Abstract
Background
Immunotherapy focuses on selectively enhancing the host’s immune response against malignant disease. It has been investigated as an important treatment modality against malignant disease for many years, but until recently its use was mostly limited to a few cancers. The advent of new immunemodulating agents in the recent past has changed the landscape for management of many solid tumors. Currently, immunotherapy offers a valuable, and in many cases, a more effective alternate to the conventional cytotoxic therapy. Colorectal cancer is a leading cause of cancer-related death. Despite progress in systemic therapy, most patients with metastatic colorectal cancer die of their disease. There is an unmet need for more effective treatments for patients with metastatic colorectal cancer. The current data support that colorectal tumors are immunoresponsive and a subset of patients with advanced disease achieve long term benefit with immunotherapy.
Objectives
This review aims to provide the current status of immunotherapy in patients with metastatic colorectal cancer.
Methods
We researched sources published in the English language between January 2000 and August 2018 and listed within the PubMed database using combinations of the key words and reviewed the proceedings of international cancer conferences and current guidelines made by major cancer societies.
Results
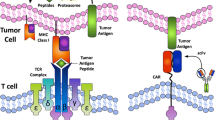
In this review, we summarize the current status of research on immunotherapy in metastatic colorectal cancer and discuss various treatment modalities including checkpoint inhibitors, cancer vaccines, adoptive cell transfer, oncolytic virus therapy, and various other agents that are under investigation with a special emphasis on immune checkpoint inhibitors. Since the toxicity profile of immunotherapy is very different from conventional cytotoxic agents and could involve any organ system, we briefly review common adverse effects and their management.

Similar content being viewed by others
References
van der Geest LG, Lam-Boer J, Koopman M et al (2015) Nationwide trends in incidence, treatment and survival of colorectal cancer patients with synchronous metastases. Clin Exp Metastasis 32:457–465
Jemal A, Siegel R, Ward E, Hao Y, Xu J, Murray T, Thun MJ (2008) Cancer statistics, 2008. CA Cancer J Clin 58:71–96
Siegel RL, Miller KD, Fedewa SA, Ahnen DJ, Meester RGS, Barzi A, Jemal A (2017) Colorectal cancer statistics, 2017. CA Cancer J Clin 67:177–193
Ahmed S, Johnson K, Ahmed O, Iqbal N (2014) Advances in the management of colorectal cancer: from biology to treatment. Int J Color Dis 29:1031–1042
Cremolini C, Schirripa M, Antoniotti C, Moretto R, Salvatore L, Masi G, Falcone A, Loupakis F (2015) First-line chemotherapy for mCRC—a review and evidence-based algorithm. Nat Rev Clin Oncol 12:607–619
Kohne CH, van Cutsem E, Wils J et al (2005) Phase III study of weekly high-dose infusional fluorouracil plus folinic acid with or without irinotecan in patients with metastatic colorectal cancer: European Organisation for Research and Treatment of Cancer Gastrointestinal Group Study 40986. J Clin Oncol 23:4856–4865
Giacchetti S, Perpoint B, Zidani R, le Bail N, Faggiuolo R, Focan C, Chollet P, Llory JF, Letourneau Y, Coudert B, Bertheaut-Cvitkovic F, Larregain-Fournier D, le Rol A, Walter S, Adam R, Misset JL, Lévi F (2000) Phase III multicenter randomized trial of oxaliplatin added to chronomodulated fluorouracil-leucovorin as first-line treatment of metastatic colorectal cancer. J Clin Oncol 18:136–147
