Abstract
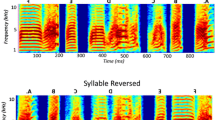
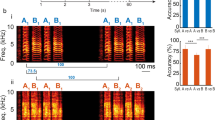
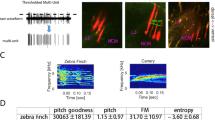
Bengalese finches (Lonchura striata var. domestica) generate more complex sequences in their songs than zebra finches. Because of this, we chose this species to explore the signal processing of sound sequence in the primary auditory forebrain area, field L, and in a secondary area, the caudomedial nidopallium (NCM). We simultaneously recorded activity from multiple single units in urethane-anesthetized birds. We successfully replicated the results of a previous study in awake zebra finches examining stimulus-specific habituation of NCM neurons to conspecific songs. Then, we used an oddball paradigm and compared the neural response to deviant sounds that were presented infrequently, with the response to standard sounds, which were presented frequently. In a single sound oddball task, two different song elements were assigned for the deviant and standard sounds. The response bias to deviant elements was larger in NCM than in field L. In a triplet sequence oddball task, two triplet sequences containing elements ABC and ACB were assigned as the deviant and standard. Only neurons in NCM that displayed broad-shaped spike waveforms had sensitivity to the difference in element order. Our results suggest the hierarchical processing of complex sound sequences in the songbird auditory forebrain.










Similar content being viewed by others
Abbreviations
- BOS:
-
Bird’s own song
- CLM:
-
Caudolateral mesopallium
- CMM:
-
Caudomedial mesopallium
- Dv:
-
Deviant stimulus
- IOI:
-
Inter-onset interval
- ISI:
-
Inter-spike interval
- NCM:
-
Caudomedial nidopallium
- RMS:
-
Root-mean-square
- St:
-
Standard stimulus
References
Anderson LA, Christianson GB, Linden JF (2009) Stimulus-specific adaptation occurs in the auditory thalamus. J Neurosci 29:7359–7363. doi:10.1523/JNEUROSCI.0793-09.2009
Astikainen P, Mällo T, Ruusuvirta T, Näätänen R (2014) Electrophysiological evidence for change detection in speech sound patterns by anesthetized rats. Front Neurosci 8:1–6. doi:10.3389/fnins.2014.00374
Beckers GJL, Gahr M (2010) Neural processing of short-term recurrence in songbird vocal communication. PLoS One 5:e11129. doi:10.1371/journal.pone.0011129
Beckers GJL, Gahr M (2012) Large-scale synchronized activity during vocal deviance detection in the zebra finch auditory forebrain. J Neurosci 32:10594–10608. doi:10.1523/JNEUROSCI.6045-11.2012
Berwick RC, Okanoya K, Beckers GJL, Bolhuis JJ (2011) Songs to syntax: the linguistics of birdsong. Trends Cogn Sci 15:113–121. doi:10.1016/j.tics.2011.01.002
Bolhuis JJ, Gahr M (2006) Neural mechanisms of birdsong memory. Nat Rev Neurosci 7:347–357. doi:10.1038/nrn1904
Bolhuis JJ, Okanoya K, Scharff C (2010) Twitter evolution: converging mechanisms in birdsong and human speech. Nat Rev Neurosci 11:747–759. doi:10.1038/nrn2931
Capsius B, Leppelsack HJ (1996) Influence of urethane anesthesia on neural processing in the auditory cortex analogue of a songbird. Hear Res 96:59–70. doi:10.1016/0378-5955(96)00038-X
Catchpole CK, Slater PJB (2008) Bird song: biological themes and variations, 2nd edn. Cambridge University Press, Cambridge
Chen J, ten Cate C (2014) Zebra finches can use positional and transitional cues to distinguish vocal element strings. Behav Processes 117:29–34. doi:10.1016/j.beproc.2014.09.004
