Abstract
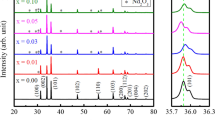
Structural transformations of Li-doped Zn1−x Li x O (0.10 ≤ x ≤ 0.70) which was synthesised by solid-state reaction were investigated. XRD carried out on powder specimens of the samples show that they are polycrystalline in nature with a hexagonal wurtzite structure having minor impurities. The result indicates the maximum limit of substitution of Zn atoms by Li is at x = 0.4. The lattice parameter a reduced from 3.01 to 2.99 Å, while c reduced from 5.21 to 5.19 Å. However, the Zn–O bond length reduced from 1.88 to 1.87 Å for the undoped, to x = 0.60 for the doped, respectively. The c/a ratio is 1.73 and is almost constant for all samples. The grain size of the (100) peak of the undoped ZnO is 41.73 nm and that of x = 0.10 is 41.76 nm. For x = 0.2–0.70, the grain size is 41.72 nm indicating that the grain size is almost independent of doping. The SEM results indicate a variation of grain size from 2.18 to 5.15 µm for the undoped ZnO to x = 0.50, which shows increase in grain size and reduction in grain boundaries as doping increases. The results show that x = 0.50 has the highest grain size and the one with the highest transition temperature is x = 0.6. DTA results indicate the structural phase transition temperature of the doped ZnO ranged from ~371 to ~409 K and increased as the amount of Li increases. A single arc is observed in all the impedance plots of the ZnO together with the presence of a relaxation process which is non-Debye. The impedance data show reduced resistance with increase in lithium content. A general increase in dielectric constant with increase in lithium content is observed.









Similar content being viewed by others
References
V. Bilgin, J. Electron. Mater. 88, 1969–1978 (2009)
A.H. Salama, J. Mater. Sci. Technol. 25, 314–315 (2009)
A. Janotti, Van de Walle, Rep. Progr. Phys. 72, 126501 (2009)
D.S. Singh, J. Nagaraju, S.B. Krupanidhi, J. Appl. Phys. A 88, 421–424 (2007)
S.H. Jeong, B.N. Park, S.-B. Lee, J.-H. Boo, J. Thin Solid Films 516, 5586–5589 (2007)
Y.L. Du, Y. Dengi, M.S. Zhang, Solid State Commun. J. 137, 78–81 (2005)
M.K.R. Khan, M.M. Rahman, I. Tanaka, Nucleus 39, 149–154 (2003)
M.K.R. Khan, M. Rahman, S.J. Mia, M. Shahajahan, Indian J. Pure Appl. Phys. 41, 211–216 (2003)
S. Polarz, A. Orlov, A. Hoffmann, M.R. Wagner, C. Rauch, R. Kirste, M. Lehmann, Chem. Matter 21, 3889–3897 (2009)
M. Ardyanian, N. Sedigh, Bull. Mater. Sci. 37, 1309–1314 (2014)
Y. Zeng, Z.Z. Ye, W.Z. Xu, L.I. Chen, D.Y. Li, L.P. Zhu, Y.L. Hu, J. Cryst. Growth 283(1), 180 (2005)
X.S. Wang, Z.C. Wu, J.F. Webb, Z.G. Liu, Appl. Phys. A 77, 561–565 (2003)
J.S. Kim, H.J. Lee, H.J. Seog, W. Kim, J. Korean Phys. Soc. 58, 640–644 (2011)
A. Onodera, N. Tamaki, K. Jin, H. Yamashita, Jpn. J. Appl. Phys. 36, 6008 (1997)
A. Sokiassian, A. Tganstsev, N. Seller, Appl. Phys. Lett. 07, 192903 (2010)
U. Ahmadu, T. Salkus, A.O. Musa, K.U. Isah, Open J. Phys. Chem. 1, 94–103 (2011)
Z. Zhou, K. Kato, T. Tomaki, M. Yoshino, H. Yukawa, M. Morinaga, K. Morita, J. Eur. Ceram. Soc. 24, 139–146 (2004)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ahmadu, U., Salaudeen, I.T. Structure, phase transition and impedance of Zn1−x Li x O (0.10 ≤ x ≤ 0.70) ceramic. Appl. Phys. A 122, 693 (2016). https://doi.org/10.1007/s00339-016-0213-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00339-016-0213-6