Tournigand C, Andre T, Achille E et al (2004) FOLFIRI followed by FOLFOX6 or the reverse sequence in advanced colorectal cancer: a randomized GERCOR study. J Clin Oncol 22:229–237
Colucci G, Gebbia V, Paoletti G, Giuliani F, Caruso M, Gebbia N, Cartenì G, Agostara B, Pezzella G, Manzione L, Borsellino N, Misino A, Romito S, Durini E, Cordio S, di Seri M, Lopez M, Maiello E, Montemurro S, Cramarossa A, Lorusso V, di Bisceglie M, Chiarenza M, Valerio MR, Guida T, Leonardi V, Pisconti S, Rosati G, Carrozza F, Nettis G, Valdesi M, Filippelli G, Fortunato S, Mancarella S, Brunetti C, Gruppo Oncologico Dell’Italia Meridionale (2005) Phase III randomized trial of FOLFIRI versus FOLFOX4 in the treatment of advanced colorectal cancer: a multicenter study of the Gruppo Oncologico Dell’Italia Meridionale. J Clin Oncol 23:4866–4875
Feliu J, Salud A, Escudero P et al (2006) XELOX (capecitabine plus oxaliplatin) as first-line treatment for elderly patients over 70 years of age with advanced colorectal cancer. Br J Cancer 94:969–975
Hurwitz H, Fehrenbacher L, Novotny W, Cartwright T, Hainsworth J, Heim W, Berlin J, Baron A, Griffing S, Holmgren E, Ferrara N, Fyfe G, Rogers B, Ross R, Kabbinavar F (2004) Bevacizumab plus irinotecan, fluorouracil, and leucovorin for metastatic colorectal cancer. N Engl J Med 350:2335–2342
Cunningham D, Humblet Y, Siena S, Khayat D, Bleiberg H, Santoro A, Bets D, Mueser M, Harstrick A, Verslype C, Chau I, van Cutsem E (2004) Cetuximab monotherapy and cetuximab plus irinotecan in irinotecan-refractory metastatic colorectal cancer. N Engl J Med 351:337–345
Jonker DJ, O'Callaghan CJ, Karapetis CS, Zalcberg JR, Tu D, Au HJ, Berry SR, Krahn M, Price T, Simes RJ, Tebbutt NC, van Hazel G, Wierzbicki R, Langer C, Moore MJ (2007) Cetuximab for the treatment of colorectal cancer. N Engl J Med 357:2040–2048
Mayer RJ, Van Cutsem E, Falcone A et al (2015) Randomized trial of TAS-102 for refractory metastatic colorectal cancer. N Engl J Med 372:1909–1919
Grothey A, Van Cutsem E, Sobrero A et al (2013) Regorafenib monotherapy for previously treated metastatic colorectal cancer (CORRECT): an international, multicentre, randomised, placebo-controlled, phase 3 trial. Lancet 381:303–312
IQWiG (2015) [A15–43] Regorafenib - Nutzenbewertung gemäß § 35a SGB V. Institut für Qualität und Wirtschaftlichkeit im Gesundheitswesen
Merika E, Saif MW, Katz A, Syrigos K, Morse M (2010) Review. Colon cancer vaccines: an update. In Vivo 24:607–628
Leslie A, Carey FA, Pratt NR, Steele RJC (2002) The colorectal adenoma-carcinoma sequence. Br J Surg 89:845–860
Vogelstein B, Kinzler KW (2004) Cancer genes and the pathways they control. Nat Med 10:789–799
Lievre A, Bachet JB, Le Corre D et al (2006) KRAS mutation status is predictive of response to cetuximab therapy in colorectal cancer. Cancer Res 66:3992–3995
Network NCC (2018) NCCN Clinical Practice Guidelines in Oncology—colon cancer (Version 2.2018)
Akizawa Y, Yamamoto T, Tamura K, Kanno T, Takahashi N, Ohki T, Omori T, Tokushige K, Yamamoto M, Saito K (2018) A novel MLH1 mutation in a Japanese family with Lynch syndrome associated with small bowel cancer. Hum Genome Var 5:13
Dawood S (2018) The evolving role of immune oncology in colorectal cancer. Chin Clin Oncol 7:17
Vu T, Datta PK (2017) Regulation of EMT in colorectal cancer: a culprit in metastasis. Cancers (Basel) 9
