Abstract
Purpose
Fetal repair of spina bifida results in improved outcomes and has therefore become a standard clinical procedure in some highly specialized centers. However, optimization of the procedure technique and timing is needed. Both might be achieved by facilitating the procedure using laboratory-grown fetal skin substitutes. The aim of this study was therefore to test in vivo the suitability of such a fetal skin substitute for an in utero application.
Methods
Collagen-based hydrogels containing fetal ovine fibroblasts were seeded with fetal ovine keratinocytes and transplanted on immuno-incompetent nu/nu rats. After 3 weeks, grafts were harvested and analyzed histologically and by immunohistochemistry.
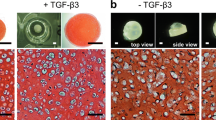
Results
Laboratory-grown fetal ovine dermo-epidermal skin substitutes showed successful engraftment at 3 weeks. Histologically, grafts revealed a neo-dermis populated by fibroblasts and with ingrowth of vessels, and an epidermis with an adult-like, mature appearance depicting clearly basal, spinous, granular, and a corneal layer. Immunostaining confirmed a physiologically organized epidermis.
Conclusion
Fetal dermo-epidermal skin substitutes of ovine origin can successfully be grafted in vivo. In a next step, we will have to test whether favorable results can also be obtained when grafts are used in utero. If so, then human fetal spina bifida repair using laboratory-grown autologous fetal skin for defect closure may be envisaged.






Similar content being viewed by others
References
Atala AAJ (2014) Translational regenerative medicine, 1st edn. Academic Press, New York
Adzick NS et al (2011) A randomized trial of prenatal versus postnatal repair of myelomeningocele. N Engl J Med 364(11):993–1004
Johnson MP et al (2016) The management of myelomeningocele study: obstetrical outcomes and risk factors for obstetrical complications following prenatal surgery. Am J Obstet Gynecol
Mazzone L et al (2014) Experimental tissue engineering of fetal skin. Pediatr Surg Int 30(12):1241–1247
Pontiggia L et al (2009) Markers to evaluate the quality and self-renewing potential of engineered human skin substitutes in vitro and after transplantation. J Invest Dermatol 129(2):480–490
Braziulis E et al (2012) Modified plastic compression of collagen hydrogels provides an ideal matrix for clinically applicable skin substitutes. Tissue Eng Part C Methods 18(6):464–474
Meuli M et al (1995) In utero surgery rescues neurological function at birth in sheep with spina bifida. Nat Med 1(4):342–347
Meuli M et al (2013) Premiere use of integra artificial skin to close an extensive fetal skin defect during open in utero repair of myelomeningocele. Pediatr Surg Int 29(12):1321–1326
Meuli M, Moehrlen U (2014) Fetal surgery for myelomeningocele is effective: a critical look at the whys. Pediatr Surg Int 30(7):689–697
Mangels KJ et al (2000) Use of bipedicular advancement flaps for intrauterine closure of myeloschisis. Pediatr Neurosurg 32(1):52–56
Moldenhauer JS et al (2015) Fetal myelomeningocele repair: the post-MOMS experience at the Children’s Hospital of Philadelphia. Fetal Diagn Ther 37(3):235–240
Mazzola CA et al (2002) Dermoid inclusion cysts and early spinal cord tethering after fetal surgery for myelomeningocele. N Engl J Med 347(4):256–259
Candi E, Schmidt R, Melino G (2005) The cornified envelope: a model of cell death in the skin. Nat Rev Mol Cell Biol 6(4):328–340
Prunieras M, Regnier M, Woodley D (1983) Methods for cultivation of keratinocytes with an air–liquid interface. J Invest Dermatol 81(1 Suppl):28s–33s
Cartlidge P (2000) The epidermal barrier. Semin Neonatol 5(4):273–280
Evans NJ, Rutter N (1986) Development of the epidermis in the newborn. Biol Neonate 49(2):74–80
Burrington JD (1971) Wound healing in the fetal lamb. J Pediatr Surg 6(5):523–528
Acknowledgments
We thank an anonymous private sponsor, the Gottfried and Julia Bangerter-Rhyner Foundation, and the Children’s Research Center of the University Children’s Hospital Zurich, Switzerland, for their generous financial support and interest in our work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Mazzone, L., Pratsinis, M., Pontiggia, L. et al. Successful grafting of tissue-engineered fetal skin. Pediatr Surg Int 32, 1177–1182 (2016). https://doi.org/10.1007/s00383-016-3977-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00383-016-3977-z