Chew SJ, Mello C, Nottebohm F et al (1995) Decrements in auditory responses to a repeated conspecific song are long-lasting and require two periods of protein synthesis in the songbird forebrain. Proc Natl Acad Sci 92:3406–3410. doi:10.1073/pnas.92.8.3406
Chew SJ, Vicario DS, Nottebohm F (1996) A large-capacity memory system that recognizes the calls and songs of individual birds. Proc Natl Acad Sci 93:1950–1955. doi:10.1073/pnas.93.5.1950
Comins JA, Gentner TQ (2013) Perceptual categories enable pattern generalization in songbirds. Cognition 128:113–118. doi:10.1016/j.cognition.2013.03.014
Dong S, Clayton DF (2009) Habituation in songbirds. Neurobiol Learn Mem 92:183–188. doi:10.1016/j.nlm.2008.09.009
Doupe AJ (1997) Song- and order-selective neurons in the songbird anterior forebrain and their emergence during vocal development. J Neurosci 17:1147–1167
Fortune ES, Margoliash D (1992) Cytoarchitectonic organization and morphology of cells of the field L complex in male zebra finches (Taenopygia guttata). J Comp Neurol 325:388–404. doi:10.1002/cne.903250306
Gentner TQ, Hulse SH, Bentley GE, Ball GF (2000) Individual vocal recognition and the effect of partial lesions to HVc on discrimination, learning, and categorization of conspecific song in adult songbirds. J Neurobiol 42:117–133
Grace JA, Amin N, Singh NC, Theunissen FE (2003) Selectivity for conspecific song in the zebra finch auditory forebrain. J Neurophysiol 89:472–487. doi:10.1152/jn.00088.2002
Hahnloser RHR, Kotowicz A (2010) Auditory representations and memory in birdsong learning. Curr Opin Neurobiol 20:332–339. doi:10.1016/j.conb.2010.02.011
Kato Y, Kato M, Hasegawa T, Okanoya K (2010) Song memory in female birds: neuronal activation suggests phonological coding. Neuroreport 21:404–409. doi:10.1097/WNR.0b013e32833730b7
Kato Y, Kato M, Okanoya K (2012) Sequential information of self-produced song is represented in the auditory areas in male Bengalese finches. Neuroreport 23:488–492. doi:10.1097/WNR.0b013e32835375ef
Kojima S, Doupe AJ (2008) Neural encoding of auditory temporal context in a songbird basal ganglia nucleus, and its independence of birds’ song experience. Eur J Neurosci 27:1231–1244. doi:10.1111/j.1460-9568.2008.06083.x
Kozlov AS, Gentner TQ (2014) Central auditory neurons display flexible feature recombination functions. J Neurophysiol 111:1183–1189. doi:10.1152/jn.00637.2013
Kröner S, Güntürkün O (1999) Afferent and efferent connections of the caudolateral neostriatum in the pigeon (Columba livia): a retro- and anterograde pathway tracing study. J Comp Neurol 407:228–260. doi:10.1002/(SICI)1096-9861(19990503)407:2<228:AID-CNE6>3.0.CO;2-2
Lewicki MS, Arthur BJ (1996) Hierarchical organization of auditory temporal context sensitivity. J Neurosci 16:6987–6998
Lu K, Vicario DS (2014) Statistical learning of recurring sound patterns encodes auditory objects in songbird forebrain. Proc Natl Acad Sci USA 111:14553–14558. doi:10.1073/pnas.1412109111
Lynch KS, Kleitz-Nelson HK, Ball GF (2013) HVC lesions modify immediate early gene expression in auditory forebrain regions of femalesongbirds. Dev Neurobiol 73:315–323. doi:10.1002/dneu.22062
Malmierca MS, Cristaudo S, Pérez-González D, Covey E (2009) Stimulus-specific adaptation in the inferior colliculus of the anesthetized rat. J Neurosci 29:5483–5493. doi:10.1523/JNEUROSCI.4153-08.2009
Margoliash D, Fortune ES (1992) Temporal and harmonic combination-sensitive neurons in the zebra finch’s HVc. J Neurosci 12:4309–4326