Knosel T, Schluns K, Stein U et al (2004) Chromosomal alterations during lymphatic and liver metastasis formation of colorectal cancer. Neoplasia 6:23–28
Knosel T, Petersen S, Schwabe H et al (2002) Incidence of chromosomal imbalances in advanced colorectal carcinomas and their metastases. Virchows Arch 440:187–194
Tabernero J, Melero I, Ros W, Argiles G, Marabelle A, Rodriguez-Ruiz ME, Albanell J, Calvo E, Moreno V, Cleary JM, Eder JP, Karanikas V, Bouseida S, Sandoval F, Sabanes D, Sreckovic S, Hurwitz H, Paz-Ares LG, Saro Suarez JM, Segal NH (2017) Phase Ia and Ib studies of the novel carcinoembryonic antigen (CEA) T-cell bispecific (CEA CD3 TCB) antibody as a single agent and in combination with atezolizumab: preliminary efficacy and safety in patients with metastatic colorectal cancer (mCRC). J Clin Oncol 35:3002–3002
Kaufman HL, Lenz HJ, Marshall J, Singh D, Garett C, Cripps C, Moore M, von Mehren M, Dalfen R, Heim WJ, Conry RM, Urba WJ, Benson AB, Yu M, Caterini J, Kim-Schulze S, DeBenedette M, Salha D, Vogel T, Elias I, Berinstein NL (2008) Combination chemotherapy and ALVAC-CEA/B7.1 vaccine in patients with metastatic colorectal cancer. Clin Cancer Res 14:4843–4849
Cameron F, Whiteside G, Perry C (2011) Ipilimumab: first global approval. Drugs 71:1093–1104
Hodi FS, O'Day SJ, McDermott DF et al (2010) Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med 363:711–723
Pardoll DM (2012) The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer 12:252–264
Blank C, Brown I, Peterson AC, Spiotto M, Iwai Y, Honjo T, Gajewski TF (2004) PD-L1/B7H-1 inhibits the effector phase of tumor rejection by T cell receptor (TCR) transgenic CD8+ T cells. Cancer Res 64:1140–1145
Schwartz RH (1992) Costimulation of T lymphocytes: the role of CD28, CTLA-4, and B7/BB1 in interleukin-2 production and immunotherapy. Cell 71:1065–1068
Haanen JB, Robert C (2015) Immune checkpoint inhibitors. Prog Tumor Res 42:55–66
Schneider H, Mandelbrot DA, Greenwald RJ, Ng F, Lechler R, Sharpe AH, Rudd CE (2002) Cutting edge: CTLA-4 (CD152) differentially regulates mitogen-activated protein kinases (extracellular signal-regulated kinase and c-Jun N-terminal kinase) in CD4+ T cells from receptor/ligand-deficient mice. J Immunol 169:3475–3479
Linsley PS, Greene JL, Brady W et al (1994) Human B7-1 (CD80) and B7-2 (CD86) bind with similar avidities but distinct kinetics to CD28 and CTLA-4 receptors. Immunity 1:793–801
Xu-Monette ZY, Zhang M, Li J, Young KH (2017) PD-1/PD-L1 blockade: have we found the key to unleash the antitumor immune response? Front Immunol 8:1597
Herbst RS, Soria JC, Kowanetz M, Fine GD, Hamid O, Gordon MS, Sosman JA, McDermott DF, Powderly JD, Gettinger SN, Kohrt HEK, Horn L, Lawrence DP, Rost S, Leabman M, Xiao Y, Mokatrin A, Koeppen H, Hegde PS, Mellman I, Chen DS, Hodi FS (2014) Predictive correlates of response to the anti-PD-L1 antibody MPDL3280A in cancer patients. Nature 515:563–567
Zak KM, Grudnik P, Guzik K, Zieba BJ, Musielak B, Dömling A, Dubin G, Holak TA (2016) Structural basis for small molecule targeting of the programmed death ligand 1 (PD-L1). Oncotarget 7:30323–30335
Iwai Y, Hamanishi J, Chamoto K, Honjo T (2017) Cancer immunotherapies targeting the PD-1 signaling pathway. J Biomed Sci 24:26
Le DT, Uram JN, Wang H et al (2015) PD-1 blockade in tumors with mismatch-repair deficiency. N Engl J Med 372:2509–2520
Le DT, Durham JN, Smith KN et al (2017) Mismatch repair deficiency predicts response of solid tumors to PD-1 blockade. Science 357:409–413
