monro JA, Mishra S, Venn B (2010) Baselines representing blood glucose clearance improve in vitro prediction of the glycemic impact of customarily consumed food quantities. Br J Nutr 103:295–305
Regand A, Chowdhury Z, Tosh SM, Wolever T, Wood P (2011) The molecular weight, solubility and viscosity of oat beta-glucan affect human glycemic response by modifying starch digestibility. Food Chem 129:297–304
Banchathanaki R, Suphantharika M (2009) Effect of different β-glucans on the gelatinisation and retrogradation of rice starch. Food Chem 114:5–14
Parada J, Aguilera JM (2009) In vitro digestibility and glycemic response of potato starch is related to granule size and degree of gelatinization. J Food Sci 74:E34–E38
Viebke C, Al-Assaf S, Phillips GO (2014) Food hydrocolloids and health claims. Bioact Carbohydr Diet Fibre 4:101–114
Chung SY, Lee SW, Rhee C (2011) Effects of various Maillard reaction products on in vitro starch hydrolysis and blood glucose responses in mice. Starch-Stärke 63:443–449
Langworthy CF, Deuel HJ (1922) Digestibility of raw rice, arrowroot, canna, cassava, taro, tree-fern, and potato starches. J Biol Chem 52:251–261
van de Velde F, van Riel J, Tromp RH (2002) Visualisation of starch granule morphologies using confocal scanning laser microscopy (CSLM). J Sci Food Agric 82:1528–1536
Hargrove JL, Greenspan P, Hartle DK, Dowd C (2011) Inhibition of aromatase and α-amylase by flavonoids and proanthocyanidins from Sorghum bicolor bran extracts. J Med Food 14:799–807
Acknowledgments
Funding for the study was provided by the Singapore Institute for Clinical Sciences, A*Star.
Conflict of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Soong, Y.Y., Quek, R.Y.C. & Henry, C.J. Glycemic potency of muffins made with wheat, rice, corn, oat and barley flours: a comparative study between in vivo and in vitro. Eur J Nutr 54, 1281–1285 (2015). https://doi.org/10.1007/s00394-014-0806-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00394-014-0806-9