Meliza C, Margoliash D (2012) Emergence of selectivity and tolerance in the avian auditory cortex. J Neurosci 32:15158–15168. doi:10.1523/JNEUROSCI.0845-12.2012
Meliza C, Chi Z, Margoliash D (2010) Representations of conspecific song by starling secondary forebrain auditory neurons: toward a hierarchical framework. J Neurophysiol 103:1195–1208. doi:10.1152/jn.00464.2009
Mello C, Nottebohm F, Clayton D (1995) Repeated exposure to one song leads to a rapid and persistent decline in an immediate early gene’s response to that song in zebra finch telencephalon. J Neurosci 15:6919–6925
Menardy F, Touiki K, Dutrieux G et al (2012) Social experience affects neuronal responses to male calls in adult female zebra finches. Eur J Neurosci 35:1322–1336. doi:10.1111/j.1460-9568.2012.08047.x
Miller-Sims VC, Bottjer SW (2014) Development of neural responsivity to vocal sounds in higher level auditory cortex of songbirds. J Neurophysiol 112:81–94. doi:10.1152/jn.00484.2013
Moore RC, Lee T, Theunissen FE (2013) Noise-invariant neurons in the avian auditory cortex: hearing the song in noise. PLoS Comput Biol 9:e1002942. doi:10.1371/journal.pcbi.1002942
Müller CM, Leppelsack H (1985) Feature extraction and tonotopic organization in the avian auditory forebrain. Exp Brain Res 59:587–599. doi:10.1007/BF00261351
Nakamura KZ, Okanoya K (2004) Neural correlates of song complexity in Bengalese finch high vocal center. Neuroreport 15:1359–1363. doi:10.1097/01.wnr.0000125782.35268.d6
Nelken I, Ulanovsky N (2007) Mismatch negativity and stimulus-specific adaptation in animal models. J Psychophysiol 21:214–223. doi:10.1027/0269-8803.21.34.214
Newport EL, Aslin RN (2004) Learning at a distance I. statistical learning of non-adjacent dependencies. Cogn Psychol 48:127–162. doi:10.1016/S0010-0285(03)00128-2
Nixdorf-Bergweiler BE, Bischof H-J (2007) A stereotaxic atlas of the brain of the zebra finch, Taeniopygia guttata. National Center for Biotechnology Information, Bethesda
Okanoya K (2004) The Bengalese finch: a window on the behavioral neurobiology of birdsong syntax. Ann N Y Acad Sci 1016:724–735. doi:10.1196/annals.1298.026
Okanoya K, Yamaguchi A (1997) Adult Bengalese finches (Lonchura striata var. domestica) require real-time auditory feedback to produce normal song syntax. J Neurobiol 33:343–356. doi:10.1002/(SICI)1097-4695
Okanoya K, Tsumaki S, Honda E (2000) Perception of temporal properties in self-generated songs by Bengalese Finches (Lonchura striata var. domestica). J Comp Psychol 114:239–245. doi:10.1037//0735
Ondracek JJM, Hahnloser RHR (2013) Advances in understanding the auditory brain of songbirds. In: Köppl C, Manley GA, Popper AN, Fay RR (eds) Insights from comparative hearing research. Springer, New York, pp 347–388
Perks KE, Gentner TQ (2015) Subthreshold membrane responses underlying sparse spiking to natural vocal signals in auditory cortex. Eur J Neurosci 41:725–733. doi:10.1111/ejn.12831
Pfenning AR, Hara E, Whitney O et al (2014) Convergent transcriptional specializations in the brains of humans and song-learning birds. Science 346:1256846. doi:10.1126/science.1256846
Phan ML, Pytte CL, Vicario DS (2006) Early auditory experience generates long-lasting memories that may subserve vocal learning in songbirds. Proc Natl Acad Sci USA 103:1088–1093. doi:10.1073/pnas.0510136103
Pinaud R, Terleph TA (2008) A songbird forebrain area potentially involved in auditory discrimination and memory formation. J Biosci 33:145–155