Overman MJ, McDermott R, Leach JL, Lonardi S, Lenz HJ, Morse MA, Desai J, Hill A, Axelson M, Moss RA, Goldberg MV, Cao ZA, Ledeine JM, Maglinte GA, Kopetz S, André T (2017) Nivolumab in patients with metastatic DNA mismatch repair-deficient or microsatellite instability-high colorectal cancer (CheckMate 142): an open-label, multicentre, phase 2 study. Lancet Oncol 18:1182–1191
Bendell J, Ciardiello F, Tabernero J, et al (2018) LBA-004 efficacy and safety results from IMblaze370, a randomised Phase III study comparing atezolizumab+cobimetinib and atezolizumab monotherapy vs regorafenib in chemotherapy-refractory metastatic colorectal cancer. Ann Oncol 29:mdy208.003-mdy208.003
Overman MJ, Lonardi S, Wong KYM, Lenz HJ, Gelsomino F, Aglietta M, Morse MA, van Cutsem E, McDermott R, Hill A, Sawyer MB, Hendlisz A, Neyns B, Svrcek M, Moss RA, Ledeine JM, Cao ZA, Kamble S, Kopetz S, André T (2018) Durable clinical benefit with nivolumab plus ipilimumab in DNA mismatch repair-deficient/microsatellite instability-high metastatic colorectal cancer. J Clin Oncol 36:773–779
Chu JN, Choi J, Ostvar S, Torchia JA, Reynolds KL, Tramontano A, Gainor JF, Chung DC, Clark JW, Hur C (2018) Cost-effectiveness of immune checkpoint inhibitors for microsatellite instability-high/mismatch repair-deficient metastatic colorectal cancer. Cancer
Parseghian CM, Patnana M, Bhosale P, Hess KR, Shih YT, Kim B, Kopetz S, Overman MJ, Varadhachary GR, Javle M, Naing A, Piha-Paul S, Hong D, le H, Subbiah V, Pant S (2018) Evaluating for pseudoprogression in colorectal and pancreatic tumors treated with immunotherapy. J Immunother 41:284–291
Bearz A, Perin T, Cancian L et al (2016) Immune checkpoint inhibitors and response analysis: a tough challenge. A case report. BMC Res Notes 9:349
Vermorken JB, Claessen AM, van Tinteren H et al (1999) Active specific immunotherapy for stage II and stage III human colon cancer: a randomised trial. Lancet 353:345–350
Schulze T, Kemmner W, Weitz J, Wernecke KD, Schirrmacher V, Schlag PM (2009) Efficiency of adjuvant active specific immunization with Newcastle disease virus modified tumor cells in colorectal cancer patients following resection of liver metastases: results of a prospective randomized trial. Cancer Immunol Immunother 58:61–69
Inoda S, Morita R, Hirohashi Y, Torigoe T, Asanuma H, Nakazawa E, Nakatsugawa M, Tamura Y, Kamiguchi K, Tsuruma T, Terui T, Ishitani K, Hashino S, Wang Q, Greene MI, Hasegawa T, Hirata K, Asaka M, Sato N (2011) The feasibility of Cep55/c10orf3 derived peptide vaccine therapy for colorectal carcinoma. Exp Mol Pathol 90:55–60
Hazama S, Nakamura Y, Tanaka H, Hirakawa K, Tahara K, Shimizu R, Ozasa H, Etoh R, Sugiura F, Okuno K, Furuya T, Nishimura T, Sakata K, Yoshimatsu K, Takenouchi H, Tsunedomi R, Inoue Y, Kanekiyo S, Shindo Y, Suzuki N, Yoshino S, Shinozaki H, Kamiya A, Furukawa H, Yamanaka T, Fujita T, Kawakami Y, Oka M (2014) A phase IotaI study of five peptides combination with oxaliplatin-based chemotherapy as a first-line therapy for advanced colorectal cancer (FXV study). J Transl Med 12:108
Morse MA, Deng Y, Coleman D, Hull S, Kitrell-Fisher E, Nair S, Schlom J, Ryback ME, Lyerly HK (1999) A phase I study of active immunotherapy with carcinoembryonic antigen peptide (CAP-1)-pulsed, autologous human cultured dendritic cells in patients with metastatic malignancies expressing carcinoembryonic antigen. Clin Cancer Res 5:1331–1338