Saffran JR, Aslin RN, Newport EL (1996) Statistical learning by 8-month-old infants. Science 274:1926–1928. doi:10.1126/science.274.5294.1926
Schneider DM, Woolley SMN (2013) Sparse and background-invariant coding of vocalizations in auditory scenes. Neuron 79:141–152. doi:10.1016/j.neuron.2013.04.038
Seki Y, Suzuki K, Osawa AM, Okanoya K (2013) Songbirds and humans apply different strategies in a sound sequence discrimination task. Front Psychol 4:447. doi:10.3389/fpsyg.2013.00447
Smulders TV, Jarvis ED (2013) Different mechanisms are responsible for dishabituation of electrophysiological auditory responses to a change in acoustic identity than to a change in stimulus location. Neurobiol Learn Mem 106:163–176. doi:10.1016/j.nlm.2013.08.010
Stripling R, Volman SF, Clayton D (1997) Response modulation in the zebra finch neostriatum: relationship to nuclear gene regulation. J Neurosci 17:3883–3893
Stripling R, Kruse AA, Clayton DF (2001) Development of song responses in the zebra finch caudomedial neostriatum: role of genomic and electrophysiological activities. J Neurobiol 48:163–180
Terleph TA, Mello CV, Vicario DS (2006) Auditory topography and temporal response dynamics of canary caudal telencephalon. J Neurobiol 66:281–292. doi:10.1002/neu.20219
Theunissen FE, Shaevitz SS (2006) Auditory processing of vocal sounds in birds. Curr Opin Neurobiol 16:400–407. doi:10.1016/j.conb.2006.07.003
Theunissen FE, Amin N, Shaevitz SS et al (2004) Song selectivity in the song system and in the auditory forebrain. Ann N Y Acad Sci 1016:222–245. doi:10.1196/annals.1298.023
Ulanovsky N, Las L, Nelken I (2003) Processing of low-probability sounds by cortical neurons. Nat Neurosci 6:391–398. doi:10.1038/nn1032
Van Heijningen CAA, de Visser J, Zuidema W, ten Cate C (2009) Simple rules can explain discrimination of putative recursive syntactic structures by a songbird species. Proc Natl Acad Sci USA 106:20538–20543. doi:10.1073/pnas.0908113106
Vates GE, Broome BM, Mello CV, Nottebohm F (1996) Auditory pathways of caudal telencephalon and their relation to the song system of adult male zebra finches. J Comp Neurol 366:613–642. doi:10.1002/(SICI)1096-9861(19960318)366:4<613:AID-CNE5>3.0.CO;2-7
Veit L, Nieder A (2013) Abstract rule neurons in the endbrain support intelligent behaviour in corvid songbirds. Nat Commun 4:2878. doi:10.1038/ncomms3878
Woolley SMN (2013) The songbird auditory system. In: Helekar SA (ed) Animal Models of Speech and Language Disorders. Springer, New York, pp 61–88
Woolley SMN, Rubel EW (1997) Bengalese finches Lonchura Striata domestica depend upon auditory feedback for the maintenance of adult song. J Neurosci 17:6380–6390
Acknowledgments
We thank Dr. Jun Nishikawa for his instruction in the surgical techniques and the use of the data acquisition software. We also thank Dr. Ryosuke Tachibana for his helpful comments in the data analysis and discussion. This work was supported by Grant in Aids from MEXT #23240033, #25590202 and #26240019 to Kazuo Okanoya, and JST ERATO Okanoya Emotional Information Project. The experimental procedures and housing conditions were approved by the Institutional Animal Care and Use Committee at the University of Tokyo.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ono, S., Okanoya, K. & Seki, Y. Hierarchical emergence of sequence sensitivity in the songbird auditory forebrain. J Comp Physiol A 202, 163–183 (2016). https://doi.org/10.1007/s00359-016-1070-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00359-016-1070-7