Caballero-Banos M, Benitez-Ribas D, Tabera J et al (2016) Phase II randomised trial of autologous tumour lysate dendritic cell plus best supportive care compared with best supportive care in pre-treated advanced colorectal cancer patients. Eur J Cancer 64:167–174
Yagiz K, Rodriguez-Aguirre ME, Lopez Espinoza F, Montellano TT, Mendoza D, Mitchell LA, Ibanez CE, Kasahara N, Gruber HE, Jolly DJ, Robbins JM (2018) A retroviral replicating vector encoding cytosine deaminase and 5-FC induces immune memory in metastatic colorectal cancer models. Mol Ther Oncolytics 8:14–26
Ruella M, Kalos M (2014) Adoptive immunotherapy for cancer. Immunol Rev 257:14–38
Turin I, Delfanti S, Ferulli F, Brugnatelli S, Tanzi M, Maestri M, Cobianchi L, Lisini D, Luinetti O, Paulli M, Perotti C, Todisco E, Pedrazzoli P, Montagna D (2018) In vitro killing of colorectal carcinoma cells by autologous activated NK cells is boosted by anti-epidermal growth factor receptor-induced ADCC regardless of RAS mutation status. J Immunother 41:190–200
Veluchamy JP, Lopez-Lastra S, Spanholtz J et al (2017) In vivo efficacy of umbilical cord blood stem cell-derived NK cells in the treatment of metastatic colorectal cancer. Front Immunol 8:87
Veluchamy JP, Spanholtz J, Tordoir M, Thijssen VL, Heideman DAM, Verheul HMW, de Gruijl TD, van der Vliet HJ (2016) Combination of NK cells and cetuximab to enhance anti-tumor responses in RAS mutant metastatic colorectal cancer. PLoS One 11:e0157830
Magee MS, Abraham TS, Baybutt TR, Flickinger JC Jr, Ridge NA, Marszalowicz GP, Prajapati P, Hersperger AR, Waldman SA, Snook AE (2018) Human GUCY2C-targeted chimeric antigen receptor (CAR)-expressing T cells eliminate colorectal cancer metastases. Cancer Immunol Res 6:509–516
Geevarghese SK, Geller DA, de Haan HA, Hörer M, Knoll AE, Mescheder A, Nemunaitis J, Reid TR, Sze DY, Tanabe KK, Tawfik H (2010) Phase I/II study of oncolytic herpes simplex virus NV1020 in patients with extensively pretreated refractory colorectal cancer metastatic to the liver. Hum Gene Ther 21:1119–1128
Weng T, Qiu X, Wang J, Li Z, Bian J (2018) Recent discovery of indoleamine-2,3-dioxygenase 1 inhibitors targeting cancer immunotherapy. Eur J Med Chem 143:656–669
Redmond WL, Ruby CE, Weinberg AD (2009) The role of OX40-mediated co-stimulation in T-cell activation and survival. Crit Rev Immunol 29:187–201
Bulliard Y, Jolicoeur R, Zhang J, Dranoff G, Wilson NS, Brogdon JL (2014) OX40 engagement depletes intratumoral Tregs via activating FcgammaRs, leading to antitumor efficacy. Immunol Cell Biol 92:475–480
Linch SN, Redmond WL (2014) Combined OX40 ligation plus CTLA-4 blockade: more than the sum of its parts. Oncoimmunology 3:e28245
Guo Z, Wang X, Cheng D, Xia Z, Luan M, Zhang S (2014) PD-1 blockade and OX40 triggering synergistically protects against tumor growth in a murine model of ovarian cancer. PLoS One 9:e89350
Hsu HC, Thiam TK, Lu YJ et al (2016) Mutations of KRAS/NRAS/BRAF predict cetuximab resistance in metastatic colorectal cancer patients. Oncotarget 7:22257–22270
Dudley JC, Lin MT, Le DT et al (2016) Microsatellite instability as a biomarker for PD-1 blockade. Clin Cancer Res 22:813–820
Kim H, Jen J, Vogelstein B, Hamilton SR (1994) Clinical and pathological characteristics of sporadic colorectal carcinomas with DNA replication errors in microsatellite sequences. Am J Pathol 145:148–156
Angelova M, Charoentong P, Hackl H, Fischer ML, Snajder R, Krogsdam AM, Waldner MJ, Bindea G, Mlecnik B, Galon J, Trajanoski Z (2015) Characterization of the immunophenotypes and antigenomes of colorectal cancers reveals distinct tumor escape mechanisms and novel targets for immunotherapy. Genome Biol 16:64
Xie QK, He WZ, Hu WM et al (2018) Tumor-infiltrating lymphocyte as a prognostic biomarker in stage IV colorectal cancer should take into account the metastatic status and operation modality. Cancer Manag Res 10:1365–1375
Galon J, Costes A, Sanchez-Cabo F, Kirilovsky A, Mlecnik B, Lagorce-Pagès C, Tosolini M, Camus M, Berger A, Wind P, Zinzindohoué F, Bruneval P, Cugnenc PH, Trajanoski Z, Fridman WH, Pagès F (2006) Type, density, and location of immune cells within human colorectal tumors predict clinical outcome. Science 313:1960–1964
Couzin J (2006) Cancer. T cells a boon for colon cancer prognosis. Science 313:1868–1869
Guidoboni M, Gafa R, Viel A et al (2001) Microsatellite instability and high content of activated cytotoxic lymphocytes identify colon cancer patients with a favorable prognosis. Am J Pathol 159:297–304
Pages F, Mlecnik B, Marliot F et al (2018) International validation of the consensus Immunoscore for the classification of colon cancer: a prognostic and accuracy study. Lancet 391:2128–2139
Villadolid J, Amin A (2015) Immune checkpoint inhibitors in clinical practice: update on management of immune-related toxicities. Transl Lung Cancer Res 4:560–575
Prieux-Klotz C, Dior M, Damotte D, Dreanic J, Brieau B, Brezault C, Abitbol V, Chaussade S, Coriat R (2017) Immune checkpoint inhibitor-induced colitis: diagnosis and management. Target Oncol 12:301–308
Zen Y, Yeh MM (2018) Hepatotoxicity of immune checkpoint inhibitors: a histology study of seven cases in comparison with autoimmune hepatitis and idiosyncratic drug-induced liver injury. Mod Pathol 31:965–973
Kao JC, Brickshawana A, Liewluck T (2018) Neuromuscular complications of programmed cell death-1 (PD-1) inhibitors. Curr Neurol Neurosci Rep 18:63
Champiat S, Lambotte O, Barreau E, Belkhir R, Berdelou A, Carbonnel F, Cauquil C, Chanson P, Collins M, Durrbach A, Ederhy S, Feuillet S, François H, Lazarovici J, le Pavec J, de Martin E, Mateus C, Michot JM, Samuel D, Soria JC, Robert C, Eggermont A, Marabelle A (2016) Management of immune checkpoint blockade dysimmune toxicities: a collaborative position paper. Ann Oncol 27:559–574
Brahmer JR, Lacchetti C, Schneider BJ, Atkins MB, Brassil KJ, Caterino JM, Chau I, Ernstoff MS, Gardner JM, Ginex P, Hallmeyer S, Holter Chakrabarty J, Leighl NB, Mammen JS, McDermott DF, Naing A, Nastoupil LJ, Phillips T, Porter LD, Puzanov I, Reichner CA, Santomasso BD, Seigel C, Spira A, Suarez-Almazor ME, Wang Y, Weber JS, Wolchok JD, Thompson JA, in collaboration with the National Comprehensive Cancer Network (2018) Management of immune-related adverse events in patients treated with immune checkpoint inhibitor therapy: American Society of Clinical Oncology clinical practice guideline. J Clin Oncol 36:1714–1768
Overman MJ, Ernstoff MS, Morse MA (2018) Where we stand with immunotherapy in colorectal cancer: deficient mismatch repair, proficient mismatch repair, and toxicity management. American Society of Clinical Oncology Educational Book, pp 239–247
Sattar J, Kartolo A, Hopman WM et al (2018) The efficacy and toxicity of immune checkpoint inhibitors in a real-world older patient population. J Geriatr Oncol
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wrobel, P., Ahmed, S. Current status of immunotherapy in metastatic colorectal cancer. Int J Colorectal Dis 34, 13–25 (2019). https://doi.org/10.1007/s00384-018-3202-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00384-018-3202-8